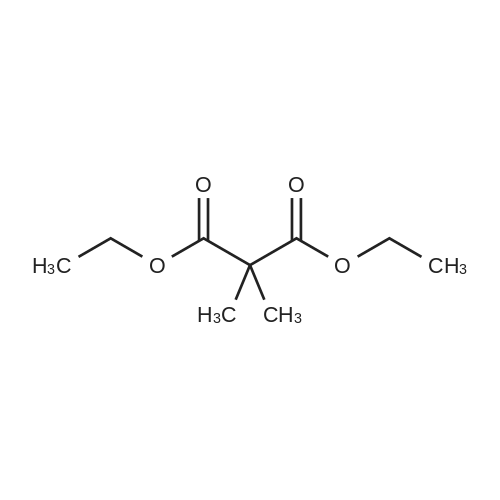
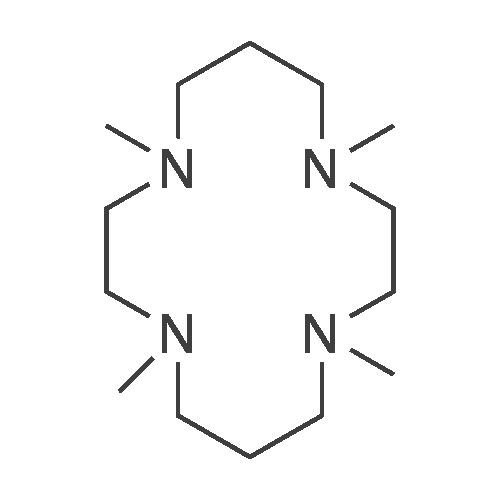
| 90% |
With phosphorus pentachloride; at 20℃; for 3h;Inert atmosphere; |
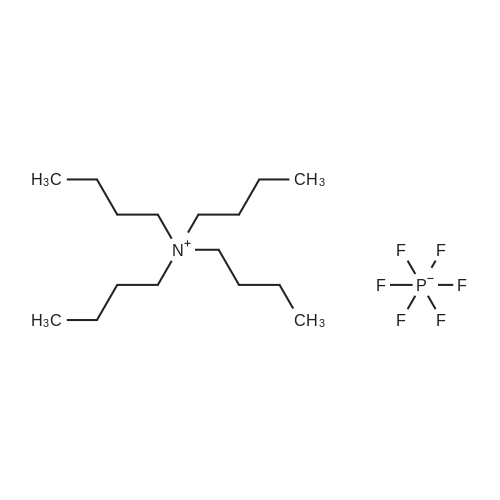
Example 8 was repeated with the difference:1. PC15 (30 mg, 6 mol %, the mol % being based onthe combined molar amount of [(n-Hu)4N] [HF4] and PC15)were used instead of FeC13.1Afterthe stirring for 3 han’‘Band ‘9F NMR spectrawere measured. In accordance to ‘9F NMR and ‘H NMR theproduct contained 100% of compound of formula (1).1 After removing the solvent the obtained white solidsubstance was dried at 50 C. in vacuo to yield 0.680 g (90%,1.66 mmol) of compound of formula (1).C/H/N Analysis calc. % (found): C, 65.14 (64.44); H, 10.36 (10.41); N, 15.99 (16.20).1‘H NMR (25 C., CDC13, 300.13 MHz, delta in ppm): 1.00 (t, 12H, CH3), 1.41 (m, 8H, CH3-CH2), 1.60 (m, 8H, CH2-CH2N), 3.12 (m, 8H, NCH2)“H NMR (25 C., CDC13, 300.13 MHz, delta in ppm): -17.6 (d, 1H, HF(CN)3, ‘J(”H-’9F)=45 Hz) ‘3C NMR (25 C., CDC13, 300.13 MHz, delta in ppm): 13.3 (s, 4C, CH3), 19.4 (t, 4C, CH2-CH3), 23.6 (s, 4C, N-CH2---CH2), 58.7 (t, 4C, NCH2), 127.2 (dq, 3C, HF(CN)3, ‘J(’3C-’9F)=38 Hz, ‘J(’3C-”H)=75 Hz) ‘9F NMR (25 C., CDC13, 300.13 MHz, delta in ppm): -210.9 (q, iF, HF(CN)3, ‘J(”H-’9F)=45 Hz)IR (ATR, 32 scans, v in cm’): 2964 (m), 2935 (m),2876 (m), 2214 (w), 1474 (m), 1381 (m), 1171 (w), 1040 (m), 960 (w), 938 (,), 903 (s), 803 (w), 736 (m), 536 (w); [(n-Hu)4N][HF4] (0.491 g, 1.49 mmol), prepared according to Preparation Description A, FeC13 (20 mg, 7 mol %, the mol % being based on the combined molar amount of [(n-Hu)4N][HF4] and FeC13) and (CH3)3SiCN (1.58 g, 1.59 mmol) were stirred under argon atmosphere at ambient temperature for 3 h.After the stirring at ambient temperature for 3 h an11H and ‘9F NMR spectra were measured. In accordance to‘9F NMR and “H NMR the product contained 100% of compound of formula (4). Afterremoving the solvent the obtained light yellow solid substance was dried at 50 C. in vacuo to yield 0.400 g (69%, 1.17 mmol) of compound of formula (4). |
| 83% |
|
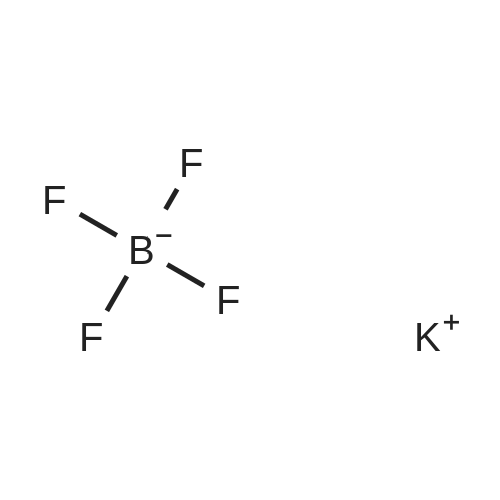
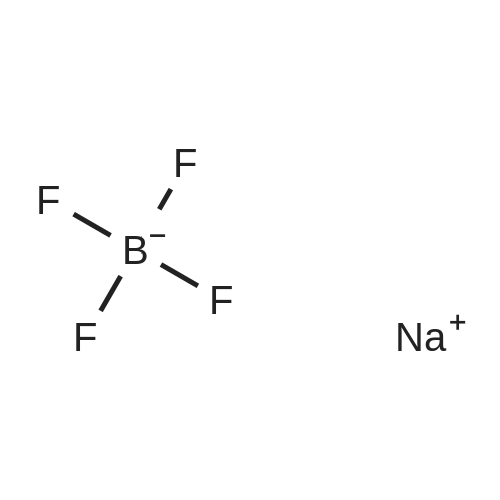
[(n-Bu)4N][BF4] (1.17 g, 3.55 mmol), prepared according to example 12, and (CH3)3SiCN (1.23 g, 12.44 mmol) were filled under argon atmosphere with a residual oxygen content of below 5 ppm and with a residual water content of below 1 ppm into a teflon tube. The teflon tube was closed using a teflon lid and place in an autoclave. The autoclave wasplaced inside a muffle furnace which was heated to 150C in 30 mm. This temperature was held for 13 h.After cooling to ambient temperature the reaction mixture was mixed with water and aqueous hydrogen peroxide (0.5 ml, 5 mmol, 30 wt%). After stirring at 60C for an hour the solution was cooled to ambient temperature. The product was extracted with dichloromethane threetimes. The organic phase was separated and dried with Mg504 and filtered. The filtrate was evaporated on a rotary evaporator. The yield of the yellow crystalline product was 0.95 g (76%, 2.71 mmol). Only one boron and one fluorine species, the one of the desired product, is visible in 11B NMR and in the 19F NMR respectively. AnalyticsMp: 60CC/H/N Analysis caic. % (found): C 65.14 (64.90), H 10.36 (10.15), N 15.99 (15.87)‘H NMR (25C, CD3CN, 300.13 MHz, delta in ppm): 0.97 (t, 12H, CH3, 3J(1H-1H) = 7.2 Hz),1.36 (m, 8H, CH3-CH2, 3J(1H-1H) = 7.6 Hz), 1.59 (m, 8H, CH2-CH2N), 3.08 (m, 8H,NCH2)‘3C NMR(25C, CD3CN, 250.13 MHz, delta in ppm): 13.78 (s, 4C, CH3), 20.30 (m, 4C, CH3-CH2), 24.29 (m, 4C, CH2-CH2N), 59.33 (m, 4C, NCH2), 127.83 (3C, CN)“B NMR (25C, CD3CN, 96.29 MHz, delta in ppm): -17.87 (d, 1B, BF(CN)3, 1J(13C-11B =44.02 Hz)‘9FNMR(25C, CD3CN, 300.13 MHz, delta in ppm): -211.73 (q, iF, 1J(11B-19F)=44.96Hz, 2J(10B-19F) = 34.74 Hz)IR (ATR, 32 scans, v in cm1): 2964 (m), 2935 (m), 2876 (m), 2214 (w), 1474 (m), 1408(w), 1382 (w), 1361(w), 1350 (w), 1322 (w), 1311 (w), 1285(w), 1244 (w), 1171 (w), 1130(w), 1110 (w), 1080 (w), 1040 (m), 991 (w), 959 (m), 938 (m), 926 (m), 903 (s), 821 (w),803 (w), 736 (m), 668 (w), 592 (w), 532 (w)RAMAN (500 mW, 400 scans cm1): 2971 (5), 2941 (7), 2878 (6), 2219 (10), 1452 (3), 1324 (2), 1113 (2), 1062 (1), 910 (1), 883 (1), 594 (1), 264 (1), 224 (1), 130 (2);_Example 1 was repeated with the differences:1. [(n-Bu)4N][BF4] (17.957 g, 54.5 mmol) and (CH3)3SiCN (21.1 g, 213 mmol) were used.2. The temperature was 140C instead of 150C, which was held for 20 h instead of 13 h.3. 5 ml of the aqueous hydrogen peroxide were used instead of 0.5 ml.A light yellow crystalline product was obtained (15.847 g, 83%, 45.2 mmol). |
| 76% |
|
Example 1: Synthesis of [(n-Bu)4N] [BF(CN)3] [0063] [(n-Bu)4N][BF4] (1.17 g, 3.55 mmol), prepared according to example 12, and (CH3)3SiCN (1.23 g, 12.44 mmol) were filled under argon atmosphere with a residual oxygen content of below 5 ppm and with a residual water content of below 1 ppm into a teflon tube. The teflon tube was closed using a teflon lid and place in an autoclave. The autoclave was placed inside a muffle furnace which was heated to 150C in 30 min. This temperature was held for 13 h. After cooling to ambient temperature the reaction mixture was mixed with water and aqueous hydrogen peroxide (0.5 ml, 5 mmol, 30 wt%). After stirring at 60C for an hour the solution was cooled to ambient temperature. The product was extracted with dichloromethane three times. The organic phase was separated and dried with MgSO4 and filtered. The filtrate was evaporated on a rotary evaporator. The yield of the yellow crystalline product was 0.95 g (76%, 2.71 mmol). Only one boron and one fluorine species, the one of the desired product, is visible in 11B NMR and in the 19F NMR respectively. Analytics Mp: 60C [0064] C/H/N Analysis calc. % (found): C 65.14 (64.90), H 10.36 (10.15), N 15.99 (15.87) 1H NMR (25C, CD3CN, 300.13 MHz, delta in ppm): 0.97 (t, 12H, CH3, 3J(1H-1H) = 7.2 Hz), 1.36 (m, 8H, CH3-CH2, 3J(1H-1H) = 7.6 Hz), 1.59 (m, 8H, CH2-CH2N), 3.08 (m, 8H, NCH2) 13C NMR (25C, CD3CN, 250.13 MHz, delta in ppm): 13.78 (s, 4C, CH3), 20.30 (m, 4C, CH3-CH2), 24.29 (m, 4C, CH2-CH2N), 59.33 (m, 4C, NCH2), 127.83 (3C, CN) 11B NMR (25C, CD3CN, 96.29 MHz, delta in ppm): -17.87 (d, 1B, BF(CN)3, 1J(13C-11B = 44.02 Hz) 19F NMR (25C, CD3CN, 300.13 MHz, delta in ppm): -211.73 (q, 1F, I1J(11B-19F) = 44.96 Hz, 2J(10B-19F) = 34.74 Hz) IR (ATR, 32 scans, ν in cm-1): 2964 (m), 2935 (m), 2876 (m), 2214 (w), 1474 (m), 1408(w), 1382 (w), 1361(w), 1350 (w), 1322 (w), 1311 (w), 1285(w), 1244 (w), 1171 (w), 1130 (w), 1110 (w), 1080 (w), 1040 (m), 991 (w), 959 (m), 938 (m), 926 (m), 903 (s), 821 (w), 803 (w), 736 (m), 668 (w), 592 (w), 532 (w) RAMAN (500 mW, 400 scans cm-1): 2971 (5), 2941 (7), 2878 (6), 2219 (10), 1452 (3), 1324 (2), 1113 (2), 1062 (1), 910 (1), 883 (1), 594 (1), 264 (1), 224 (1), 130 (2) |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 HazMat Fee +
HazMat Fee +

 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping