|
|
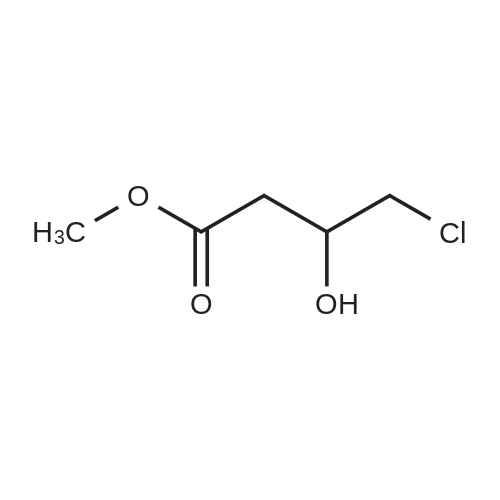
EXAMPLE 11 12 g 2-(1-methylethyl)-4-imidazolidinone hydrochloride ((5); R4 =H, R5 =isopropyl) (0.073 moles) are shaken with 20 ml water and treated with 5 g potassium carbonate (0.036 moles). 8.5 g methyl 3,4-epoxybutanoate (0.073 moles) and 12 ml acetone are added. The mixture is stirred for 45 h at 70 C. After evaporating, the residue is chromatographed on silica, eluding with 8:2 ethyl acetate/methanol. The main fractions are combined and evaporated. 2.5 g of methyl 2-(1-methylethyl)-4-oxoimidazolidine-beta-hydroxybutanoate, m.p. 109-122 C., are obtained as a white powder (14.4%). 2.0 g of this compound (0.0082 mole) are heated to boiling for 15 h with 8.8 ml DMF and 2.2 ml water. After evaporation in vacuo, the residue is taken up with methanol, and filtered. 0.74 g oxiracetam is obtained as a white crystalline powder, m.p. 167-70 |
|
|
EXAMPLE 13 A solution of 0.65 g 2-methylpropyl 2-phenyl-4-oxoimidazolidine-beta-hydroxybutanoate in 1.5 ml dimethyl sulfoxide and 0.5 ml water is heated at reflux for 16 h. After evaporating, the residue is chromatographed on silica, eluding with 7:3 ethyl acetate/methanol. Oxiracetam is obtained as a white powder, m.p. 167-70 C. |
|
|
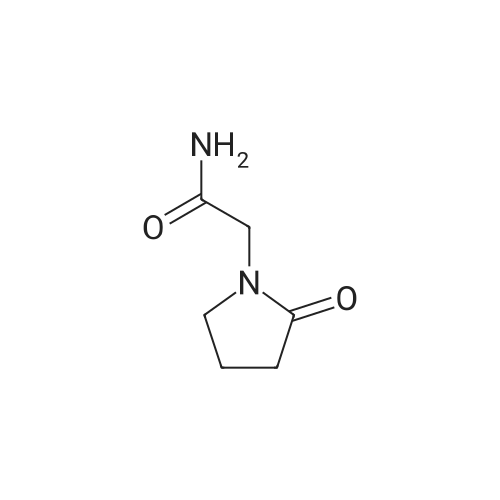
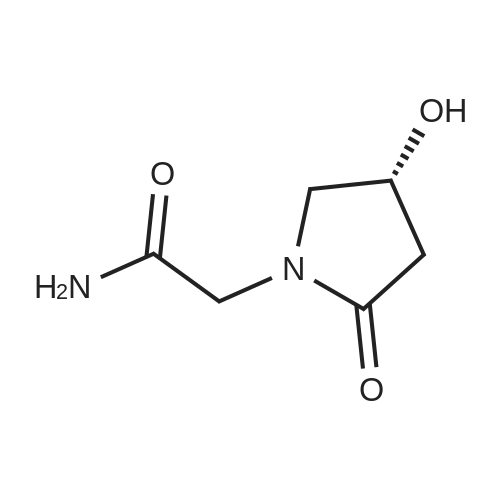
B. 4-Hydroxy-2-oxo-1-pyrrolidineacetamide (oxiracetam). A solution of compound A (1 g) in 25 ml ethanol is hydrogenated at ambient temperature and pressure in the presence of 100 mg 5% Pd/C. The catalyst is filtered off and the solvent evaporated to give 0.72 g 2-methylpropyl 4-(carbamoylmethylamino)-3-hydroxybutanoate. This compound is dissolved in situ in 10 ml acetonitrile and heated at reflux for 8 h. After evaporation of the solvent, the residue is crystallized from ethanol. Oxiracetam is obtained as a white crystalline powder of m.p. 167-70 C. |
|
|
EXAMPLE 2 1 g of 2,2-dimethyl-4-imidazolidinone ((5); R4 =R5 =CH3) (8.76 mmoles) is dissolved in 5 ml acetonitrile. 1 g 2-methylpropyl 3,4-epoxybutanoate ((4); X=isobutyl) (6.32 mmoles) is added and the mixture is boiled for 30 h with stirring. The mixture is cooled, the solvent is decanted off, the resulting dark solid mass is taken up in ethanol, and the solid oxiracetam obtained is filtered off, m.p. 167-70 C. |
|
|
EXAMPLE 3 0.650 g 2,2-dimethyl-4-imidazolidinone ((5); R4 =R5 =CH3) (5.7 mmoles) is dissolved in 5 ml isopropanol. 1 g 2-methylpropyl 3,4-epoxybutanoate ((4); X=isobutyl) (6.32 mmoles) is added. The mixture is boiled for 9 h. A further 1 g of epoxyester is added and boiling is continued for a further 9 h. After cooling, the solvent is decanted and the dark mass obtained is taken up in ethanol. The solid obtained is filtered, to give oxiracetam, m.p. 167-70 C. |
|
|
EXAMPLE 4 1 g of 2,2-dimethyl-4-imidazolidinone ((5); R4 =R5 =CH3) (8.76 mmoles) are heated at 165 C. (external temperature) for 1 h 30 min with 2 g 2-methylpropyl 3,4-epoxybutanoate ((4); X=isobutyl) (12.64 mmoles). 0.17 ml of water and 5 ml ethanol are added, and boiling is continued for a further 2 h. The mixture is cooled and filtered. Oxiracetam is obtained as a white crystalline powder, m.p. 167-70 C. |
|
|
EXAMPLE 6 0.5 g 2,2-dimethyl-4-imidazolidinone ((5); R4 =R5 =CH3) (4.38 mmoles) are heated at 115 C. (external temperature) for 12 h with stirring by a magnetic stirrer, with 0.57 g ethyl 3,4-epoxybutanoate ((4); X=C2 H5) (4.38 mmoles). The mixture is cooled, taken up with methanol and filtered. Oxiracetam is obtained, m.p. 167-70 C. |
|
|
This compound is dissolved in 3 ml acetonitrile and 1 ml water, and boiled for 72 h. After evaporating, the product is taken up with ethanol and filtered, oxiracetam is obtained as a white crystalline powder, m.p. 167-70 C. |
|
|
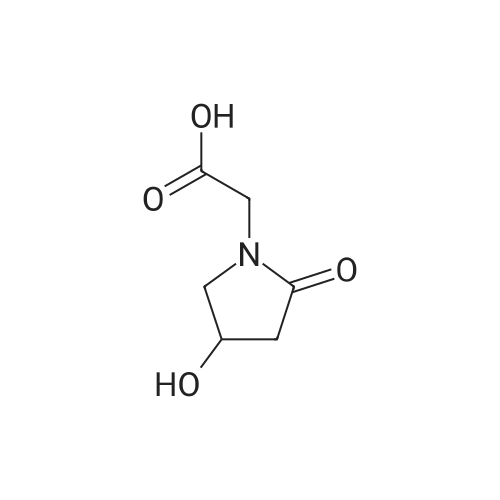
8 6.52 g 2-(1-methylethyl)-4-imidazolidinone hydrochloride ((5); R4 =H, R5 =isopropyl) (0.04 moles) are dissolved in 40 ml water and treated with 2.8 g potassium carbonate (0.02 mole). 7 g 2-methylpropyl 3,4-epoxybutanoate are added ((4); X=isobutyl) (0.044 mole) and 25 ml acetone. The mixture is heated at 60 C. for 48 h with stirring. The mixture is reduced to a small volume, the solid is filtered and washed with ether. 3.5 g of a white solid, m.p. 135-40 C. is obtained. The liquor from which this solid was separated is evaporated to dryness and chromatographed on silica, eluding with 8:2 ethylacetate/methanol. The main fractions are collected and evaporated to give a further 2.9 g compound of m.p. 135-40 C., giving a total of 6.4 g 2-methylpropyl 2-(1-methylethyl)-4-oxo-1-imidazolidine-beta-hydroxybutanoate ((2); R3 =R4 =H, R5 =isopropyl, X=isobutyl). 1 g of this compound (3.5 mmoles) is heated to boiling for 8 h in 6 ml dimethyl sulfoxide+2 ml water. After evaporation, the residue is taken up with acetone, filtered, dried in vacuo and crystallized from methanol. 0.32 g Oxiracetam is obtained as a white crystalline powder, m.p. 167-70 C. (yield 57.8%). |
|
|
EXAMPLE 9 5.8 g of 2-(1-methylethyl)-4-imidazolidinone hydrochloride ((5); R4 =H, R5 =isopropyl) (0.035 moles) are shaken in 10 ml water and treated with 2.4 g potassium carbonate (0.0174 moles). 4.5 g ethyl 3,4-epoxybutanoate and 6 ml acetone are added, and stirring is maintained for 45 h at 70 C. After evaporation, the residue is chromatographed on silica, eluding with 8:2 ethyl acetate/methanol. The main fractions are collected and evaporated. The oil that remains is taken up with ethyl acetate. After being left to stand overnight the white compound obtained is filtered: 1.3 g ethyl 2-(1-methylethyl)-4-oxo-1-imidazolidine-beta-hydroxybutanoate m.p. 118-122 C. 0.95 g of this compound (3.7 mmoles) are heated to boiling for 15 h in 4 ml DMF and 1 ml water. After evaporation in vacuo, the residue is taken up with methanol, and the solution is filtered. 0.37 g oxiracetam is obtained as a white crystalline powder, m.p. 167-70 C. (63.2%). |
|
|
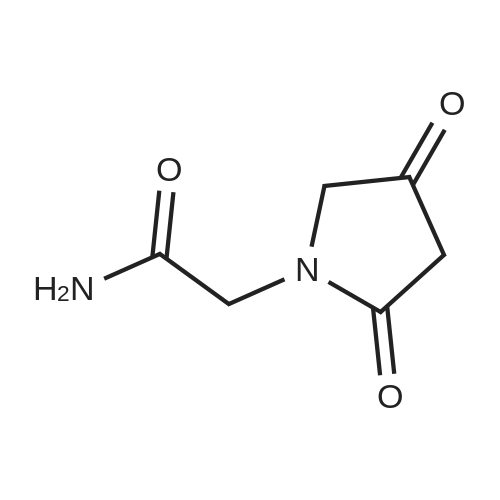
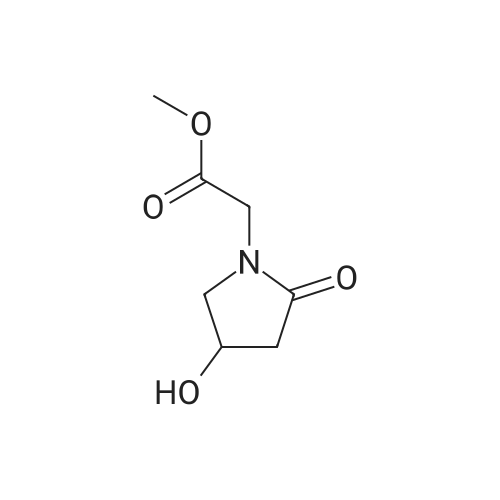
EXAMPLE 1 2-(4-Hydroxypyrrolidin-2-on-1-yl) acetamide To a mixture containing 648 g ethyl iminodiacetate in 3600 ml anhydrous methylene chloride and 572 ml triethylamine at 0 C., a solution of 619 g 2-carbethoxyacetylchloride in 1100 ml methylene chloride is added dropwise and under stirring, while checking that the reaction temperature does not exceed 10-15 C. The mixture is kept under stirring for 2 hours at room temperature and allowed to stand overnight, thereafter washed with water, made anhydrous and evaporated under vacuo. Ethyl N-(2-carbethoxyacetyl)-imino-diacetate, in the form of an oil, is dissolved in anhydrous benzene and added at room temperature to a solution of 75.6 g sodium in 2700 ml of absolute ethyl alcohol. The solution is refluxed for 6 hours, cooled to room temperature, repeatedly extracted with water, the aqueous extracts collected together and acidified to pH 1 with hydrogen chloride, and a precipitate containing 2-(3-carbethoxy-4-hydroxy-Delta3 -pyrrolidin-2-on-1-yl) ethyl acetate, which purified by recrystallization melts at 175-179 C., is obtained. Twenty grams of 2-(3-carbethoxy-4-hydroxy-Delta3 -pyrrolidin-2-on-1-yl) ethyl acetate are added to 200 ml warm anhydrous acetonitrile and 1.8 ml water. The mixture is refluxed for about 20 minutes and thereafter cooled on an ice bath and evaporated under vacuo to give 2-(pyrrolidino-2,4-dion-1-yl) ethyl acetate melting at 87-91 C. To 22.25 g 2-(pyrrolidino-2,4-dion-1-yl) ethyl acetate in 445 ml anhydrous dimethoxyethane cooled to 0 C., 1.52 g sodium borohydride are added; the mixture is allowed to stand for 10 minutes on an ice bath and then for 30 minutes at room temperature. The solution is acidified with 20% hydrochloric acid, filtered in vacuo, evaporated in vacuo, taken up with methylene chloride and made anhydrous over magnesium sulphate; by filtration and evaporation, in vacuo and successive chromatography 2-(4-hydroxy-pyrrolidin-2-on-1-yl) ethyl acetate having a boiling point of 180 C. (with decomposition) is separated. To a solution of 8.9 g 2-(4-hydroxypyrrolidin-2-on-1-yl) ethyl acetate in 300 ml methyl alcohol is added at 0 C. gaseous ammonia and is then allowed to stand overnight. The solvent is removed in vacuo, the residue is taken up with methyl alcohol, filtered over charcoal, and slowly added to 200 ml isopropyl ether. 2-(4-Hydroxypyrrolidin-2-on-1-yl) acetamide melting at 161-163 C. precipitates. |
|
|
EXAMPLE 2 R(+) 2-(4-Hydroxypyrrolidin-2-on-1-yl)-acetamide Operating is as described in Example 1 and using as the starting material R(-) gamma-amino-beta-hydroxybutyric acid, there is obtained, via ethyl R(+) 2-(4-hydroxypyrrolidin-2-on-1-yl)-acetate (b.p. 179 C./0.8 mm.Hg.), R(+) 2-(4-hydroxypyrrolidin-2-on-1-yl)-acetamide; m.p. 135-136 C.; [alpha]D =+36.2 (water, c+1). |
|
In water; ethyl acetate; |
(c) The above reaction solution was filtered, and the residue was fully washed with ethanol and the filtrate was concentrated. The concentrate was dissolved in water, and extracted by adding ethyl acetate 7 times to the weight of the filtrate. The aqueous phase was concentrated, and separated with column chromatography. Finally, aqueous ammonia of 23 wt % was added and then the reaction continued for 4h at 22 C to obtain crude oxiracetam, wherein the amount of 23wt% aqueous ammonia is 13 times to the weight of the product separated by column chromatography; |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping