| 53% |
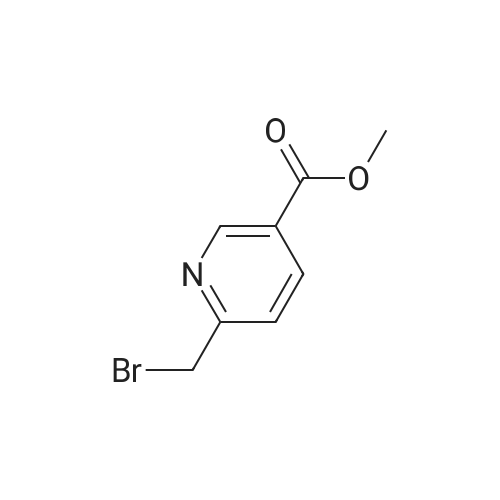
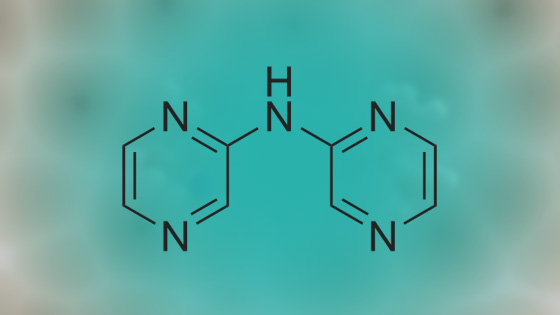
Stage #1: di(pyrazin-2-yl)amine With sodium hydride In N,N-dimethyl-formamide at 5℃; for 0.333333h; Inert atmosphere;
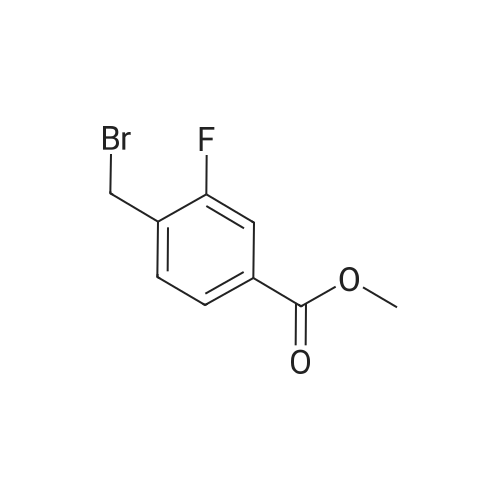
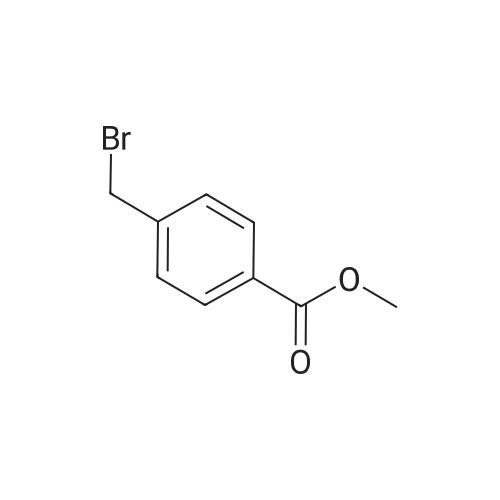
Stage #2: Methyl 4-(bromomethyl)benzoate In N,N-dimethyl-formamide at 70℃; for 1h; Inert atmosphere; |
GG
NaH (60%, 48.5mg, 1 .21 mmol) was added to a solution of (3) (200mg, 1 .15mmol) in DMF (7ml_) at 5°C under N2(g). The reaction mixture was stirred for 20min then methyl 4-(bromomethyl)benzoate (344mg, 1 .5mmol) was added as a solution in DMF (3ml_). The stirring was continued at 70°C for 1 h. Reaction cooled to rt and poured onto water (100ml_). Brine (25mL) was added and extracted with EtOAc (2 x 100ml_). Combined organic was dried over Na2S04, filtered and concentrated in vacuo. The residue was purified by flash column chromatography with CH2CI2/EtOAc (1 :0-0: 1 ) then EtOAc/MeOH (1 :0-4: 1 ) to give (4) (196 mg, 53%). 1 H NMR (500 MHz, Chloroform-d), δΗ ppm: 8.59-8.65 (m, 2H), 8.23-8.26 (m, 2H), 8.16 (d, J=2.5 Hz, 2H), 7.94 (d, J=8.3 Hz, 2H), 7.38 (d, J=8.2 Hz, 2H), 5.50 (s, 2H), 3.86 (s, 3H). LCMS (ES): Found 321 .9 [M+H]+. |
| 53% |
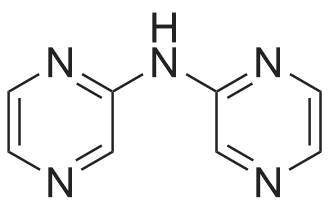
Stage #1: di(pyrazin-2-yl)amine With sodium hydride In N,N-dimethyl-formamide; mineral oil at 5℃; for 0.333333h; Inert atmosphere;
Stage #2: Methyl 4-(bromomethyl)benzoate In N,N-dimethyl-formamide; mineral oil at 70℃; for 1h; |
GG
NaH (60%, 48.5mg, 1.21 mmol) was added to a solution of (3) (200mg, 1.15mmol) in DMF (7mL) at 5°C under N2(g). The reaction mixture was stirred for 20min then methyl 4-(bromomethyl)benzoate (344mg, 1.5mmol) was added as a solution in DMF (3ml_). The stirring was continued at 70°C for 1 h. Reaction cooled to rt and poured onto water (100ml_). Brine (25ml_) was added and extracted with EtOAc (2 x 100ml_). Combined organic was dried over Na2S04, filtered and concentrated in vacuo. The residue was purified by flash column chromatography with CH2CI2/EtOAc (1 :0-0:1) then EtOAc/MeOH (1 :0-4: 1) to give (4) (196 mg, 53%). 1 H NMR (500 MHz, Chloroform-d), δΗ ppm: 8.59-8.65 (m, 2H), 8.23-8.26 (m, 2H), 8.16 (d, J=2.5 Hz, 2H), 7.94 (d, J=8.3 Hz, 2H), 7.38 (d, J=8.2 Hz, 2H), 5.50 (s, 2H), 3.86 (s, 3H). (0478) LCMS (ES): Found 321.9 [M+H]+. |
| 53% |
Stage #1: di(pyrazin-2-yl)amine With sodium hydride In N,N-dimethyl-formamide at 5℃; Inert atmosphere;
Stage #2: Methyl 4-(bromomethyl)benzoate In N,N-dimethyl-formamide at 70℃; for 1h; |
4-[Bis(pyrazin-2-yl)amino]methyl}-N-hydroxybenzamide
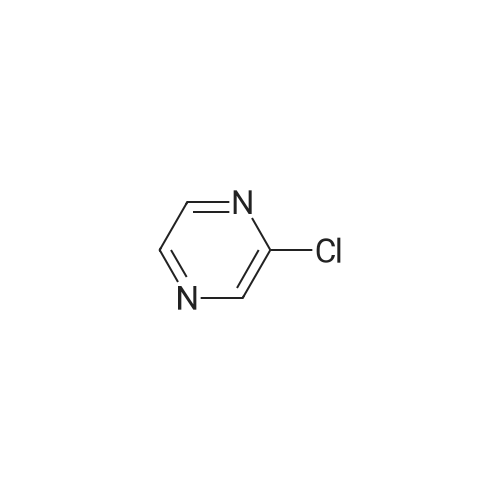
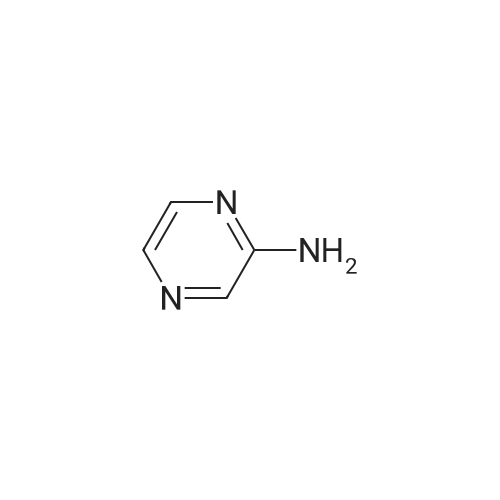
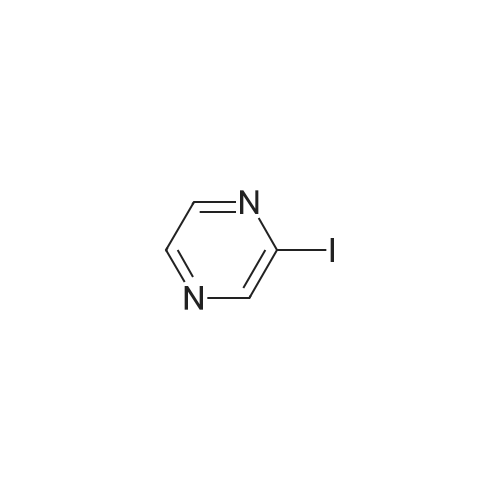
A solution of 2-iodopyrazine (1) (1.2g, 5.83mmol), pyrazin-2-amine (2) (609mg, 6.4mmol), CS2CO3 (3.80g, 11.7mmol) and Xantphos (148mg, 0.26mmol) in dioxane (25ml_) was purged with Il2(g) for lOmin. Pd2(dba)3 (107mg, 0.12mmol) was added and mixture was heated to 90°C for 3h. Reaction cooled to rt and poured onto water (200ml_), extracted with EtOAc (2 x 150ml_) and CH2CI2-IPA (150mL, 4: 1). Combined organics were dried over Na2SC>4, filtered and concentrated in vacuo. Flash column chromatography with heptane/ EtOAc (4: 1- 0: 1) then EtOAc/MeOH (1 :0-3: 1) yielded (3) as an off white solid (210 mg, 51%). XH NMR (500 MHz, Chloroform-d), 6H ppm: 8.99 (d, 7= 1.4 Hz, 2H), 8.30 (dd, 7=2.6, 1.5 Hz, 2H), 8.11 (d, 7=2.7 Hz, 2H). (0135) LCMS (ES) : Found 174.1 [M + H]+. (0136) NaH (60%, 48.5mg, 1.21mmol) was added to a solution of (3) (200mg, 1.15mmol) in DMF (7ml_) at 5°C under N2(g). The reaction mixture was stirred for 20min then methyl 4-(bromomethyl)benzoate (344mg, 1.5mmol) was added as a solution in DMF (3ml_). The stirring was continued at 70°C for lh. Reaction cooled to rt and poured onto water (lOOmL). Brine (25ml_) was added and extracted with EtOAc (2 x lOOmL). Combined organic was dried over Na2S04, filtered and concentrated in vacuo. The residue was purified by flash column chromatography with ChhCh/EtOAc (1 :0-0: 1) then EtOAc/MeOH (1 :0-4: 1) to give (4) (196 mg, 53%). (0137) XH NMR (500 MHz, Chloroform-d), 6H ppm: 8.59-8.65 (m, 2H), 8.23-8.26 (m, 2H), 8.16 (d, .7=2.5 Hz, 2H), 7.94 (d, 7=8.3 Hz, 2H), 7.38 (d, 7=8.2 Hz, 2H), 5.50 (s, 2H), 3.86 (s, 3H). (0138) LCMS (ES) : Found 321.9 [M + H]+. (0139) A solution of (4) (0.09ml_, 0.61mmol) in 0.85M hydroxylamine in MeOH (10 mL) was stirred at rt for 72h. Solvent concentrated to dryness and the residue purified by reverse phase HPLC to give Example GG (23 mg, 12%). (0140) XH NMR (500 MHz, Methanol-*), d ppm: 8.66 (d, 7= 1.3 Hz, 2H), 8.28-8.36 (m, 2H), 8.16 (d, 7=2.6 Hz, 2H), 7.67 (d, 7=8.2 Hz, 2H), 7.45 (d, 7=8.2 Hz, 2H), 5.56 (s, 2H). (0141) LCMS (ES) : Found 323.1 [M + H]+. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping