| 59% |
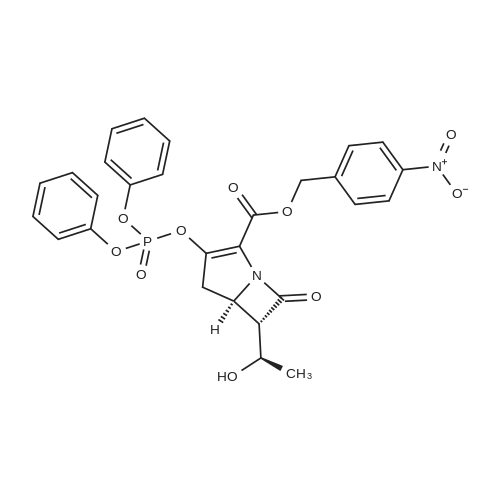
Stage #1: (5R,6S)-6-[(R)-1-hydroxyethyl]-3,7-dioxo-1-azabicyclo[3.2.0]heptane-2-carboxylic acid p-nitrobenzyl ester With trifluoromethylsulfonic anhydride; triethylamine In dichloromethane at -78℃; for 0.5h; Inert atmosphere;
Stage #2: t-butyldimethylsiyl triflate In dichloromethane at -78℃; for 0.5h; Inert atmosphere;
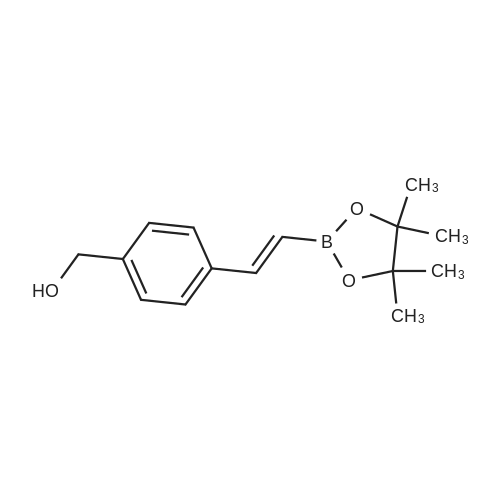
Stage #3: (E)-(4-(2-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)vinyl)phenyl)methanol With tris-(dibenzylideneacetone)dipalladium(0); potassium hydroxide In dichloromethane; water; N,N-dimethyl-formamide at -78 - 30℃; for 1h; Inert atmosphere; |
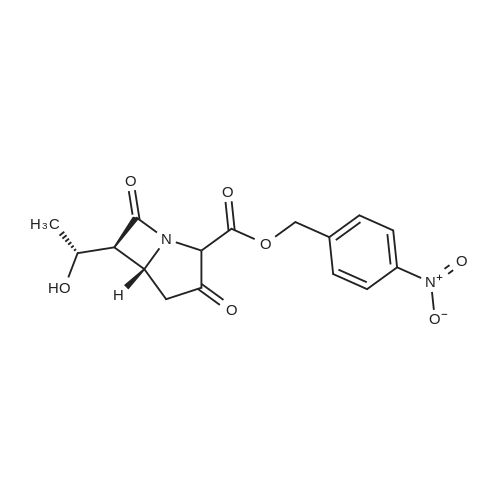
8.A A. Preparation of (5R,6S)-4-nitrobenzyl 6-((R)-1-((tert-butyldimethylsilyl)oxy)ethyl)-3-((E)-4-(hydroxymethyl)styryl)-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate 99
Under nitrogen atmosphere, (5R,6S)-4-nitrobenzyl 6-((R)-1 -hydroxyethyl)- 3,7-dioxo-1 -azabicyclo[3.2.0]heptane-2-carboxylate 92 (from Ark Pharm) (4.10 g, 1 1 .8 mmol, 1 .00 equiv) is dissolved in anhydrous DCM (39 ml_, 0.30 M). At - 78 °C, triethylamine (1 .58 ml_, 1 1 .3 mmol, 0.960 equiv) is added, followed by dropwise addition of trifluoromethanesulfonic anhydride (from Sigma Aldrich) (1 .90 ml_, 1 1 .3 mmol, 0.960 equiv). The resulting reaction mixture is stirred at -78 °C for 0.5 hour and then triethylamine (1 .80 ml_, 13.0 mmol, 1 .10 equiv) is added, followed by dropwise addition of fe/f-butyldimethylsilyl trifluoromethanesulfonate (2.97 ml_, 13.0 mmol, 1 .10 equiv). The resulting mixture is stirred at -78 °C for 0.5 hour. A solution of (E)-(4-(2-(4, 4,5,5- tetramethyl-1 ,3,2-dioxaborolan-2-yl)vinyl)phenyl)methanol 98 (from AK Scientific) (3.06 g, 1 1 .8 mmol, 1 .00 equiv) in anhydrous DMF (24 ml_, 0.50 M) is then added to the mixture, followed by addition of tris(dibenzylideneacetone)dipalladium(0) Pd2(dba)3 (0.22 g, 0.24 mmol, 0.020 equiv) and 6 M aqueous potassium hydroxide (5.9 ml_, 35 mmol, 3.0 equiv). The resulting reaction mixture is warmed to 30 °C and stirred at this temperature for 1 hour. The reaction mixture is cooled to rt and is diluted in AcOEt (100 ml_). This organic layer is washed with water (100 ml_) and the aqueous wash is extracted with AcOEt (100 ml_). The combined organic extracts are dried over magnesium sulfate, filtered and then concentrated under reduced pressure. The residue is purified by column chromatography on silica gel eluting with 0-100% AcOEt/hexanes. The desired product 99 is obtained as pale yellow solid (4.03 g, 6.96 mmol, 59% yield). 1H NMR (500 MHz, CDCI3) d (ppm) 8.25-8.20 (m, 2H), 7.91 (d, 1 H, J = 16.0 Hz), 7.71 -7.66 (m, 2H), 7.48-7.44 (m, 2H), 7.36-7.31 (m, 2H), 6.70 (d, 1 H, J = 17.0 Hz), 5.48 (d, 1 H, J = 13.5 Hz), 5.30 (d, 1 H, J = 14.0 Hz), 4.31 -4.20 (m, 2H), 3.22 (dd, 1 H, J = 17.5 Hz, J = 9.5 Hz), 3.18 (dd, 1 H, J = 5.5 Hz, J = 3.0 Hz), 3.10 (dd, 1 H, J = 17.5 Hz, J = 9.0 Hz), 1 .29 (d, 3H, J = 6.0 Hz), 0.89 (s, 9H), 0.10 (s, 6H). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping