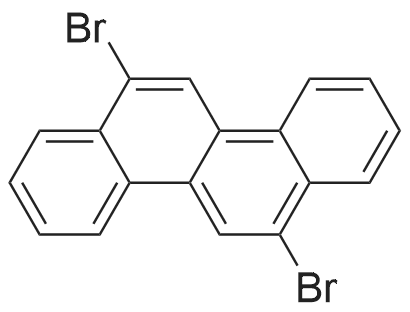
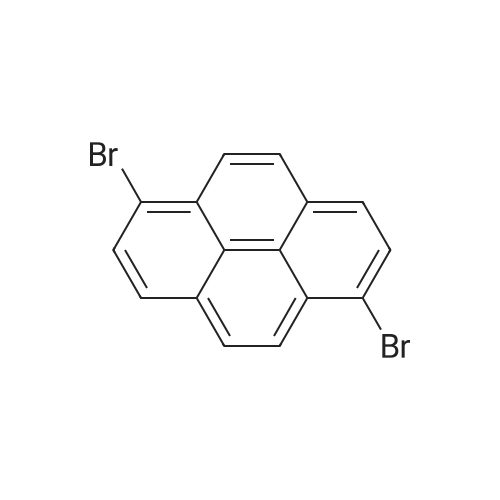
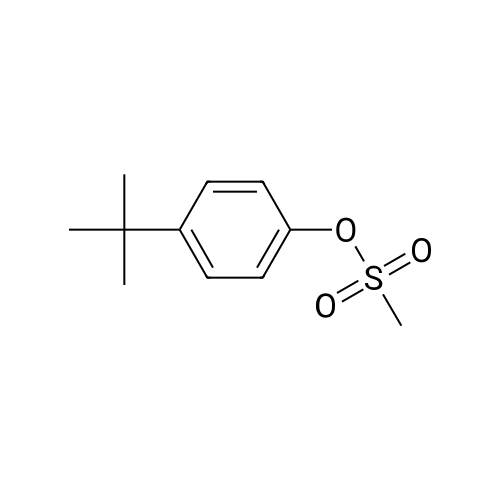
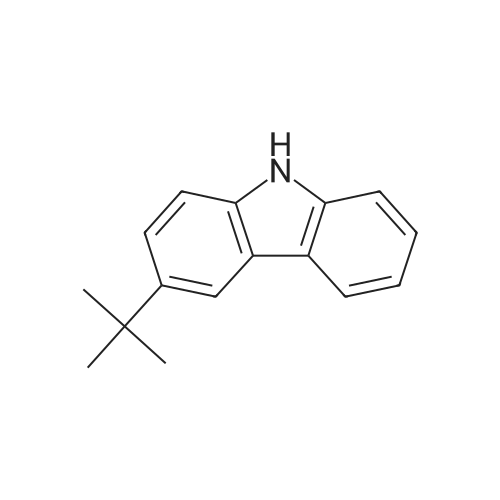
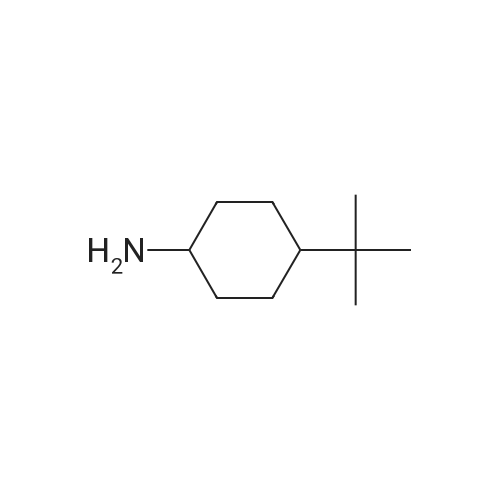
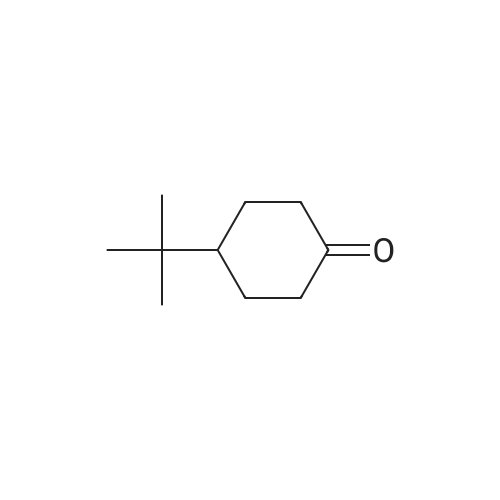
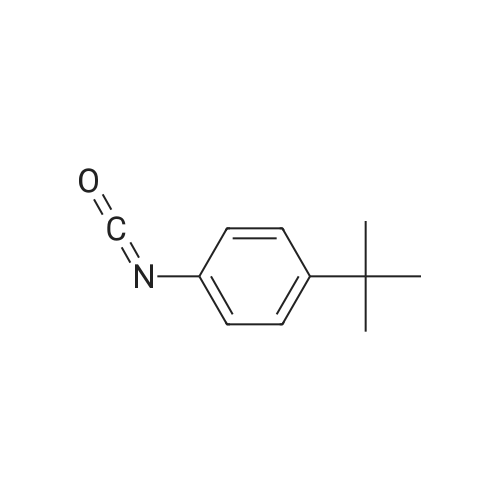
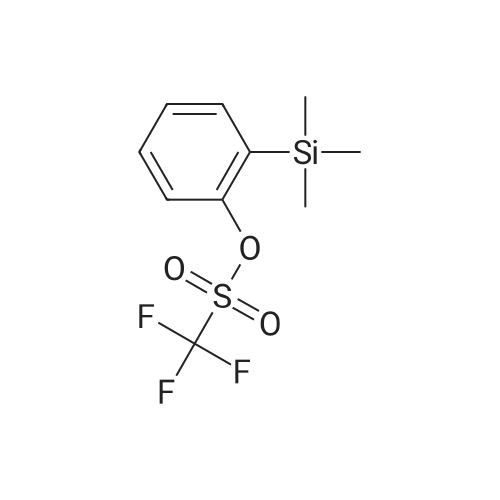
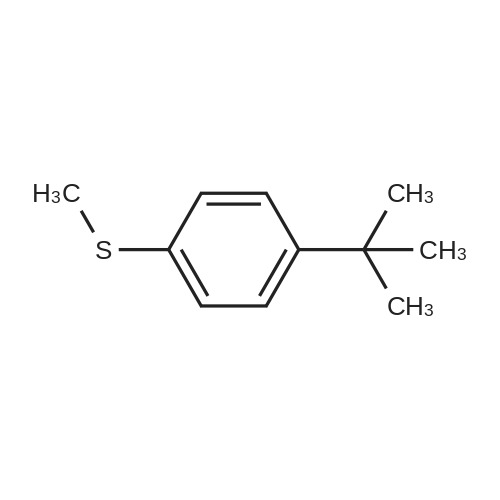
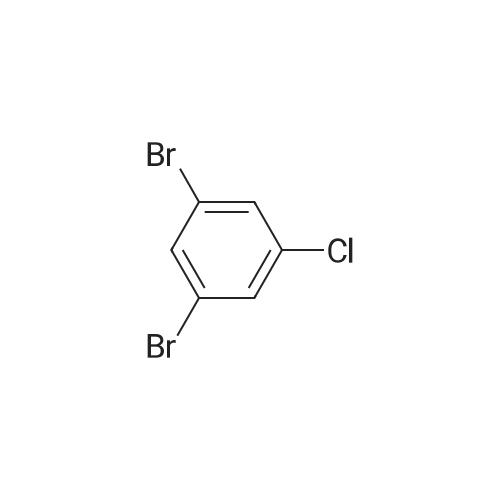
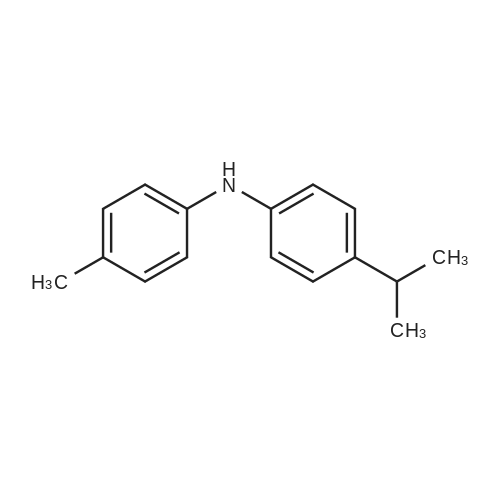
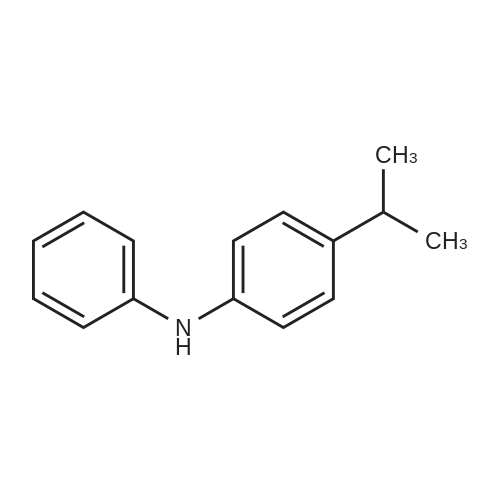
|
With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate; In toluene; at 100℃; for 24h; |
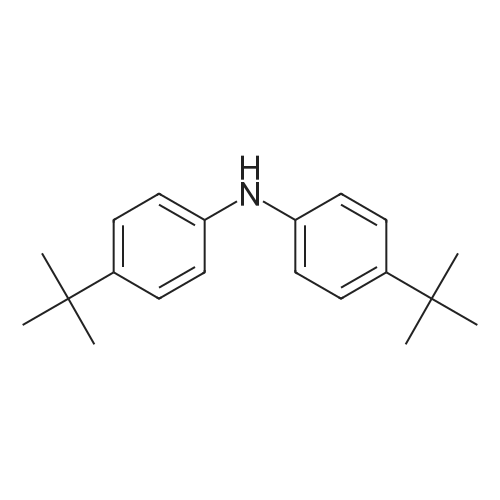
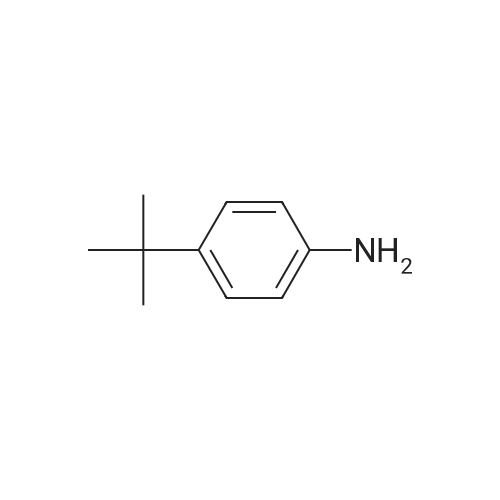
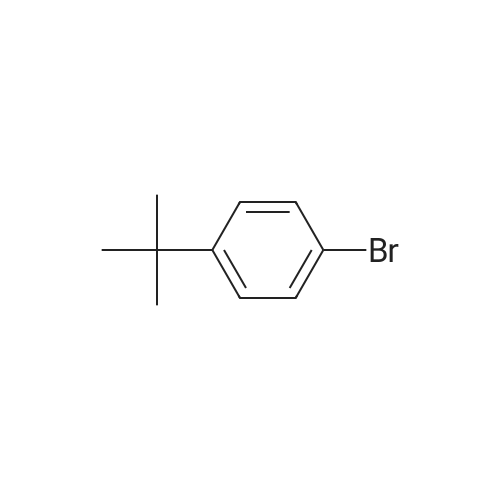
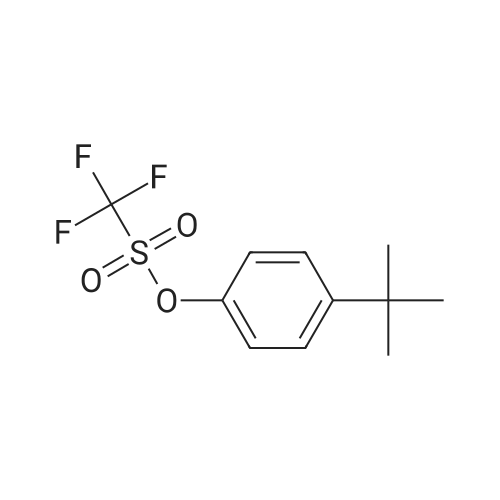
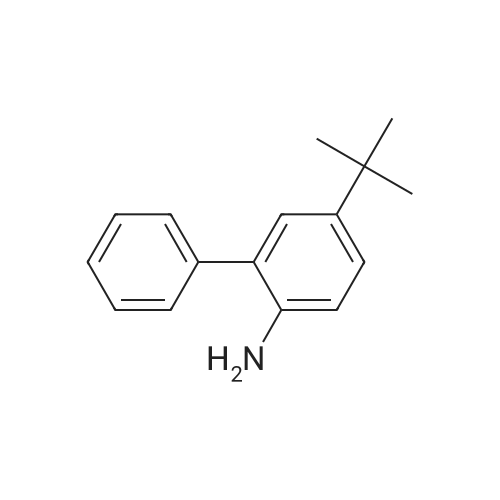
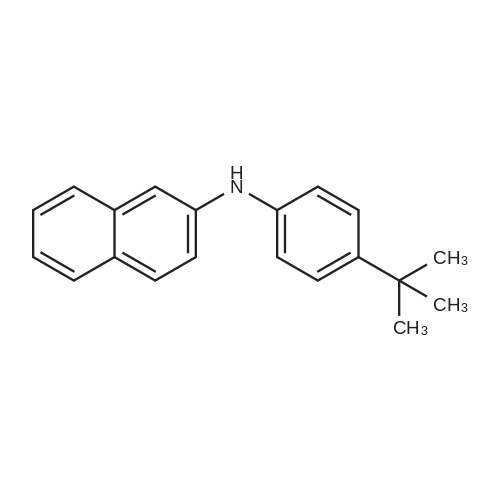
General procedure: 4-bromo-1,1'-biphenyl (5.6g, 24mmol) was dissolved in toluene, [1,1'-biphenyl]-4-amine (3.4g, 20mmol), Pd2(dba)3 (0.5g, 0.6mmol), P(t-Bu)3 (0.2g, 2mmol), NaOt-Bu (5.8g, 60mmol), toluene (300 mL) was added to each of the back, and refluxed under strring for 24 hours at 100C. After the reaction was terminated with water and extracted with ether. The organic layer was dried over MgSO4 and concentrated and to the resulting organic silicagel column and re-crystallization the final compound 6.2g (Yield 80%). |
|
With tris-(dibenzylideneacetone)dipalladium(0); triphenylphosphine; sodium t-butanolate; In toluene; at 100℃; for 24h; |
General procedure: To a round bottom flask Sub 1-3-1-1 (1.9g, 20mmol), Sub 1-3-2-1 (5.5g, 20mmol), Pd2 (dba) 3 (0.9g, 1mmol), PPh3 (0.5g , 2mmol), NaOt-Bu (5.8g, 60mmol), were added to toluene (210mL), respectively, and refluxed under stirring for 24 hours at 100C . Ether and a water extraction of organic layer after MgSO4to dry a formed after silicagel column Sub 1-3 (9) are the and recrystallization 5.2g a (yield: 64%)obtained. |
|
With tris-(dibenzylideneacetone)dipalladium(0); triphenylphosphine; sodium t-butanolate; In toluene; at 100℃; for 24h; |
General procedure: To a round bottom flask Sub 2-1-1 (1.9g, 20mmol), Sub 2-2-1 (5.5g, 20mmol), Pd2 (dba) 3 (0.9g, 1mmol), PPh3 (0.5g, 2mmol), NaOt-Bu (5.8g, 60mmol), were added to toluene (210mL), respectively, and stirred at reflux for 24 hours, 100 C thereby. The organic layer was dried over MgSO4, and extracted with water and ether and recrystallized silica gel column and the resulting organic one was concentrated to a Sub 2-9 5.2g (yield: 64%) was obtained. |
|
With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate; In toluene; at 100℃; for 24h; |
General procedure: 4-bromo-1,1'-biphenyl (5.6 g, 24 mmol) was dissolved in toluene and then [1,1'-biphenyl] -4-amine (3.4 g, 20 mmol), Pd2 (dba) (0.2 g, 2 mmol), NaOt-Bu (5.8 g, 60 mmol) and toluene (300 mL) were added to the reaction mixture, and the mixture was refluxed at 100 C for 24 hours. After the reaction was completed, the reaction mixture was extracted with ether and water. The organic layer was dried over MgSO 4 and concentrated. The resulting organic material was purified by silicagel column and recrystallized to obtain 6.2 g (yield: 80%) of final compound. |
|
With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate; In toluene; at 100℃; for 24h; |
General procedure: 4-bromo-1,1'-biphenyl (5.6 g, 24 mmol) was dissolved in toluene and then [1,1'-biphenyl] -4-amine (3.4 g, 20 mmol), Pd2 (dba) (0.2 g, 2 mmol), NaOt-Bu (5.8 g, 60 mmol) and toluene (300 mL) were added to the reaction mixture, and the mixture was refluxed at 100 C for 24 hours. After the reaction was completed, the reaction mixture was extracted with ether and water. The organic layer was dried over MgSO 4 and concentrated. The resulting organic material was purified by silicagel column and recrystallized to obtain 6.2 g (yield: 80%) of final compound. |
|
With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate; In toluene; at 100℃; |
General procedure: To a round bottom flask was added an amine compound (1 eq.),Bromine compound (1.1 eq.),Pd2 (dba) 3 (0.05 mol%),P (t-Bu) 3 (0.1 eq.),NaOt-Bu (3 eq.),toluene (10.5 mL / 1 mmol) is added and the reaction proceeds at 100 C.After completion of the reaction, the reaction mixture was extracted with ether and water. The organic layer was dried over MgSO 4 and concentrated. The resulting organic material was subjected to silicagel column and recrystallization to obtain product Sub 7. |
|
With tris-(dibenzylideneacetone)dipalladium(0); tri-tert-butyl phosphine; sodium t-butanolate; In toluene; at 100℃; |
General procedure: A round bottom flask was charged with an amine compound (1 eq), a bromine compound (1.1 eq), Pd2(dba)3 (0.05 mol%), P(t-Bu) 3 (0.1 equivalent), NaOt-Bu (3 equivalents)toluene (10.5 mL / 1 mmol) is added and the reaction proceeds at 100 C. After the reaction was completed, the reaction mixture was extracted with ether and water. The organic layer was dried over MgSO4 and concentrated. The resulting organic material was subjected to silicagel column and recrystallization to obtain product Sub 7. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping