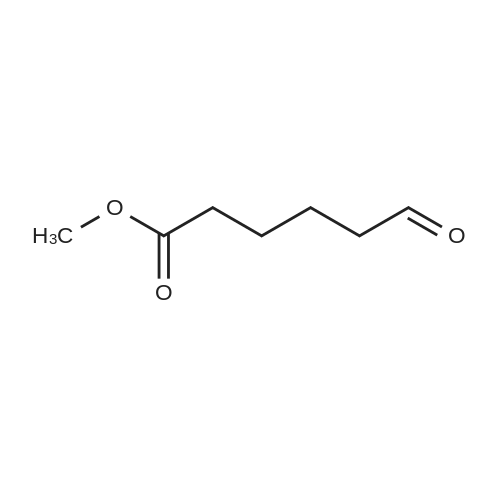
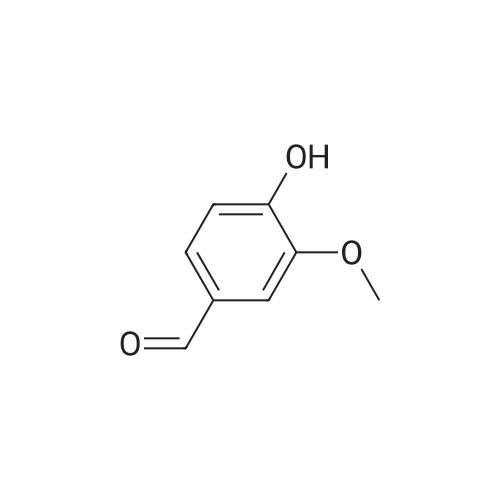
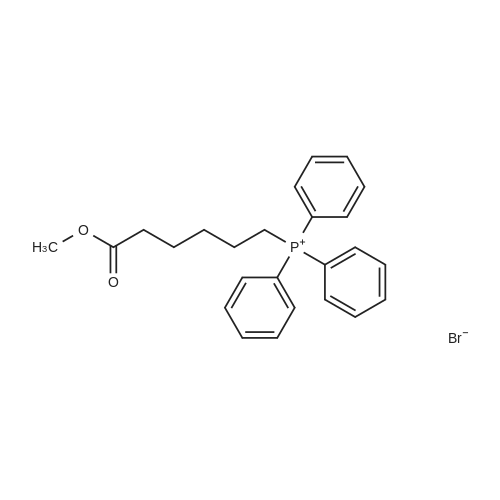
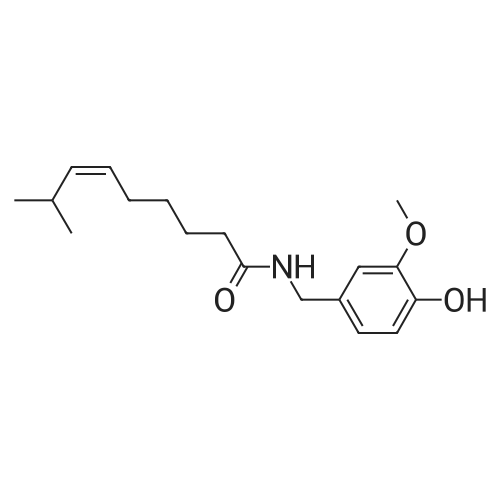
| 8.73 kg |
With 9-bora-bicyclo[3.3.1]nonane; In tetrahydrofuran; at 20 - 25℃;Inert atmosphere; Large scale; |
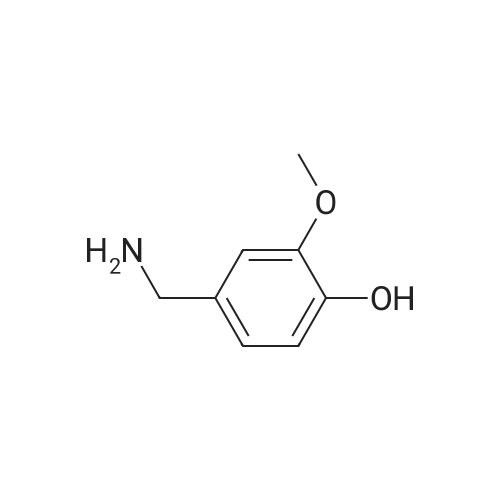
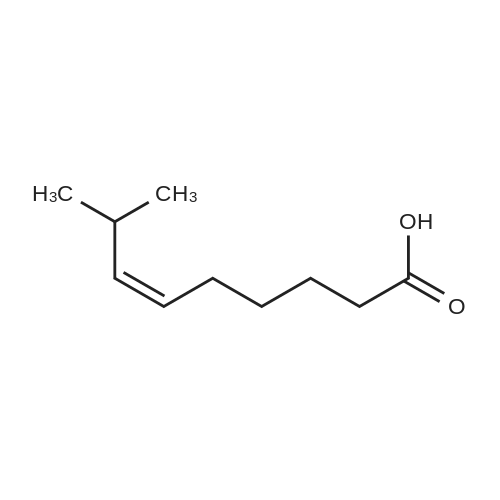
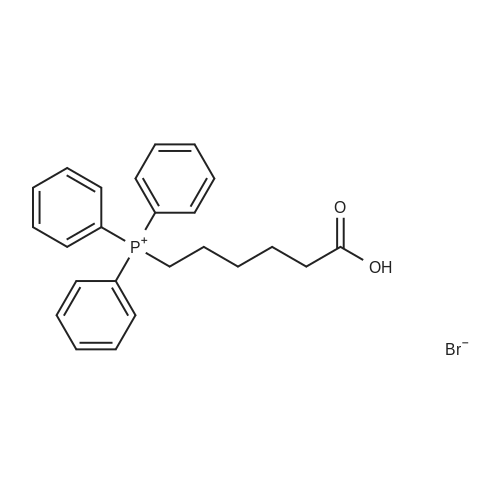
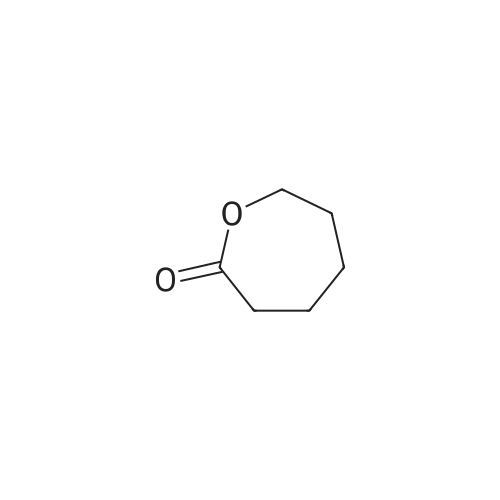
Under nitrogen protection,Add Zucapsaicin intermediate IV 12.0kg to anhydrous tetrahydrofuran 84L with stirring.(39.55 mol, HPLC purity 99.32%),Add 0.5 mol/L of 9-BBN (9-borobicyclo[3.3.1]nonane) tetrahydrofuranLiquid 99L (49.5 mol);Stir at 20 to 25 C for 6 to 8 hours. Samples are taken every hour for HPLC testing starting at 4 hours.Until the rutheid intermediate IV is not more than 1.0% and the E isomer is not more than 0.2%.In the fourth hour, the intermediate IV of Zucapsaicin was 1.71%.The E isomer was not detected; the fifth phase of the Zucapsaicin intermediate IV was 1.26%.The E isomer is about 0.01%; the 6th hour of the ruthenium intermediate IV is 0.82%,The E isomer is 0.03%; the seventh hour of the Zucapsaicinintermediate IV is 0.66%.The E isomer is 0.06%; the 8th hour, the Zucapsaicin intermediate IV is 0.52%,The E isomer is 0.08%. Therefore, the reaction was terminated after 6 to 8 hours. Another 2 mL of the reaction solution was taken, and the reaction was continued under the conditions. In the 12th hour, the intermediate IV of the Zucapsaicin was 0.35%, the E isomer was 0.18%, and the intermediate IV of the hexacycline was 0.22%. The E isomer was 0.26%; the hexadecane intermediate IV was 0.07% at the 24th hour and the E isomer was 0.39%. 45 g (0.75 mol) of acetic acid was added to 4.45 Kg (139 mol) of methanol, and the mixed solution was added to the reaction system for quenching. Heat to 45 ~ 50 C for 3 to 4 hours, cool to 0 ~ 5 C,Adding a mass concentration of 3% aqueous hydrochloric acid (the mass concentration refers to the mass of hydrogen chloride as a percentage of the total mass of the aqueous hydrochloric acid solution) and stirring for 1 hour. It is then treated to near neutral with a mass concentration of 7% aqueous solution of sodium bicarbonate (the mass concentration means the mass of sodium bicarbonate as a percentage of the total mass of the aqueous solution of sodium bicarbonate), and the insoluble matter is removed by filtration. Concentrated in vacuum (temperature 35 C ~ 55 C,The pressure is -0.08MPa~-0.1MPa). Most of the organic solvent is removed, cooled to room temperature, and extracted twice with isopropyl ether 40L; the organic phase is combined.Stirring, standing, and mass concentration of 10% aqueous sodium chloride solution (the mass concentration refers to the mass of sodium chloride as a percentage of the total mass of sodium chloride water)The water layer is separated. The organic layer is concentrated in a vacuum (temperature 35 C ~ 45 C, pressure -0.08 MPa ~ -0.1 MPa) to remove about 1/2 of the solvent, heated to 50 ~ 60 C,Slowly add 40 L of n-heptane, and then cool to 10 to 15 C and stir for 2 to 3 hours. Filter, rinse three times with n-heptane, and drain. Add 35L of isopropyl ether to the wet product.Heat to 50 ~ 60 C, slowly add n-heptane 35L,Cool again to 10-15 C and stir for 2 to 3 hours. filter,Rinse three times with n-heptane and drain. Drying under vacuum (vacuum degree -0.01 MPa to -0.1 MPa, temperature 45 to 55 C) for 12 to 16 hours,The Zucapsaicin I 8.73 Kg was obtained, the yield was 72.3%, and the total yield in three steps was 67.0%. HPLC purity 99.92%, E isomer 0.04%, other largest single An impurity of 0.05%. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping