Alternatived Products of [ 582-77-4 ]
Product Details of [ 582-77-4 ]
| CAS No. : | 582-77-4 |
MDL No. : | MFCD00041331 |
| Formula : |
C14H13NO
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
211.26
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 582-77-4 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 582-77-4 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 582-77-4 ]
- Downstream synthetic route of [ 582-77-4 ]
- 1
-
 [ 582-77-4 ]
[ 582-77-4 ]

-
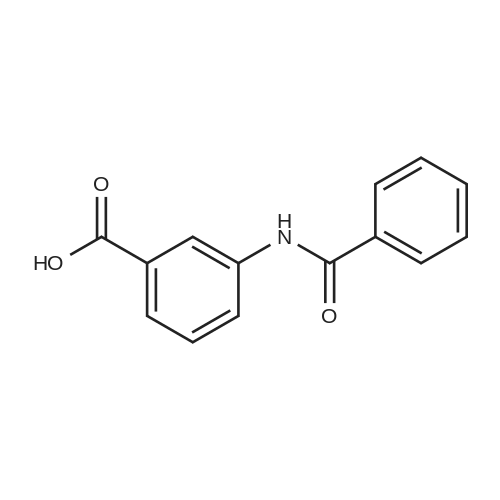 [ 587-54-2 ]
[ 587-54-2 ]
| Yield | Reaction Conditions | Operation in experiment |
| 30% |
With potassium permanganate In water for 1.5h; Heating; |
|
- 2
-
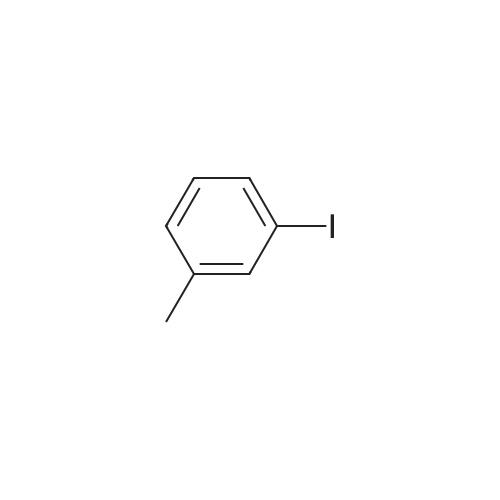 [ 625-95-6 ]
[ 625-95-6 ]

-
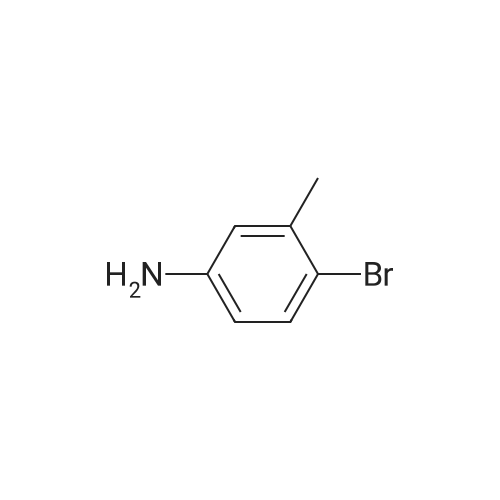
 [ 55-21-0 ]
[ 55-21-0 ]

-
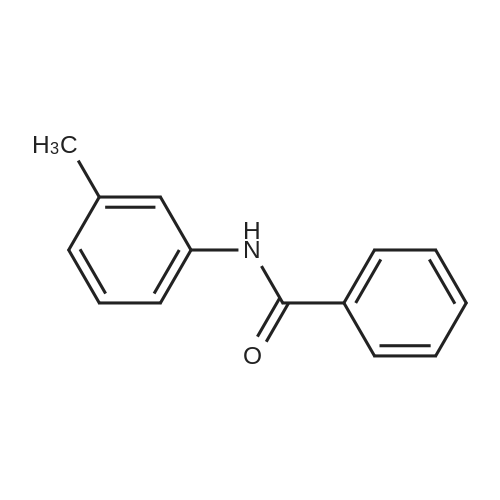
 [ 582-77-4 ]
[ 582-77-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 89% |
With 2-Phenyl-1,3,2-dioxaborinane In 1,4-dioxane at 115℃; for 30h; Inert atmosphere; |
|
| 79% |
With potassium phosphate; N,N`-dimethylethylenediamine In toluene at 135℃; for 24h; |
|
| 75% |
With cobalt(II) oxalate dihydrate; caesium carbonate; N,N`-dimethylethylenediamine In water at 120℃; for 24h; |
|
| 55% |
With iron(III) chloride; potassium phosphate monohydrate; air; N,N`-dimethylethylenediamine In water at 125℃; for 36h; |
|
| 45% |
With copper(l) iodide; manganese(II) fluoride; (±)-trans-1,2-diaminocyclohexane; potassium hydroxide In water at 60℃; for 24h; |
|
| 24% |
With potassium phosphate monohydrate; cobalt(II) chloride hexahydrate; N,N`-dimethylethylenediamine In water at 120℃; for 36h; |
|

Reference:
[1]Sen, Abhijit; Dhital, Raghu N.; Sato, Takuma; Ohno, Aya; Yamada, Yoichi M.A.
[ACS Catalysis, 2020, vol. 10, # 24, p. 14410 - 14418]
[2]Correa, Arkaitz; Bolm, Carsten
[Angewandte Chemie - International Edition, 2007, vol. 46, # 46, p. 8862 - 8865]
[3]Tan, Bryan Yong-Hao; Teo, Yong-Chua
[Organic and biomolecular chemistry, 2014, vol. 12, # 38, p. 7478 - 7481]
[4]Teo, Yong-Chua
[Advanced Synthesis and Catalysis, 2009, vol. 351, # 5, p. 720 - 724]
[5]Teo, Yong-Chua; Yong, Fui-Fong; Ithnin, Idzham Khalid; Yio, Siew-Hui Trionna; Lin, Zhiyin
[European Journal of Organic Chemistry, 2013, # 3, p. 515 - 524]
[6]Teo, Yong-Chua; Chua, Guan-Leong
[Chemistry - A European Journal, 2009, vol. 15, # 13, p. 3072 - 3075]
- 3
-
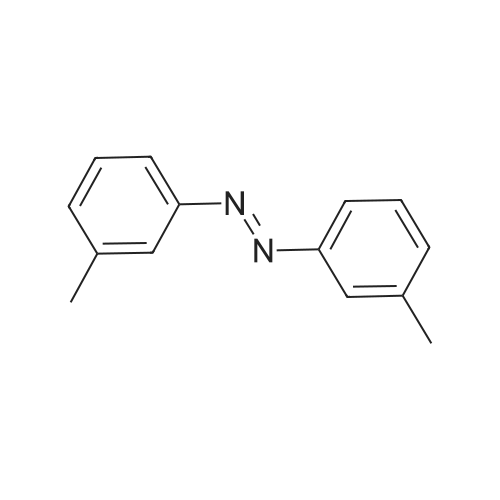 [ 588-04-5 ]
[ 588-04-5 ]

-
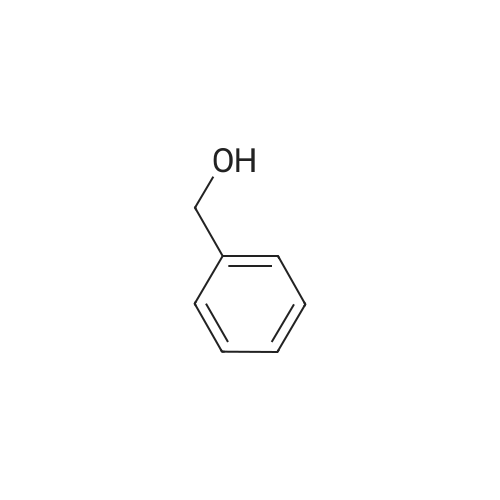 [ 100-51-6 ]
[ 100-51-6 ]

-
 [ 582-77-4 ]
[ 582-77-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 34% |
With tert.-butylhydroperoxide; water In acetonitrile at 120℃; for 24h; Sealed tube; |
4.2. Typical procedure for TBHP-mediated reaction of alcoholswith azobenzenes
General procedure: The mixture of azobenzenes 1 (0.25 mmol), alcohols 2(0.5 mmol), TBHP (1 mmol) and CH3CN (1 mL) were added into a sealed tube under air. After being stirred vigorously at 120 °C for 24 h, the mixture was evaporated under vacuum. The corresponding product was isolated by silica gel column chromatography with a petroleum ether/ethyl acetate mixture as eluent. |
- 4
-
 [ 582-77-4 ]
[ 582-77-4 ]

-
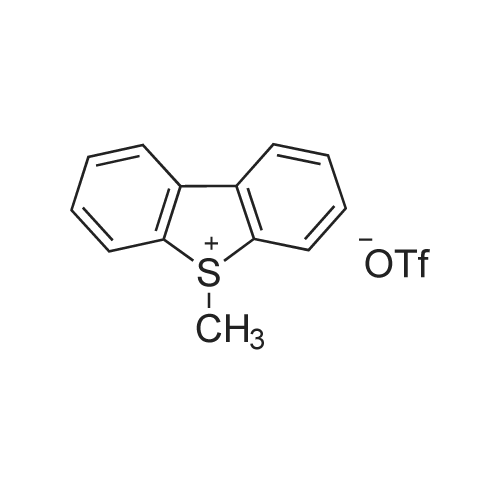 [ 112359-25-8 ]
[ 112359-25-8 ]

-
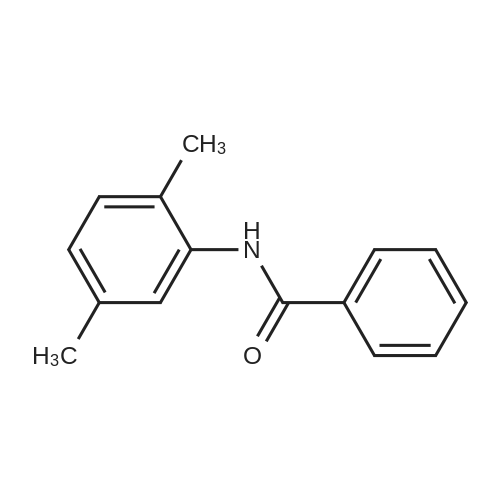 [ 71114-52-8 ]
[ 71114-52-8 ]
| Yield | Reaction Conditions | Operation in experiment |
| 84% |
With palladium diacetate; copper(II) acetate monohydrate; trifluoroacetic acid In 1,2-dichloro-ethane at 50℃; for 5h; Sealed tube; |
|
- 5
-
 [ 582-77-4 ]
[ 582-77-4 ]

-
 [ 65854-91-3 ]
[ 65854-91-3 ]

-
N-(5′-chloro-4-methyl-2′-pivalamido[1,1′-biphenyl]-2-yl)benzamide
[ No CAS ]
- 6
-
 [ 201230-82-2 ]
[ 201230-82-2 ]

-
 [ 2996-92-1 ]
[ 2996-92-1 ]

-
 [ 108-44-1 ]
[ 108-44-1 ]

-
 [ 582-77-4 ]
[ 582-77-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 83% |
With copper (II)-fluoride; bis(triphenylphosphine)palladium(II) dichloride In acetonitrile at 80℃; for 24h; Schlenk technique; |
|
| 83% |
With copper (II)-fluoride; bis-triphenylphosphine-palladium(II) chloride In acetonitrile at 80℃; for 24h; Sealed tube; |
6 Preparation of N-(m-tolyl)benzamide
0.75 mmol of trimethoxyphenylsilane, 10 mol% of Pd(PPh3)Cl2 and 2equiv.CuF2 were dissolved in 3 mL of CH3CN Shrek tube (with magnetic stirrer), and the reaction tube was closed and passed through the branch of the reaction tube. Add carbon monoxide, fill it with balloons and then empty it, repeat it three times until all the air is emptied, then fill the balloon, then add 0.5 mmol of m-methylaniline to the reaction tube and heat and stir at 80 °C. The reaction was carried out by TLC. After the reaction was completed, the mixture was extracted with ethyl acetate (10 mL), and the organic phase was combined and concentrated to silica gel column chromatography to give 87.6 mg of white solid compound (yield: 83%). : |
Reference:
[1]Zhang, Jin; Hou, Yanyan; Ma, Yangmin; Szostak, Michal
[Journal of Organic Chemistry, 2019, vol. 84, # 1, p. 338 - 345]
[2]Current Patent Assignee: SHAANXI UNIVERSITY OF SCIENCE AND TECHNOLOGY - CN109180518, 2019, A
Location in patent: Paragraph 0097; 0098; 0099; 0100; 0102

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping