Alternatived Products of [ 98639-91-9 ]
Product Details of [ 98639-91-9 ]
| CAS No. : | 98639-91-9 |
MDL No. : | |
| Formula : |
C8H8N2OS
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
180.23
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 98639-91-9 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 98639-91-9 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 98639-91-9 ]
- 1
-
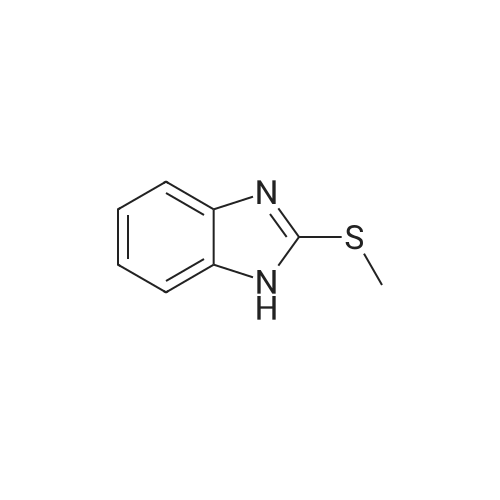 [ 7152-24-1 ]
[ 7152-24-1 ]

-
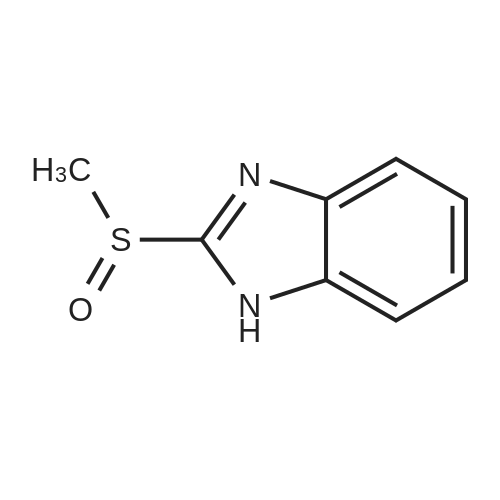 [ 98639-91-9 ]
[ 98639-91-9 ]
| Yield | Reaction Conditions | Operation in experiment |
| 85% |
With tert.-butylhydroperoxide; bis(acetylacetonate)oxovanadium; In decane; dichloromethane; at 20℃; |
t-BuOOH (solution 5.5 M in decane, 1 mL) and V0(acac)2 (5%, 0.25 mmol, 66 mg)were added in sequence to a solution of commercially available 2-(methylthio)-benzoimidazole (5 mmol, 820 mg) in CH2Cl2 (25 ml). The mixture was stirred at rt and monitored by thin-layer chromatography (TLC). At the end of there action, the solventwasremovedunderreducedpressureandthecrudewasdirectlypurifiedby flashchromatography (CHCl3/EtOAc) to obtain pure 1 as a white solid (765 mg, 4.2 mmol, 85% yield). |
Reference:
[1]Journal of Chemical Research,2005,p. 796 - 799
[2]Synthetic Communications,2015,vol. 45,p. 1783 - 1791
[3]Synthetic Communications,2005,vol. 35,p. 775 - 784
[4]Journal of the American Chemical Society,2004,vol. 126,p. 7800 - 7811
[5]Green Chemistry,2017,vol. 19,p. 4061 - 4066
[6]Journal of Organic Chemistry,1987,vol. 52,p. 4620 - 4622
[7]Organic Letters,2007,vol. 9,p. 2265 - 2268
[8]Bioorganic and Medicinal Chemistry Letters,2008,vol. 18,p. 1573 - 1576
[9]Russian Journal of Organic Chemistry,2011,vol. 47,p. 124 - 130
- 2
-
 [ 98639-91-9 ]
[ 98639-91-9 ]

-
 [ 7152-24-1 ]
[ 7152-24-1 ]
| Yield | Reaction Conditions | Operation in experiment |
| 99% |
With 1,3,5-trichloro-2,4,6-triazine; potassium iodide; In acetonitrile; at 20℃; for 0.25h; |
General procedure: To a flask containing a stirred mixture of sulfoxide (1 mmol) in acetonitrile (5 mL), TCT (1 mmol, 0.184 g) and KI (2.5 mmol, 0.42 g) were added. The mixture was stirred at room temperature and monitored by TLC. On completion of the reaction, H2O (10 mL) was added to the reaction mixture which was then extracted with EtOAc (4 × 5 mL) and the combined extracts were dried (MgSO4). The filtrate was evaporated and the corresponding sulfide was obtained as the sole product (Table 2). |
- 3
-
 [ 7152-24-1 ]
[ 7152-24-1 ]

-
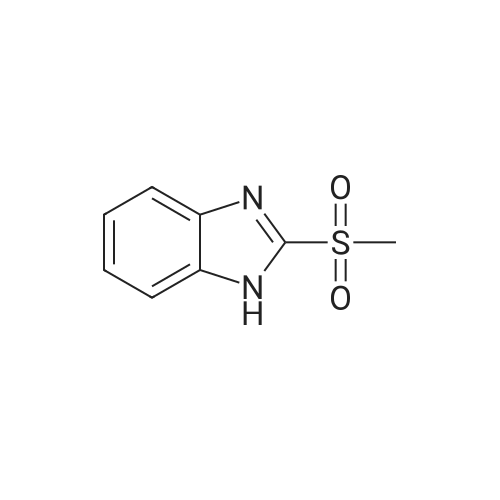 [ 57159-81-6 ]
[ 57159-81-6 ]

-
 [ 98639-91-9 ]
[ 98639-91-9 ]
- 4
-
 [ 98639-91-9 ]
[ 98639-91-9 ]

-
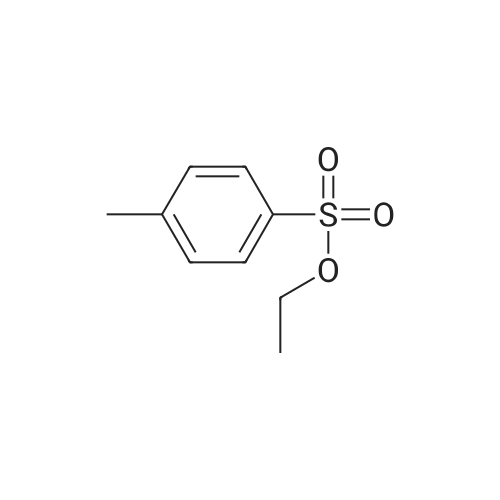 [ 80-40-0 ]
[ 80-40-0 ]

-
2-(methylsulfinyl)-N-ethyl-benzimidazole
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 86% |
|
Galvanostatic electrolysis was performed in a two-compartment cell [15] equipped with platinum spirals as cathode (apparent area ?1 cm2) and anode (apparent area ?0.5 cm2) using applied constant current I = 25 mA and total charge Q = 1.1 electrons per molecule 1. The catholyte starting material was 1 (18 mg, 0.1 mmol), CH3CN (0.3 ml), Et4NBF4 (21 mg, 0.1 mmol), and the anolyte was1-butyl-3-methylimidazolium tetrafluoroborate (0.3 ml). At the end of the electrolysis, MeI (0.11 mmol, 7 muL) was added to the cathode compartment and the reaction was prolonged at rt under magnetic stirring for 2 h. The mixture was then concentrated in vacuum and directly purified by flash chromatography (eluent mixture: hexane/AcEt) to obtain pure product 3a in 76% yield as an amorphous solid. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping






