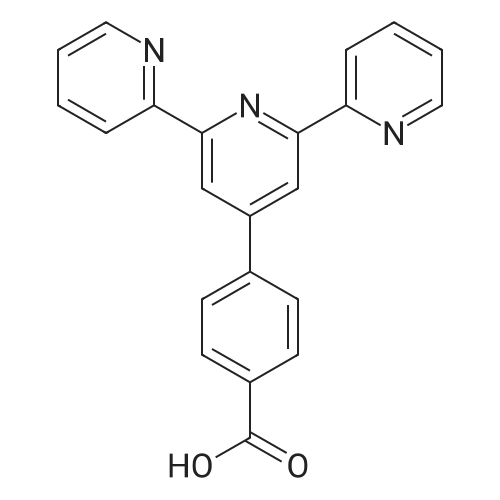
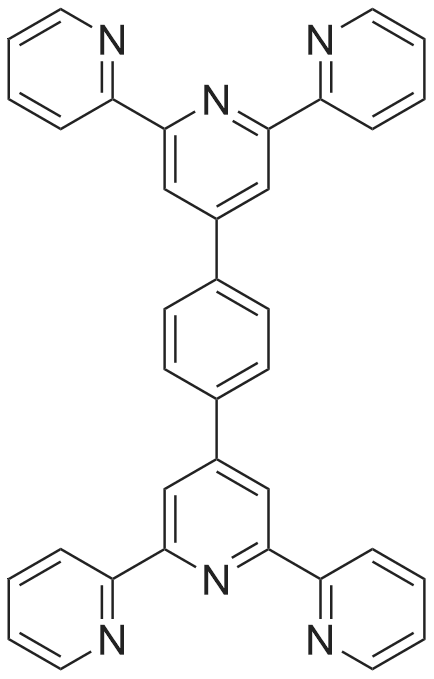
| 77.7% |
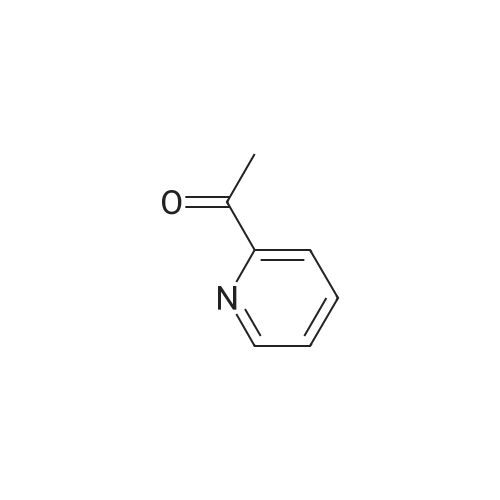
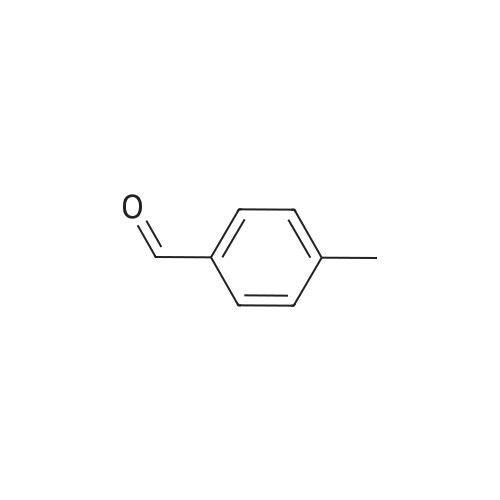
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide for 0.5h;
Stage #2: With ammonium acetate; acetic acid for 2h; Reflux; |
|
| 74% |
With ammonium hydroxide; potassium hydroxide In ethanol; water at 34℃; for 24h; |
|
| 72% |
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide for 0.666667h;
Stage #2: With ammonium acetate In acetic acid for 3h; Heating; |
|
| 69.8% |
With ammonia In ethanol for 36h; Alkaline conditions; |
Synthesis of tpy-PhCH3
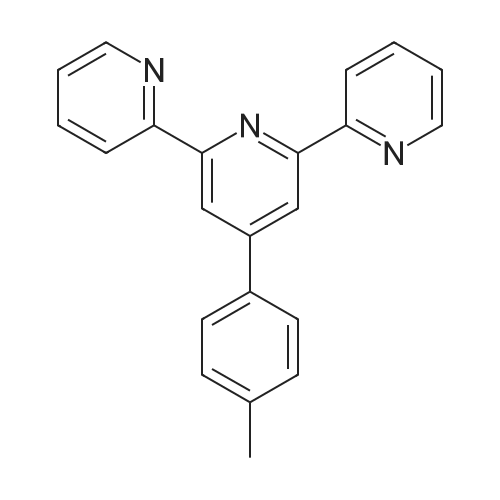
Synthesis of tpy-PhCH3[36]: A mixture of 4-methylbenzaldehyde (2.40 g, 0.02 mol), 2-acetylpyridine (4.85 g,0.04 mol),potassium hydroxide(3.08 g, 0.55 mol) and concentrated ammonia (50 mL, 25%) in anhydrous ethanol(100 mL) was stirred at room temperature under air atmosphere for 24 h. Other concentrated ammonia (10 mL 25%)was added and stirred at 60C for another 12 h. Theprecipitatewasseparatedby centrifugation,washedthree times withdeionizedwater(5 mL) and anhydrous ethanol(5 mL), andcrystallization from ethanol to yield tpy-PhCH3 (4.51 g,69.8% yield) as awhite solid.1H NMR (CDCl3):d(ppm) 2.43(s, 3H), 7.32 (dd, 2H, JD1.2 Hz, JD2.2 Hz), 7.34 (t, 2H, JD2.4 Hz), 7.85 (t, 2H, JD2.0 Hz), 7.86 (d, 2H, JD1.6 Hz),8.68 (d, 2H, JD8.0 Hz), 8.74 (d, 2H, JD0.8 Hz), 8.75 (s,2H). ESI-MS (positive): m/z 324.4 (100%) |
| 63% |
With ammonia; sodium hydroxide In ethanol; water at 34℃; for 24h; |
|
| 63% |
With ammonium hydroxide; sodium hydroxide In ethanol at 35℃; for 24h; |
|
| 62% |
With ammonia; sodium hydroxide In water at 34℃; for 24h; |
|
| 62% |
With ammonium hydroxide; sodium hydroxide In ethanol at 34℃; for 24h; |
|
| 60% |
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide at 20℃; for 2.16h;
Stage #2: With ammonia |
Methyl phenyl terpyridine [1] (MPT)
Methyl phenyl terpyridine [1] (MPT) was prepared using Krohnke methodology by condensation of 2-acetyl pyridine and p-tolualdehyde in 60% yield. 1.65 g (41 mmol) of crushed NaOH was added to the 30 ml of Polyethylene Glycol (PEG-300) with constant stirring at room temperature. 5 g (41.6 mmol) of 2-acetyl pyridine was added and after 10 minutes 2.472 g (20.6 mmol) of p- tolualdehyde was added with continuous stirring. The colorless solution turned to light yellow. After two hours the color changed to orange and then to pink. 25 ml of liquid NH3(18N) was added. The white precipitate formed was washed with water and recrystallized from ethanol. Yield %- 60%, HNMR-(CDCl3, 400 MHz, ppm) 2.4 (s, 3 H) d7.49 (m,2 H), 8.01 (m, 2 H), 8.15 (m, 4H), 8.72 (d,2 H), 8.80 (d, 2 H), 8.98 (s, 2 H),.5, UV-Vis- 281 nm |
| 60% |
With ammonium hydroxide; potassium hydroxide In ethanol at 20℃; for 24h; Inert atmosphere; Schlenk technique; |
|
| 60% |
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With potassium hydroxide In ethanol for 0.0833333h;
Stage #2: With ammonium hydroxide In ethanol at 28℃; for 12h; |
|
| 60% |
With ammonium hydroxide; potassium hydroxide In ethanol at 20℃; |
|
| 53.4% |
Stage #1: 2-acetylpyridine With potassium hydroxide In ethanol; water at 20℃; for 0.5h;
Stage #2: 4-methyl-benzaldehyde In ethanol; water at 20℃; for 0.333333h;
Stage #3: With ammonium hydroxide In ethanol; water at 50℃; for 24h; |
|
| 49% |
With potassium hydroxide; ammonium hydroxide In ethanol at 20℃; for 2h; |
|
| 49% |
With ammonium hydroxide; potassium hydroxide In ethanol at 20℃; for 12h; |
|
| 48% |
With ammonia; potassium hydroxide In ethanol; water for 36h; Reflux; |
|
| 46% |
With potassium hydroxide; ammonium hydroxide In methanol for 72h; |
|
| 46% |
With ammonia; sodium hydroxide In ethanol; water at 35℃; for 24h; |
|
| 40% |
With ammonium hydroxide; sodium hydroxide In methanol; water for 12h; Reflux; |
|
| 40% |
With ammonium hydroxide; sodium hydroxide In methanol for 12h; Reflux; |
|
| 40% |
With ammonium hydroxide; potassium hydroxide In ethanol at 80℃; |
4.3.1. Synthesis of 4-tolyl-tpy ligand
The 4-tolyl-tpy ligand was synthesized as per the reportedprocedure. To the ethanolic solution of 2-acetyl pyridine (4.84 g,40 mmol) and 4-methyl-benzaldehyde (2.4 g, 20 mmol) taken in around bottom flask, potassium hydroxide (2.5 g, 44.55 mmol) wasadded followed by 80 ml ammonia solution. The reaction mixturewas refluxed overnight at 80 °C, and a yellow colored precipitatewas obtained which was filtered and washed several times withpurified water to make it free from alkali followed by washing withdiethyl ether (3 x 5 mL). The light yellow precipitate was recrystallizedin dichloromethane to give yellow crystalline solid (4.8 g,14.8 mmol, yield = 40% based on 2-acetyl pyridine). ESI-MScalculated for [M+H]+, i.e. [(C22H17N3)+H+ = 324.15 and foundm/z 324.14. 1H NMR 400 MHz {δ, (J, Hz), CDCl3}: 8.73 (m, 4H), 8.66(d, 8.8, 2H), 7.88-7.81 (m, 4H), 7.35e7.30 (m, 4H), 2.42 (s, 3H). 13CNMR (100 MHz, CDCl3) δ 156.34, 155.84, 150.18, 149.09, 139.10,136.88, 135.47, 129.66, 127.17, 123.78, 121.39, 118.65, 21.29. |
| 40% |
With ammonium acetate In neat (no solvent) at 120℃; for 1h; |
|
| 39% |
With ammonia; potassium hydroxide In ethanol; water at 20℃; for 8h; |
2.3.1. Synthesis of 4'-(4-chlorophenyl)-2,2':6',2''-terpyridine (4-cptpy)
General procedure: 2-Acetylpyridine (2.42 g, 20.0 mmol) was added to 70 mL ethanolic solution of 4-chlorobenzaldehyde (1.4 g, 10.0 mmol). KOH pellets (1.4 g, 26 mmol) and aqueous NH3 (30 mL, 25%, 0.425 mol) were added to the solution and was then stirred at room temperature for 8 h (Scheme 1). An off-white solid was formed which was collected by filtration and washed with H2O (3 x 10 mL) and ethanol (2 x 5 mL). Recrystallization from CHCl3-MeOH gave white crystalline solid. Yield: 1.48 g, 43% |
| 39% |
With ammonia; sodium hydroxide In ethanol; water at 20℃; for 4h; |
|
| 33% |
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide In methanol; water at -10 - 20℃;
Stage #2: With ammonium acetate In methanol; water for 24h; Reflux; |
|
| 24% |
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With acetamide; ammonium acetate for 2h; Reflux;
Stage #2: With sodium hydroxide In water for 2h; Reflux; |
|
| 20% |
With acetamide; ammonium acetate at 160℃; for 2h; |
2.2.1. Synthesis of L1 and L2
General procedure: The syntheses of both L1 and L2 were based on literature procedure[24,37,38]. A mixture of acetamide (36.6 g, 0.6 mmol), ammoniumacetate (23.6 g, 0.3 mmol), p-tolualdehyde (2.48 g,20.6 mmol) and 2-acetylpyridine (5 g, 41.2 mmol) were heated at160 C with stirring for 2 h in 250 ml round-bottom flask, thenthe resulting solution was cooled to 120 C, and the aqueoussodium hydroxide (10% solution, 20 ml) was added over 20 minwith heating to 120 C for 2 h. The black brown paste was formedwhen the solution was cooled to room temperature and filteredout, and then washed with water and cool C2H5OH. The obtainedsolid was dissolved into CHCl3 and separated using column chromatography(Al2O3, n-hexane: ethyl acetate = 8:1, v/v), rotaryevaporating and the white crystalline solid L1 and L2 wereobtained, individually. The structure of L1 and L2 are shown inScheme 1. L1 1.9 g, 20%, 1H NMR (400 MHz, CDCl3, 300 K): d 8.78 (2H, s),8.73 (2H, m), 8.69(2H, d), 7.88 (2H, td), 7.84 (2H, d), 7.39 (2H,dd), 7.36 (2H, d), 2.43 (3H, s) (Fig. S1A). Selected FT-IR (KBr, m,cm1): 1584, 1466, 1392, 1113, 789, 619, 504. ESI-MS: m/z324.15 for [L1 + H]+. |
| 20% |
With ammonium hydroxide; potassium hydroxide In methanol; water for 48h; Reflux; |
Synthesis of 4′-p-tolyl-2,2′; 6′,2″-terpyridine (ttpy).
Synthesis of this ligand resembledpreviously reported procedures (35, 36); however, these procedureswere found to yield unsatisfactory purity considering the application. In ourhands, 2-acetylpyridine (7.83 g, 0.065 mol and KOH aqueous solution (5 mL,15 wt %) were stirred briefly in methanol (60 mL) at room temperature (∼5min). p-tolualdehyde (3.56 g, 0.03 mol) and concentrated ammonium hydroxide(25 mL) were then added and the mixture heated to reflux withvigorous stirring for 48 h. After cooling, the reaction mixture was decantedinto a large separatory funnel. To this was added 600 mL of water and600 mL of dichloromethane. After agitation, the dichloromethane layer wasremoved, washed once more with 600 mL water, then separated and driedover sodium sulfate. The dichloromethane was removed by distillation andthe remaining residue was recrystallized from 95:5 ethanol/water to yield2.45 g of slightly impure material. High purity was achieved by flash chromatographyusing 688 g of alumina, previously deactivated by thoroughmixing with water (5% by mass) and using toluene as the mobile phase.Yield = 1.91 g (20%); 1H NMR (600 MHz, d6-DMSO) δ ppm: 2.40 (s, 3H, Htolyl),7.40 (d, J = 8.0 Hz, 2H, Htolyl 3,5), 7.53 (dd, J = 7.5 Hz, J = 5.0 Hz, 2H, H5,5′′),7.83 (d, J =8.0 Hz, 2H, Htolyl 2,6), 8.04 (t, J = 7.5 Hz, 2H, H4,4″), 8.67 (d, J = 8.0Hz, 2H, H3,3′′), 8.70 (s, 2H, H3′,5′), 8.76 (d, J = 5.0 Hz, 2H, H6,6″). ESI(+)MS found(calcd) for C22H17N3H: 324.1511 (324.1501). |
| 19% |
Multistep reaction.; |
|
|
With acetamide; sodium hydroxide; ammonium acetate; dihydrogen peroxide; iron(III) 1.) reflux, 4 h; Yield given; Multistep reaction; |
|
|
With ammonia; potassium hydroxide In ethanol; water Reflux; |
|
|
With ammonia; potassium hydroxide In ethanol; water at 20℃; for 8h; |
2.3. Synthesis of ligands
General procedure: All tridentate ligands were synthesized similarly by following literature procedure [24]. 2-Acetyl pyridine (20.0 mmol) was added to an ethanolic solution of various aldehydes (10.0 mmol in 70 mL EtOH). KOH pellets (26 mmol) and aqueous NH3 (25%, 0.425 mol) were added to the solution and was stirred at room temperature for 8 h. An off-white solid formed which was collected by filtration, followed by washings with H2O (3 x 10 mL) and EtOH (2 x 5 mL). Crystallization from CHCl3-MeOH system gives a white crystalline solid. The proposed reaction is shown in Scheme 1. |
|
With ammonia In ethanol at 20℃; for 8h; Alkaline conditions; |
|
|
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With potassium <i>tert</i>-butylate In tetrahydrofuran
Stage #2: With ammonium acetate In tetrahydrofuran |
|
|
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide In ethanol
Stage #2: With ammonium hydroxide |
|
|
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With potassium <i>tert</i>-butylate In tetrahydrofuran at 20℃; Inert atmosphere;
Stage #2: With ammonium acetate In tetrahydrofuran; methanol for 4h; Reflux; Inert atmosphere; |
|
| 0.5% |
With ammonium hydroxide; potassium hydroxide In ethanol at 20℃; for 48h; |
|
|
With ammonium hydroxide; sodium hydroxide In ethanol at 34℃; for 24h; |
|
|
With ammonium hydroxide; sodium hydroxide In methanol for 6h; Reflux; |
|
|
With ammonia; potassium hydroxide In ethanol at 20℃; |
Preparation of ligands L1-L6
General procedure: The one-pot preparation method of Krohnke type 4'-aryl-2,2':6',2''-terpyridine ligands was performed to obtainligands L1-L5 according to the literature [23] with aslight modification. 2-Acetylpyridine (0.56 cm3,5.0 mmol) was added to a solution of 0.25 cm3 benzaldehyde(2.5 mmol) in 18 cm3 ethanol. After addition of the mixture of 0.280 g KOH (5.0 mmol) and 0.5 cm3 NH3(25 %, 6.5 mmol), the solution was stirred overnight at room temperature, during which time as orange suspension was appeared. The solid was collected by filtration and washed with EtOH (3 9 6 cm3). Then, the crude solid product was recrystallized by cooling the hot super saturated ethanolic solution. The preparation method of diketone ligand (L6) was performed similar to the above procedure for the preparation of substituted phenyl terpyridines without the addition of ammonia. The prepared ligand was characterized in good agreement with the literature [24]. |
|
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With sodium hydroxide In ethanol
Stage #2: With ammonium hydroxide |
|
|
With ammonium hydroxide; potassium hydroxide In ethanol for 38h; Reflux; |
|
|
Stage #1: 2-acetylpyridine; 4-methyl-benzaldehyde With potassium <i>tert</i>-butylate In tetrahydrofuran at 20℃; for 16h;
Stage #2: With ammonium acetate In ethanol Reflux; |
|
|
Alkaline conditions; |
|
|
With ammonium hydroxide; sodium hydroxide In ethanol |
|
|
With ammonium hydroxide; sodium hydroxide In ethanol at 25℃; for 12h; |
1 Example 1 Preparation of benzylamine terpyridine
Weigh p-tolualdehyde (1 mmol), 2-acetylpyridine (4 mmol), sodium hydroxide (4.2 mmol), ammonia water (3 mmol) into the reaction bottle, add ethanol, and magnetically stir at 25 ° C for 12 hours. After the reaction, the precipitate was filtered, and the recrystallized product obtained by recrystallization with ethanol was p-methylphenyl terpyridine. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping