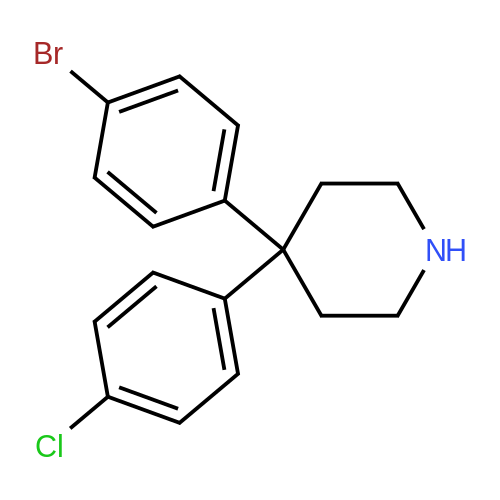
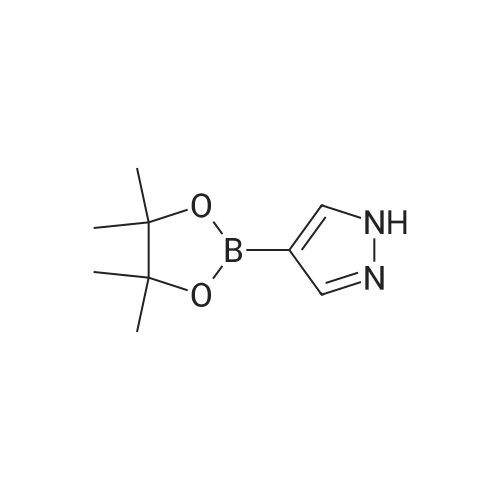
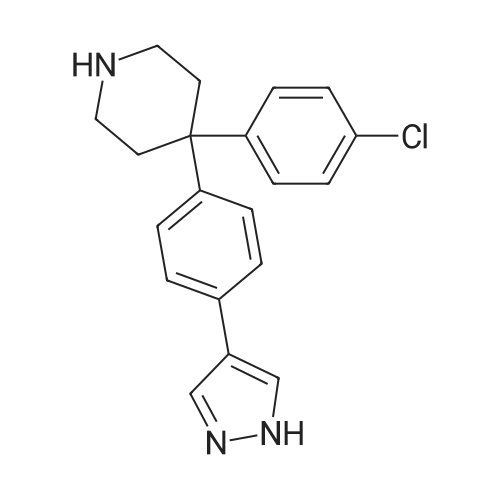
Alternatived Products of [ 857531-00-1 ]
Product Details of [ 857531-00-1 ]
CAS No. : 857531-00-1
MDL No. : MFCD18384971
Formula :
C20 H20 ClN3
Boiling Point : -
Linear Structure Formula : -
InChI Key : LZMOSYUFVYJEPY-UHFFFAOYSA-N
M.W :
337.85
Pubchem ID : 11175137
Synonyms :
Calculated chemistry of [ 857531-00-1 ]
Physicochemical Properties
Num. heavy atoms : 24
Num. arom. heavy atoms : 17
Fraction Csp3 : 0.25
Num. rotatable bonds : 3
Num. H-bond acceptors : 2.0
Num. H-bond donors : 2.0
Molar Refractivity : 102.2
TPSA : 40.71 Ų
Pharmacokinetics
GI absorption : High
BBB permeant : Yes
P-gp substrate : Yes
CYP1A2 inhibitor : Yes
CYP2C19 inhibitor : No
CYP2C9 inhibitor : No
CYP2D6 inhibitor : Yes
CYP3A4 inhibitor : Yes
Log Kp (skin permeation) : -5.34 cm/s
Lipophilicity
Log Po/w (iLOGP) : 2.53
Log Po/w (XLOGP3) : 4.25
Log Po/w (WLOGP) : 4.02
Log Po/w (MLOGP) : 3.69
Log Po/w (SILICOS-IT) : 5.36
Consensus Log Po/w : 3.97
Druglikeness
Lipinski : 0.0
Ghose : None
Veber : 0.0
Egan : 0.0
Muegge : 0.0
Bioavailability Score : 0.55
Water Solubility
Log S (ESOL) : -4.94
Solubility : 0.00389 mg/ml ; 0.0000115 mol/l
Class : Moderately soluble
Log S (Ali) : -4.82
Solubility : 0.00516 mg/ml ; 0.0000153 mol/l
Class : Moderately soluble
Log S (SILICOS-IT) : -8.28
Solubility : 0.00000176 mg/ml ; 0.0000000052 mol/l
Class : Poorly soluble
Medicinal Chemistry
PAINS : 0.0 alert
Brenk : 0.0 alert
Leadlikeness : 1.0
Synthetic accessibility : 2.36
Safety of [ 857531-00-1 ]
Application In Synthesis of [ 857531-00-1 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
Downstream synthetic route of [ 857531-00-1 ]
1
[ 857531-01-2 ]
[ 269410-08-4 ]
[ 857531-00-1 ]
Yield Reaction Conditions Operation in experiment
33%
With potassium phosphate In various solvent(s) at 135℃; for 30h; microwave irradiation;
14.B 14B.
14B. 4-(4-Chloro-phenyl)-4-[4(1H-pyrazol-4-yl)-phenyl]-piperidine 4-(4-Bromo-phenyl)-4-(4-chloro-phenyl)-piperidine was reacted with 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole following the procedure set out in Example 1 to give the title compound. LC/MS: (PS-A3) Rt 7.22 [M+H]+ 338.08. 1H NMR (Me-d3-OD) δ 2.64-2.74 (4H, m), 3.22-3.25 (4H, m), 7.33-7.45 (6H, m), 7.65 (2H, d), 8.37 (2H, s).
Reference:
[1]Saxty, Gordon; Woodhead, Steven J.; Berdini, Valerio; Davies, Thomas G.; Verdonk, Marcel L.; Wyatt, Paul G.; Boyle, Robert G.; Barford, David; Downham, Robert; Garrett, Michelle D.; Carr, Robin A.
[Journal of Medicinal Chemistry, 2007, vol. 50, # 10, p. 2293 - 2296]
[2]Current Patent Assignee: UNIVERSITY OF LONDON; OTSUKA HOLDINGS CO LTD; CANCER RESEARCH UK - US2010/166699, 2010, A1
2
[ CAS Unavailable ]
[ 857531-00-1 ]
Yield Reaction Conditions Operation in experiment
Multi-step reaction with 2 steps
1: 92 percent / AlCl3 / 2 h / 0 °C
2: 33 percent / K3 PO4 / bis(tri-t-butylphosphine)palladium(0) / various solvent(s) / 30 h / 135 °C / microwave irradiation
Reference:
[1]Saxty, Gordon; Woodhead, Steven J.; Berdini, Valerio; Davies, Thomas G.; Verdonk, Marcel L.; Wyatt, Paul G.; Boyle, Robert G.; Barford, David; Downham, Robert; Garrett, Michelle D.; Carr, Robin A.
[Journal of Medicinal Chemistry, 2007, vol. 50, # 10, p. 2293 - 2296]
3
[ 57988-58-6 ]
[ 857531-00-1 ]
Yield Reaction Conditions Operation in experiment
Multi-step reaction with 2 steps
1: 92 percent / AlCl3 / 2 h / 0 °C
2: 33 percent / K3 PO4 / bis(tri-t-butylphosphine)palladium(0) / various solvent(s) / 30 h / 135 °C / microwave irradiation
Reference:
[1]Saxty, Gordon; Woodhead, Steven J.; Berdini, Valerio; Davies, Thomas G.; Verdonk, Marcel L.; Wyatt, Paul G.; Boyle, Robert G.; Barford, David; Downham, Robert; Garrett, Michelle D.; Carr, Robin A.
[Journal of Medicinal Chemistry, 2007, vol. 50, # 10, p. 2293 - 2296]

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping