Alternatived Products of [ 64385-02-0 ]
Product Details of [ 64385-02-0 ]
| CAS No. : | 64385-02-0 |
MDL No. : | MFCD30533929 |
| Formula : |
C7HI5O2
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | LNLWJOZRHQPFML-UHFFFAOYSA-N |
| M.W : |
751.60
|
Pubchem ID : | 12256098 |
| Synonyms : |
|
Application In Synthesis of [ 64385-02-0 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 64385-02-0 ]
- 1
-
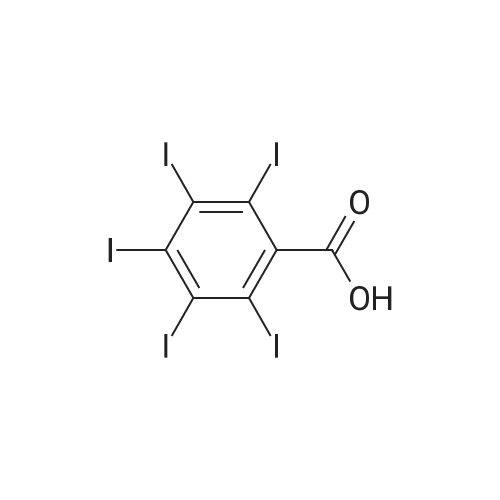 [ 64385-02-0 ]
[ 64385-02-0 ]

-
 [ 536-74-3 ]
[ 536-74-3 ]

-
 [ 123568-37-6 ]
[ 123568-37-6 ]
| Yield | Reaction Conditions | Operation in experiment |
| 32% |
With copper(l) iodide; triethylamine at 50℃; for 12h; |
|
- 2
-
 [ 65-85-0 ]
[ 65-85-0 ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 49% |
With iodine; periodic acid In sulfuric acid 1.) room temperature, 1 d, 2.) 95 deg C, 1 d; |
|
- 3
-
 [ 64349-92-4 ]
[ 64349-92-4 ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With water |
|
| Yield | Reaction Conditions | Operation in experiment |
|
entspr. Saeure 4, KI; |
|
- 5
-
 [ 109-06-8 ]
[ 109-06-8 ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]

-
C7HI5O2*C6H7N
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 79% |
In diethyl ether; N,N-dimethyl-formamide at 20℃; for 48h; |
Preparation of 1
PIBA (80 mg, 0.09 mmol) was dissolved in 3 mL of DMF, after which 2-methylpyridine (10 μL, 0.11 mmol) was added. Slow diffusion of diethyl ether vapor at room temperature for 2 days results in the formation of pale yellowcrystals of 1 with a yield of ≈79%. |
- 6
-
 [ 108-89-4 ]
[ 108-89-4 ]

-
copper(II) nitrate trihydrate
[ No CAS ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]

-
 [ 33513-42-7 ]
[ 33513-42-7 ]

-
Cu(2+)*2C7I5O2(1-)*C6H7N*3.5C3H7NO*0.5H2O
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 81% |
In diethyl ether Sealed tube; |
2.1 Preparation of 1-4
General procedure: In all cases, PIBA was dissolved in DMF, followed by addition of corresponding pyridine and, after that, solution of Cu(NO3)2·3H2O in DMF (see Table 1 for details). After that, the mixture was placed into bigger vial containing diethyl ether and closed. Slow diffusion of Et2O results in formation of crystalline samples of 1-4 (all brownish-green) suitable for XRD (within 1-5days). Yields: 79% (1), 83% (2), 87% (3), 81% (4). |
- 7
-
 [ 110-86-1 ]
[ 110-86-1 ]

-
copper(II) nitrate trihydrate
[ No CAS ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]

-
C24H10CuI10N2O4
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 79% |
In diethyl ether; N,N-dimethyl-formamide Sealed tube; |
2.1 Preparation of 1-4
General procedure: In all cases, PIBA was dissolved in DMF, followed by addition of corresponding pyridine and, after that, solution of Cu(NO3)2·3H2O in DMF (see Table 1 for details). After that, the mixture was placed into bigger vial containing diethyl ether and closed. Slow diffusion of Et2O results in formation of crystalline samples of 1-4 (all brownish-green) suitable for XRD (within 1-5days). Yields: 79% (1), 83% (2), 87% (3), 81% (4). |
- 8
-
 [ 591-22-0 ]
[ 591-22-0 ]

-
copper(II) nitrate trihydrate
[ No CAS ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]

-
C28H18CuI10N2O4
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 83% |
In diethyl ether; N,N-dimethyl-formamide Sealed tube; |
2.1 Preparation of 1-4
General procedure: In all cases, PIBA was dissolved in DMF, followed by addition of corresponding pyridine and, after that, solution of Cu(NO3)2·3H2O in DMF (see Table 1 for details). After that, the mixture was placed into bigger vial containing diethyl ether and closed. Slow diffusion of Et2O results in formation of crystalline samples of 1-4 (all brownish-green) suitable for XRD (within 1-5days). Yields: 79% (1), 83% (2), 87% (3), 81% (4). |
- 9
-
 [ 108-48-5 ]
[ 108-48-5 ]

-
copper(II) nitrate trihydrate
[ No CAS ]

-
 [ 64385-02-0 ]
[ 64385-02-0 ]

-
C28H18CuI10N2O4
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 87% |
In diethyl ether; N,N-dimethyl-formamide Sealed tube; |
2.1 Preparation of 1-4
General procedure: In all cases, PIBA was dissolved in DMF, followed by addition of corresponding pyridine and, after that, solution of Cu(NO3)2·3H2O in DMF (see Table 1 for details). After that, the mixture was placed into bigger vial containing diethyl ether and closed. Slow diffusion of Et2O results in formation of crystalline samples of 1-4 (all brownish-green) suitable for XRD (within 1-5days). Yields: 79% (1), 83% (2), 87% (3), 81% (4). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping






