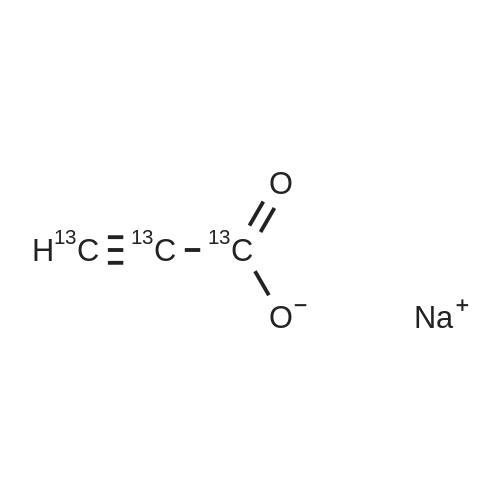
|
With hydrogen; In methanol; at 20℃; for 1h; |
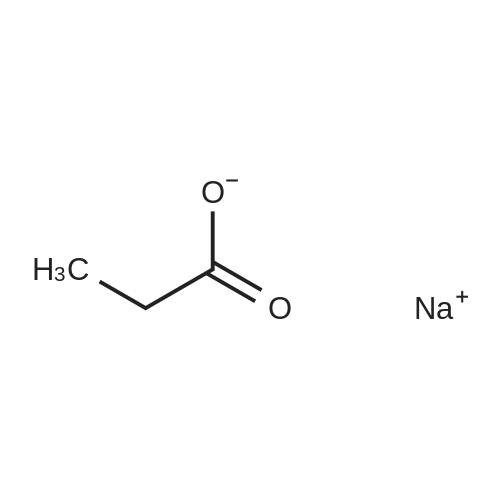
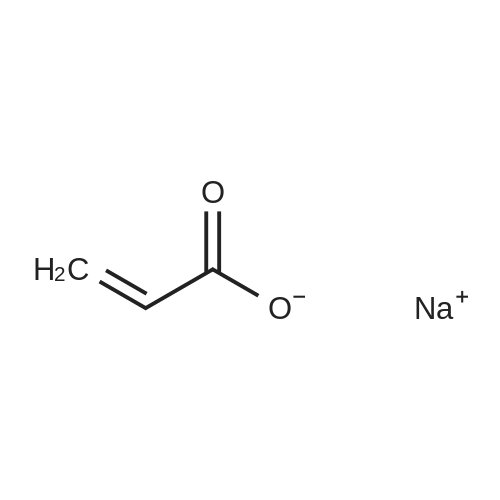
Survey Reactions with Hydrogen[0057]Prior to conducting reactions using deuterium, a series of survey reactions were herein performed using hydrogen gas to determine the optimum reaction conditions that would maximize the yield of non-deuterated sodium acrylate (4), while minimizing the amount of sodium propiolate (2-Na) remaining and the amount of sodium propionate (5) produced. Experimental details are provided below.[0058]In a typical procedure, a 250-mL round bottom flask was charged with sodium propiolate (2-Na, 1.15 g, 12.5 mmol), the appropriate Lindlar catalyst (e.g., 200 mg, 5 wt % Pd(0) on CaCO3 with 3 wt % of PbCO3), hydroquinone (10 mg), and a Teflon-coated stir bar. The appropriate solvent or solvent mixture (40 mL) was added and the solution stirred to dissolve the propiolate. The desired amount of quinoline (0 wt %, 50 wt %, 250 wt %, or 500 wt % of the catalyst, depending on the experiment) was then added via syringe. The flask was sealed with a rubber septum, sealed with parafilm, and evacuated and N2 back-filled three times by means of a needle attached to a double-manifold vacuum line. A plastic 5-mL syringe barrel with the large end cut off was fitted with two He-quality latex balloons (one balloon inserted inside a second balloon), using rubber bands and parafilm. The balloon assembly was purged with nitrogen at least three times. For reactions involving NO nitrogen blanketing atmosphere, the reaction flask was evacuated one more time. The balloon assembly was evacuated, filled with ca. 600-800 mL of hydrogen gas (>2-fold excess), and the syringe barrel fitted with a needle that was then inserted into the septum of the reaction flask. The solution was stirred at room temperature for up to 24 hours, with a small amount (?0.2 mL) of solution removed periodically by syringe. Those aliquots were diluted with 0.4 mL of D2O for analysis by 1H and 13C NMR. For reactions involving nominally 1 atm nitrogen blanketing atmosphere, the syringe barrel of the evacuated/deflated balloon assembly was fitted with a needle that was inserted through the septum of the nitrogen-filled reaction flask. An amount of hydrogen corresponding to a total of 13.3±0.3 mmol (the uncertainty being due to the day-to-day variance in lab temperature and pressure) was syringed into the reaction flask through the septum, using a 100-mL gas-tight syringe equipped with a valve, upon which the balloon was observed to inflate. The solution was stirred at room temperature for about 5 hours. A small amount (?0.2 mL) of solution was removed by syringe and diluted with 0.4 mL of D2O for 1H and 13C NMR at reaction times of 1, 2, and 3 hours. After 5 hours of stirring, (the balloon was completely deflated, indicating that the hydrogen had been consumed), the solution was filtered to remove the catalyst, and washed (to remove quinoline) with 5 mL×3 of CH3OH. The combined washing and reaction solvent was removed by rotary evaporation, and the solid that remained was suspended in 50 mL of ether and stirred for 10 minutes. The solid was then filtered, washed with additional ether (10 mL×3), and dried under vacuum to afford generally 1.10 to 1.15 g of a grey solid. 13C{1H} NMR (no NOE, see above) was used to determine the final distribution of sodium propiolate (2-Na), sodium acrylate (4), and sodium propionate (5).[0059]It was previously reported that treatment of sodium propiolate in 0.1 M NaOH at 40 C. with hydrogen over Pt-black (L. D. Volkova, et al., React. Kinet. Catal. Lett. 1985, 29, 345-351) could produce sodium acrylate. On the basis of this work, the partial reduction of sodium propiolate using the Lindlar catalyst (200 mg per 1.15 g of 2-Na) was herein first investigated in 0.05 M NaOH. However, as shown in Table 1 below, after 24 hours of stirring with a ?2-fold excess of hydrogen, only unreacted sodium propiolate (2-Na) was observed in the NMR spectrum.Switching to a 1:1 mixture of 0.05 M NaOH:CH3OH also resulted in only unreacted 2-H after 24 hours, and no reaction was also observed when the solvent was deionized water (2 hours). However, when pure CH3OH was used as the solvent, in the presence of a ?2-fold excess of hydrogen, complete over-reduction to sodium propionate (5) was observed within one hour. In this case, the balloon was completely deflated and, due to the excessive consumption of hydrogen, the flask was under negative pressure. This reaction was then repeated with the addition of 1.0 g of quinoline (500% on the weight of the catalyst), which is known to retard the over-reduction reaction. After 1 hour, a distribution ratio of 21:75:4 of 2-Na:4:5 was observed.[0061]In those first experiments, the reaction flask was placed under partial vacuum (evacuated and nitrogen refilled three times, then evacuated again until the solvent bubbled), and a balloon containing the ca. 2-fold excess hydrogen gas was attached to the flask. To better control the reaction and prevent over-reduction, in subsequent reactions a gas t... |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping