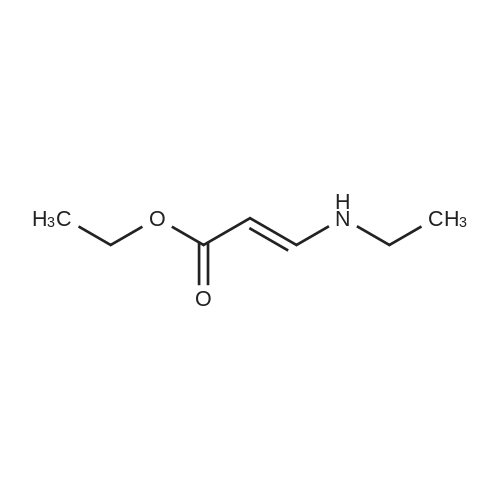
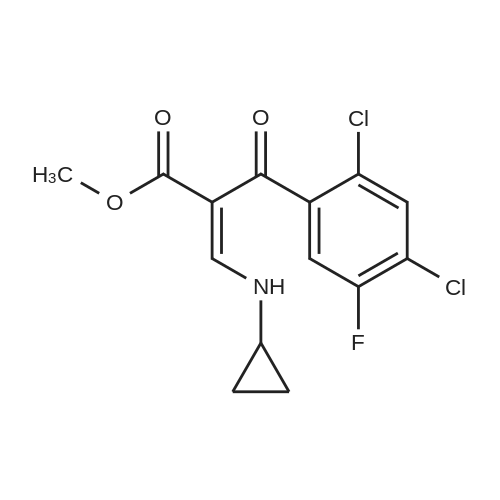
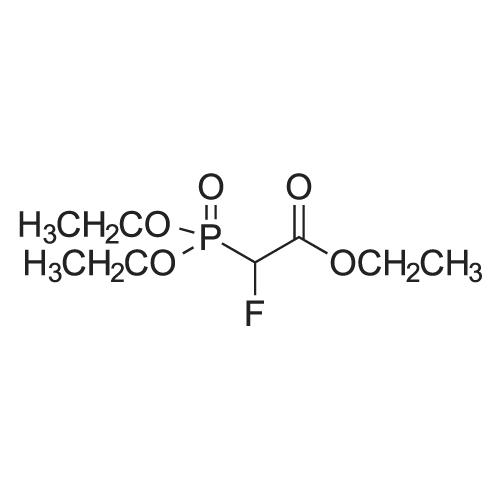
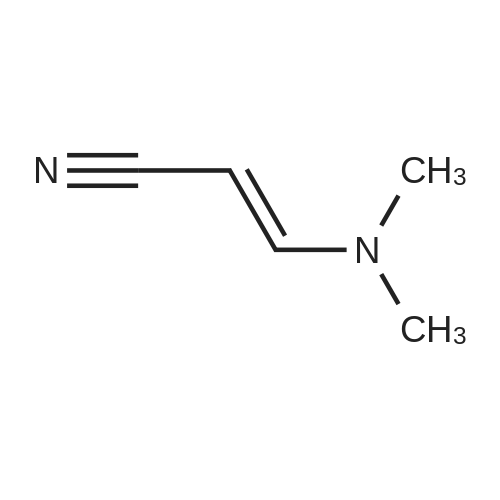
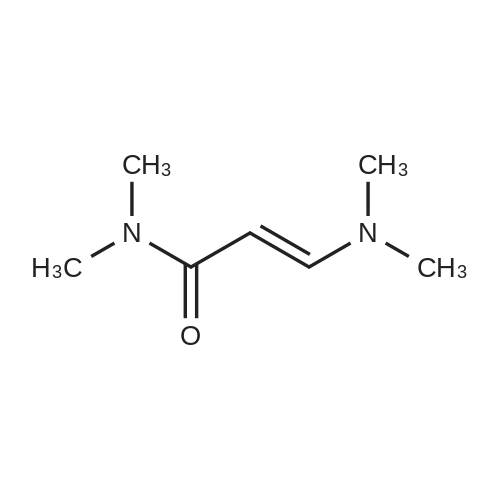
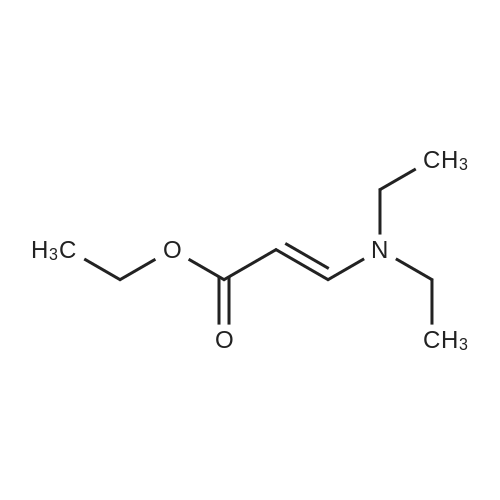
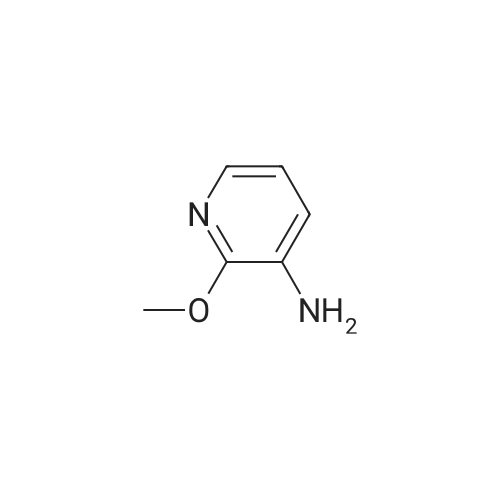
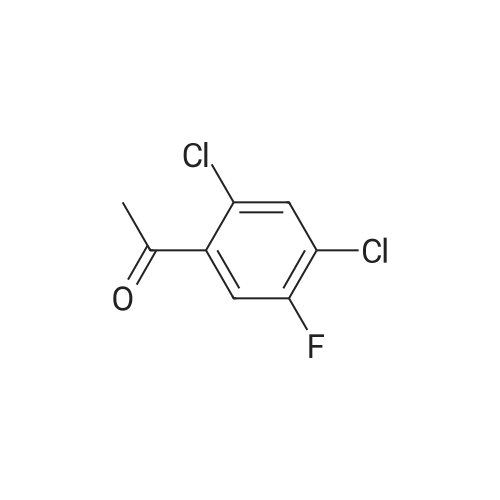
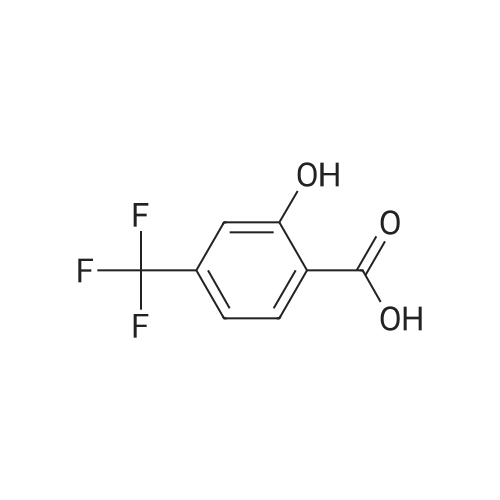
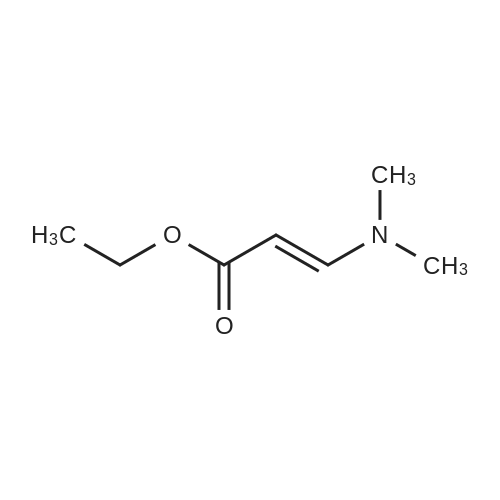
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
24.b EXAMPLE 24 STR31
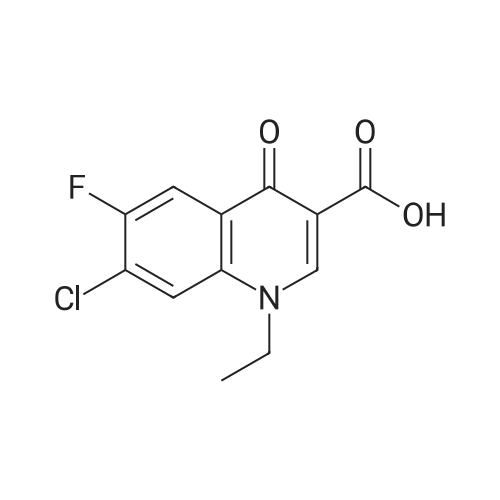
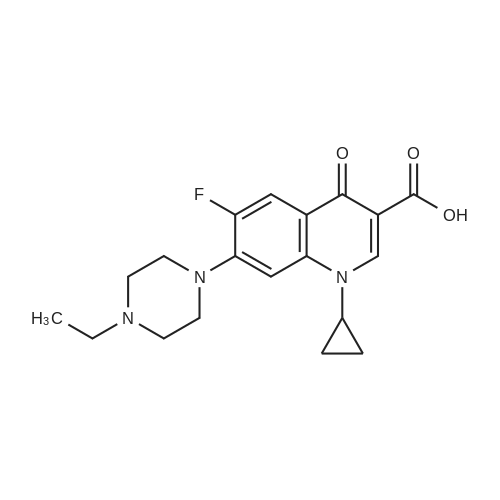
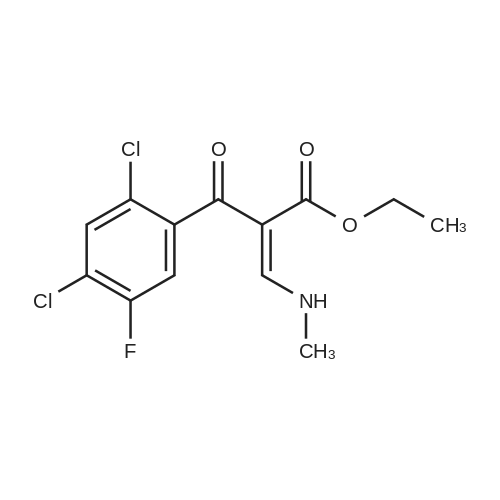
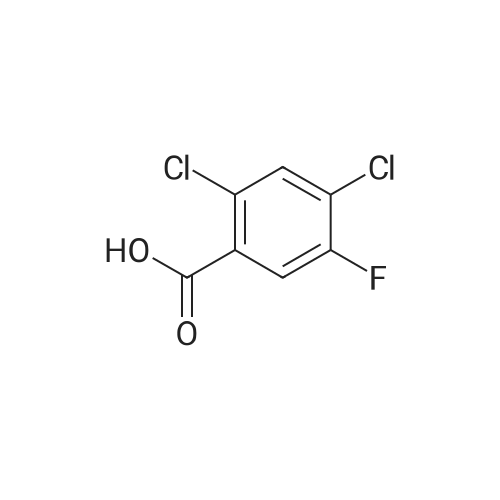
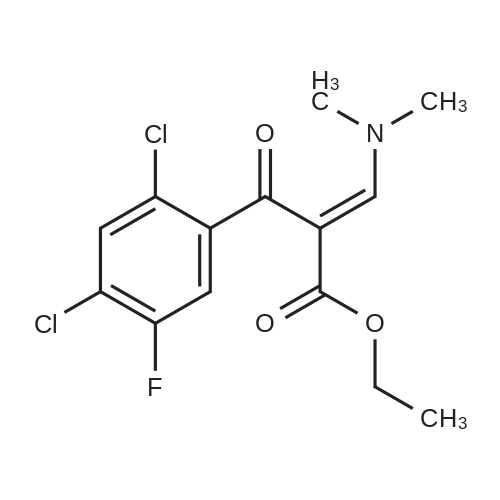
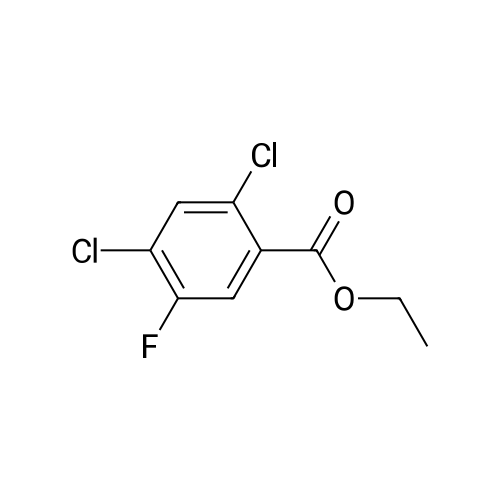
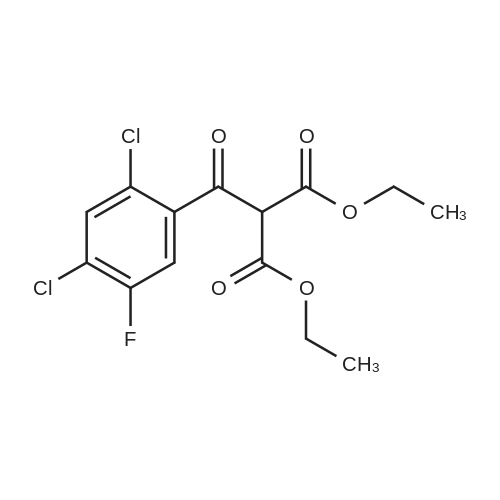
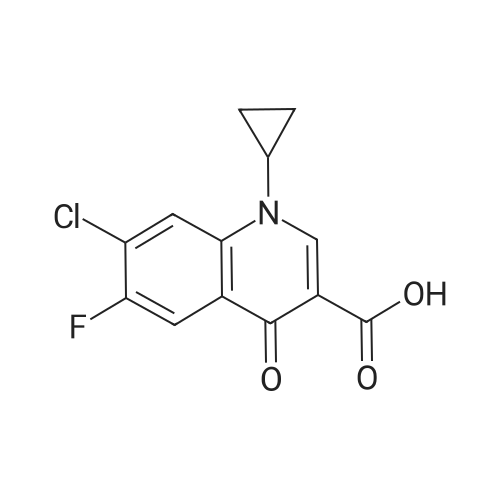
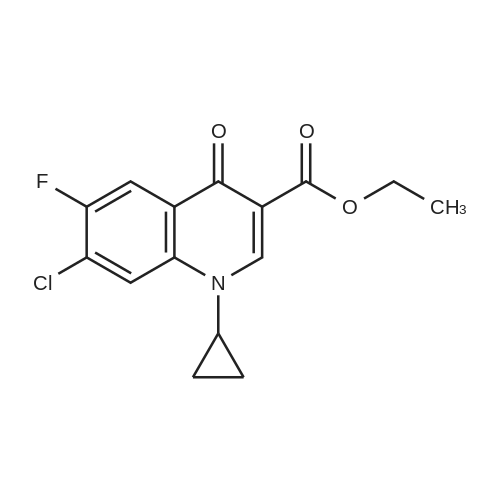
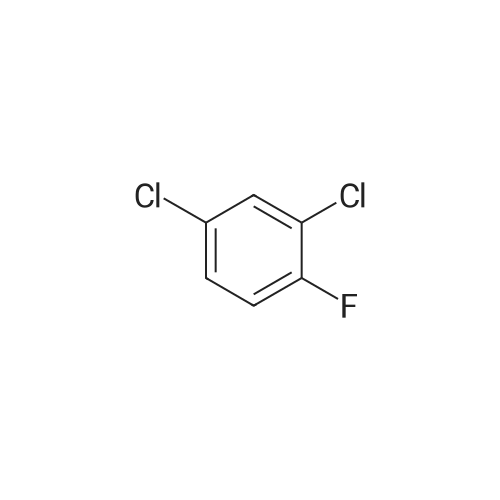
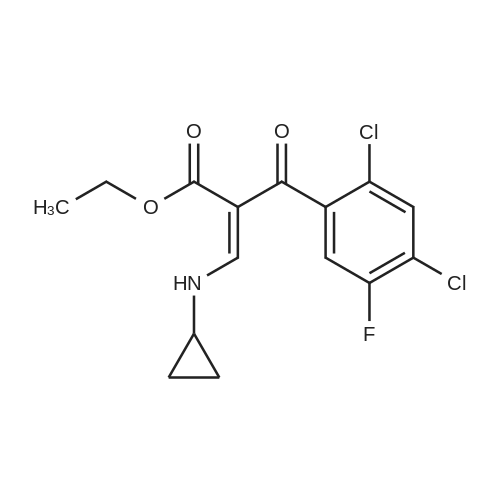
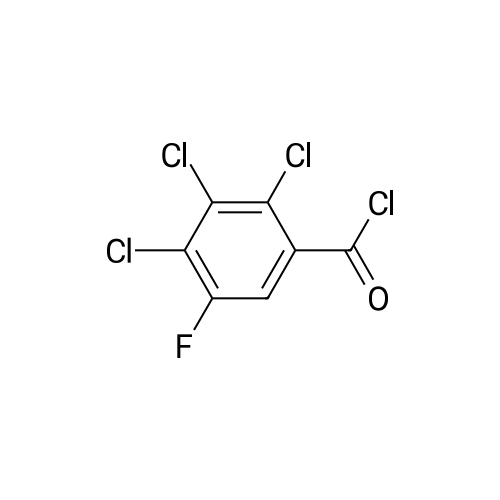
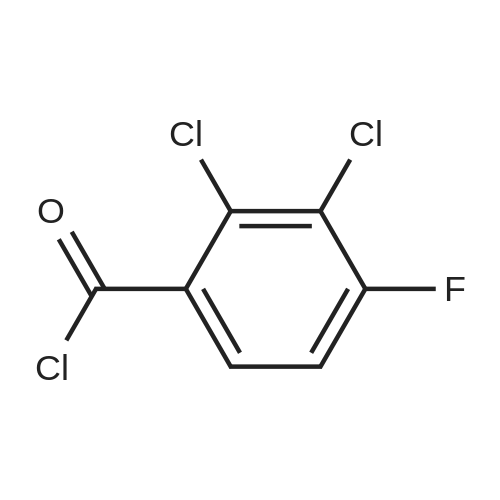
(b) The 7-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-quinoline-3-carboxylic acid used as the starting material was prepared as follows: 24.3 g of magnesium turnings were suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride were added and, when the reaction had started, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether was added dropwise, a vigorous reflux being observed. After the reaction had ceased, the mixture was heated at the boil for a further 2 hours and was cooled with dry ice/acetone at -5° C. to -10° C. and a solution of 227.5 g of 2,4-dichloro-5-fluoro-benzoyl chloride in 100 ml of absolute ether was slowly added dropwise at this temperature. The mixture was stirred for 1 hour at 0° C. to -5° C. and was allowed to reach room temperature overnight, and a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid was allowed to run in while cooling with ice. The phases were separated and were extracted twice with ether. The combined ether solutions were washed with saturated NaCl solution and dried with Na2 SO4, and the solvent was stripped off in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluoro-benzoyl-malonate were obtained as the crude product. |
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
A (Preparation of the starting material II): STR9
EXAMPLE A (Preparation of the starting material II): STR9 24.3 g of magnesium turnings are suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride are added and, when the reaction has started up, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether is added dropwise, vigorous reflux being observed. After the reaction has moderated, the mixture is heated to boiling for 2 hours, then cooled down to -5° C. to -10° C. with dry ice/acetone and, at this temperature, a solution of 227.5 g of 2,4-dichloro-5-fluorobenzoyl chloride (1) in 100 ml of absolute ether is slowly added dropwise. The mixture is stirred at 0° to -5° C. for 1 hour, allowed to reach room temperature overnight and, while cooling in ice, a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid is allowed to run in. The phases are separated and the aqueous phase is extracted twice more with ether. The combined ether solutions are washed with saturated NaCl solution, dried with Na2 SO4 and the solvent is removed in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluorobenzoylmalonate (3) are obtained as a crude product. |
|
With magnesium In Carbon tetrachloride; (2S)-N-methyl-1-phenylpropan-2-amine hydrate; ethanol |
A EXAMPLE A STR18
The 7-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-quinoline-3-carboxylic acid VIa used as a starting material is prepared as follows: 24.3 g of magnesium turnings are suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride are added, and, when the reaction has started, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether is added dropwise, vigorous refluxing taking place. After the reaction has died down, the mixture is heated at the boil for a further 2 hours and cooled to -5° C. to -10° C. with dry ice/acetone, and a solution of 227.5 g of 2,4-dichloro-5-fluoro-benzoyl chloride (1) in 100 ml of absolute ether is slowly added dropwise at this temperature. The mixture is stirred for 1 hour at 0° C. to -5° C., and is allowed to reach room temperature overnight, and a mixture of 400 ml of ice water and 25 ml of concentrated sulphuric acid is allowed to run in, while cooling with ice. The phases are separated, and further extracted twice with ether. The combined ether solutions are washed with saturated NaCl solution and dried with Na2 SO4, and the solvent is stripped off in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluoro-benzoyl-malonate (3) are obtained as a crude product. |
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
A EXAMPLE A (Preparation of the starting material II) STR42
EXAMPLE A (Preparation of the starting material II) STR42 24.3 g of magnesium turnings are suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride are added and, when the reaction has started up, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether is added dropwise, vigorous reflux being observed. After the reaction has moderated, the mixture is heated to boiling for 2 hours, then cooled down to -5° C. to -10° C. with dry ice/acetone and, at this temperature, a solution of 227.5 g of 2,4-dichloro-5-fluorobenzoyl chloride (1) in 100 ml of absolute ether is slowly added dropwise. The mixture is stirred at 0° to -5° C. for 1 hour, allowed to reach room temperature overnight and, while cooling in ice, a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid is allowed to run in. The phases are separated and the aqueous phase is extracted twice more with ether. The combined ether solutions are washed with saturated NaCl solution, dried with Na2 SO4 and the solvent is removed in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluorobenzoylmalonate (3) are obtained as a reaction product. |
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
A EXAMPLE A STR19
The 7-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-3-quinolinecarboxylic acid VIa used as the starting material is prepared as follows: 24.3 g of magnesium filings are suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride are added and, when the reaction has started, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether is added dropwise, whereupon vigorous reflux can be observed. When the reaction has subsided, the mixture is heated at the boiling point for a further 2 hours and cooled to -5° C. to -10° C. with dry ice/acetone, and a solution of 227.5 g of 2,4-dichloro-5-fluorobenzoyl chloride (1) in 100 ml of absolute ether is slowly added dropwise at this temperature. The mixture is stirred at 0° C. to -5° C. for 1 hour and allowed to come to room temperature overnight, and a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid is allowed to run in, while cooling with ice. The phases are separated and subsequently extracted twice with ether. The combined ether solutions are washed with saturated NaCl solution and dried with Na2 SO4 and the solvent is stripped off in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluoro-benzoyl-malonate (3) are obtained as the crude product. |
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
24.b EXAMPLE 24 STR31
(b) The 7-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-quinoline-3-carboxylic acid used as the starting material was prepared as follows: 24.3 g of magnesium turnings were suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride were added and, when the reaction had started, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether was added dropwise, a vigorous reflux being observed. After the reaction had ceased, the mixture was heated at the boil for a further 2 hours and was cooled with dry ice/acetone at -5° C. to -10° C. and a solution of 227.5 g of 2,4-dichloro-5-fluoro-benzoyl chloride in 100 ml of absolute ether was slowly added dropwise at this temperature. The mixture was stirred for 1 hour at 0° C. to -5° C. and was allowed to reach room temperature overnight, and a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid was allowed to run in while cooling with ice. The phases were separated and were extracted twice with ether. The combined ether solutions were washed with saturated NaCl solution and dried with Na2 SO4, and the solvent was stripped off in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluoro-benzoyl-malonate were obtained as the crude product. |
|
With magnesium In Carbon tetrachloride; ice-water; ethanol |
24.b EXAMPLE 24 STR31
(b) The 7-chloro-1-cyclopropyl-6-fluoro-1,4-dihydro-4-oxo-quinoline-3-carboxylic acid used as the starting material was prepared as follows: 24.3 g of magnesium turnings were suspended in 50 ml of anhydrous ethanol. 5 ml of carbon tetrachloride were added and, when the reaction had started, a mixture of 160 g of diethyl malonate, 100 ml of absolute ethanol and 400 ml of anhydrous ether was added dropwise, a vigorous reflex being observed. After the reaction had ceased, the mixture was heated at the boil for a further 2 hours and was cooled with dry ice/acetone at -5° C. to -10° C. and solution of 227.5 g of 2,4-dichloro-5-fluoro-benzoyl chloride in 100 ml of absolute ether was slowly added dropwise at this temperature. The mixture was stirred for 1 hour at 0° C. to -5° C. and was allowed to reach room temperature overnight, and a mixture of 400 ml of ice-water and 25 ml of concentrated sulphuric acid was allowed to run in while cooling with ice. The phases were separated and were extracted twice with ether. The combined ether solutions were washed with saturated NaCl solution and dried with Na2 SO4, and the solvent was stripped off in vacuo. 349.5 g of diethyl 2,4-dichloro-5-fluoro-benzoyl-malonate were obtained as the crude product. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 HazMat Fee +
HazMat Fee +

 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping