Alternatived Products of [ 957111-25-0 ]
Product Details of [ 957111-25-0 ]
| CAS No. : | 957111-25-0 |
MDL No. : | MFCD09027269 |
| Formula : |
C48H30O2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
638.75
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 957111-25-0 ]
Application In Synthesis of [ 957111-25-0 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 957111-25-0 ]
- 1
-
 [ 1261302-60-6 ]
[ 1261302-60-6 ]

-
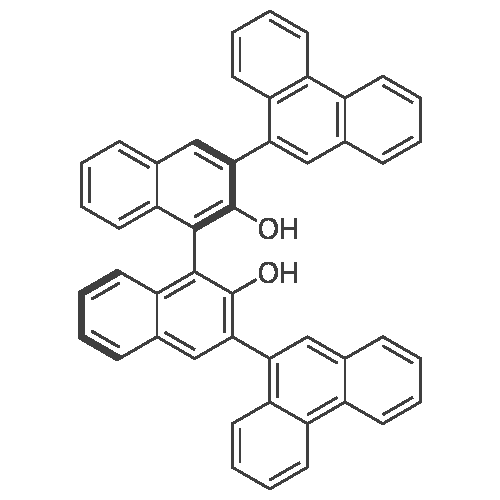 [ 1058734-56-7 ]
[ 1058734-56-7 ]
| Yield | Reaction Conditions | Operation in experiment |
| 99% |
With hydrogenchloride In 1,4-dioxane; water at 98 - 100℃; for 12h; |
|
| 94% |
With hydrogenchloride In 1,4-dioxane; water at 60℃; for 12h; |
|
|
With hydrogenchloride; water |
|
|
With hydrogenchloride In 1,4-dioxane; water |
|

Reference:
[1]Hu, Wenhao; Zhou, Jing; Xu, Xinfang; Liu, Weijun; Gong, Liuzhu
[Organic Syntheses, 2011, vol. 88, p. 406 - 417]
[2]Jia, Ji; Liu, Xiaoming; Ma, Si; Xia, Hong; Zhang, Yuwei; Zhang, Zhenwei
[Journal of Materials Chemistry A, 2021, vol. 9, # 45, p. 25369 - 25373]
[3]Location in patent: scheme or table
Rueping, Magnus; Sugiono, Erli; Schoepke, Fenja R.
[Synlett, 2010, # 6, p. 852 - 865]
[4]Location in patent: scheme or table
Rueping, Magnus; Nachtsheim, Boris J.; Koenigs, Rene M.; Ieawsuwan, Winai
[Chemistry - A European Journal, 2010, vol. 16, # 44, p. 13116 - 13126]
- 2
-
 [ 421-85-2 ]
[ 421-85-2 ]

-
 [ 1058734-56-7 ]
[ 1058734-56-7 ]

-
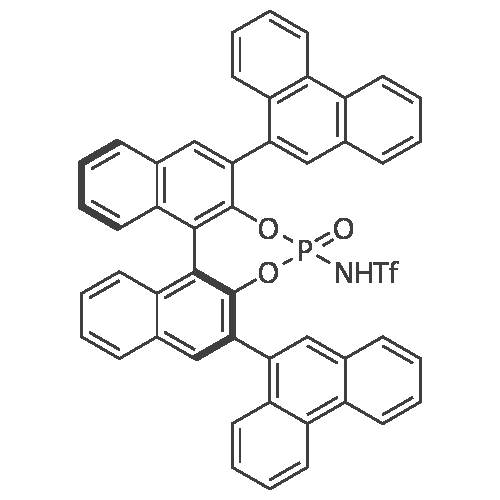 [ 1256334-83-4 ]
[ 1256334-83-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 54% |
Stage #1: (R)-3,3′-di-9-phenanthrenyl-1,1′-bi-2-naphthol With N,N-dimethyl-4-aminopyridine; triethylamine In dichloromethane at 0℃; for 0.166667h;
Stage #2: With trichlorophosphate In dichloromethane at 20℃; for 1.083h;
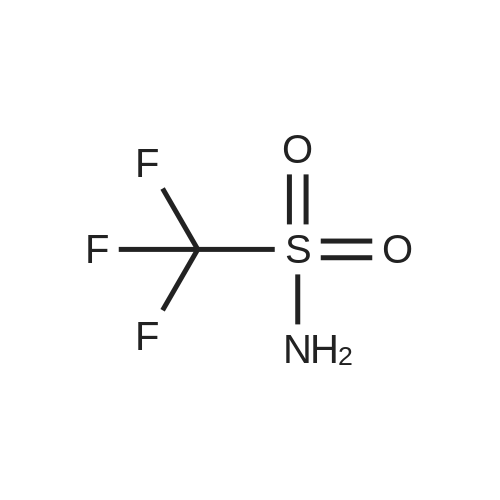
Stage #3: Trifluoromethanesulfonamide In dichloromethane; acetonitrile for 12h; Reflux; |
|
| 42% |
Stage #1: (R)-3,3′-di-9-phenanthrenyl-1,1′-bi-2-naphthol With N,N-dimethyl-4-aminopyridine; triethylamine; trichlorophosphate In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere;
Stage #2: Trifluoromethanesulfonamide In dichloromethane; acetonitrile for 12h; Inert atmosphere; Reflux; |
|
|
Stage #1: (R)-3,3′-di-9-phenanthrenyl-1,1′-bi-2-naphthol With N,N-dimethyl-4-aminopyridine; triethylamine; trichlorophosphate In dichloromethane
Stage #2: Trifluoromethanesulfonamide With propiononitrile Reflux; |
|
Reference:
[1]Cheng, Aolin; Li, Yingkun; Ma, Jiguo; Wang, Xinxu; Zhang, Yi; Zhao, Baoguo; Zhao, Guoqing; Zhao, Pengyuan
[Chinese Journal of Chemistry, 2020, vol. 38, # 6, p. 565 - 569]
[2]Momiyama, Norie; Jongwohan, Chanantida; Ohtsuka, Naoya; Chaibuth, Pawittra; Fujinami, Takeshi; Adachi, Kiyohiro; Suzuki, Toshiyasu
[Journal of Organic Chemistry, 2022, vol. 87, # 14, p. 9399 - 9407]
[3]Jolit, Anais; Dickinson, Cody F.; Kitamura, Kei; Walleser, Patrick M.; Yap, Glenn P. A.; Tius, Marcus A.
[European Journal of Organic Chemistry, 2017, vol. 2017, # 40, p. 6067 - 6076]
- 3
-
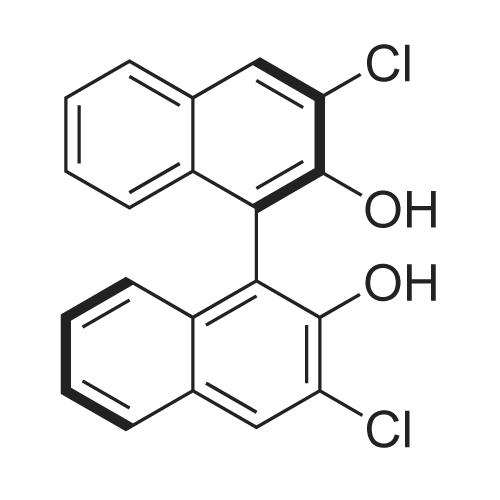 [ 287111-92-6 ]
[ 287111-92-6 ]

-
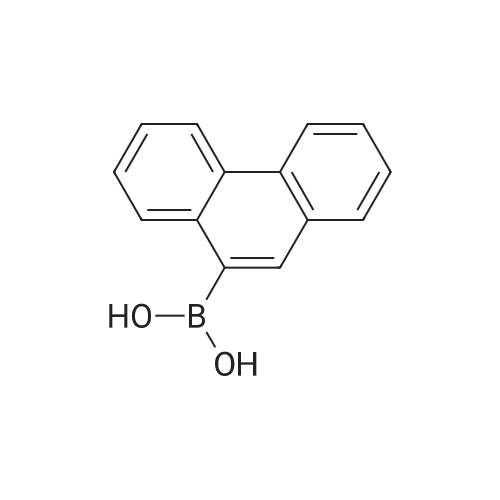 [ 68572-87-2 ]
[ 68572-87-2 ]

-
 [ 1058734-56-7 ]
[ 1058734-56-7 ]
| Yield | Reaction Conditions | Operation in experiment |
| 2.1 g |
With bis-triphenylphosphine-palladium(II) chloride; sodium carbonate In water; N,N-dimethyl-formamide at 90℃; |
2.S2
S2. After the completion of the reaction monitored by TLC, the intermediate I was subjected to a Suzuki coupling reaction. The specific steps are: using the intermediate I and (4-(phenanthrene-9-yl)phenyl)boronic acid as raw materials, In an alkaline environment (with Na2CO3 as the base), a mixed solution of DMF and water in a weight ratio of 1:0.4 is used as a solvent, under the catalysis of a palladium catalyst (Ph3P)2PdCl2, react at 90°C for 6h, 2.1 g of intermediate II was obtained, Among them, the feed ratio of intermediate I, (4-(phenanthrene-9-yl)phenyl)boronic acid, (Ph3P)2PdCl2, Na2CO3, and solvent is 1g:0.5g:0.04g:0.01g:15ml; |
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping











