Alternatived Products of [ 17495-12-4 ]
Product Details of [ 17495-12-4 ]
| CAS No. : | 17495-12-4 |
MDL No. : | MFCD04145136 |
| Formula : |
C12H11N5O
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
241.25
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 17495-12-4 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 17495-12-4 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 17495-12-4 ]
- Downstream synthetic route of [ 17495-12-4 ]
- 1
-
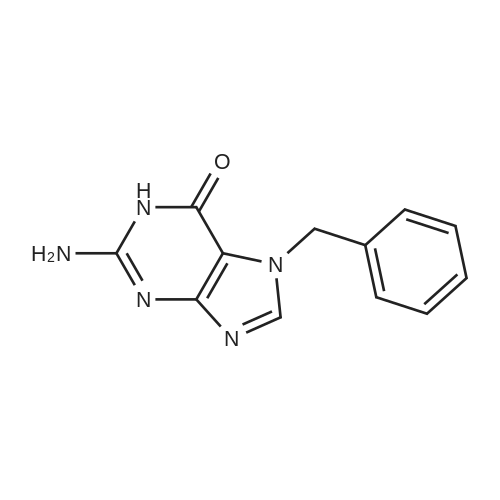 [ 17495-12-4 ]
[ 17495-12-4 ]

-
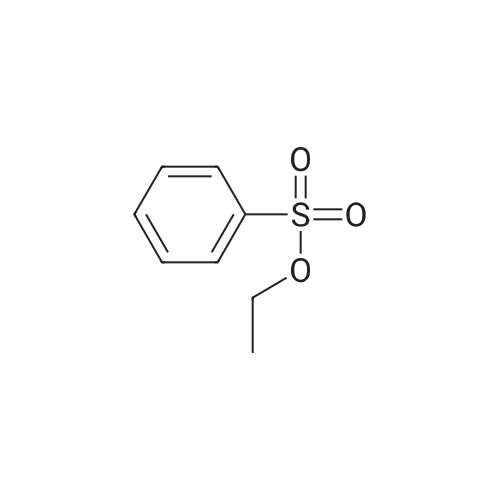 [ 515-46-8 ]
[ 515-46-8 ]

-
2-amino-7-benzyl-9-ethyl-6-oxo-6,7-dihydro-1<i>H</i>-purin-9-ium; benzenesulfonate
[ No CAS ]
- 2
-
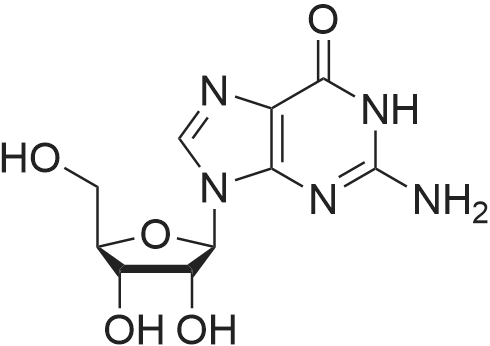
 [ 100-39-0 ]
[ 100-39-0 ]

-
 [ 118-00-3 ]
[ 118-00-3 ]

-
 [ 17495-12-4 ]
[ 17495-12-4 ]
| Yield | Reaction Conditions | Operation in experiment |
| 85% |
Stage #1: benzyl bromide; GUANOSINE In dimethyl sulfoxide at 20℃; for 24h;
Stage #2: With hydrogenchloride In dimethyl sulfoxide at 70℃; for 2h; |
|
|
In dimethyl sulfoxide at 20℃; for 4h; |
|
|
Stage #1: benzyl bromide; GUANOSINE In dimethyl sulfoxide at 50℃; for 3h;
Stage #2: With hydrogenchloride In dimethyl sulfoxide at 20 - 70℃; for 5h; |
5.16 Intermediate 5.16
To a mixture of 2-amino-9-((2R,3R,4S,5R)-3,4-dihydroxy-5-hydroxymethyl-tetrahydro- furan-2-yl)-1 ,9-dihydro-purin-6-one (50.0 g, 177 mmol) in DMSO (133 mL) bromomethyl-benzene (25.2 mL, 212 mmol) was added dropwise. The resulting mixture was stirred for 3 h at 50 °C. The mixture was cooled to rt and HCI solution (4 mol/l, 102 mL, 406 mmol) was added dropwise. The mixture was stirred for 5 h at 70 °C, then at rt overnight. The obtained precipitate was filtered, washed with cold MeOH and dried to obtain the product.MS (ESI+): (M+H)+242HPLC: RT = 0.28 min, Method D |
| 41.8 g |
In dimethyl sulfoxide at 50℃; for 4h; |
Intermediate 20.1
To a mixture of 2-amino-9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yI]-6,9-dihydro-1 H-purin-6-one (60 g, 211.8 mmol) in DMSO (160 ml, 2.25 mol)(bromomethyl)benzene (30.2 ml, 254.2 mmol) was added dropwise and the mixture wasstirred 4 h at 50°C. The mixture was cooled and HCI (4 mol/l, 122 mL, 487.2 mmol) wasadded dropwise and the mixture was stirred 2h at 70°C. The mixture was cooled,filtered, washed with MeOH and dried to obtain 41.8 g of the product.MS (ESI): (M+H) 242HPLC: RT = 0.28 mm, Method D |
|
Multi-step reaction with 2 steps
1: dimethyl sulfoxide / 24 h / 20 - 70 °C
2: hydrogenchloride / dimethyl sulfoxide; lithium hydroxide monohydrate / 2 h / 70 °C |
|

Reference:
[1]Kalayanov, Genadiy; Jaksa, Suzana; Scarcia, Tommaso; Kobe, Joze
[Synthesis, 2004, # 12, p. 2026 - 2034]
[2]Balssa, Frederic; Bonnaire, Yves
[Journal of labelled compounds and radiopharmaceuticals, 2007, vol. 50, # 1, p. 33 - 41]
[3]Current Patent Assignee: HYDRA BIOSCIENCES, INC; C.H. Boehringer Sohn AG & Co. KG - WO2019/11802, 2019, A1
Location in patent: Page/Page column 38
[4]Current Patent Assignee: C.H. Boehringer Sohn AG & Co. KG; HYDRA BIOSCIENES; HYDRA BIOSCIENCES, INC - WO2020/120450, 2020, A1
Location in patent: Page/Page column 106-107
[5]Baranowski, Marek R.; Basquin, Jerome; Jemielity, Jacek; Kowalska, Joanna; Kozarski, Mateusz; Kubacka, Dorota; Panecka‐hofman, Joanna; Strzelecka, Dominika; Wojcik, Radoslaw
[Pharmaceuticals, 2022, vol. 15, # 5]
- 3
-
 [ 17495-12-4 ]
[ 17495-12-4 ]

-
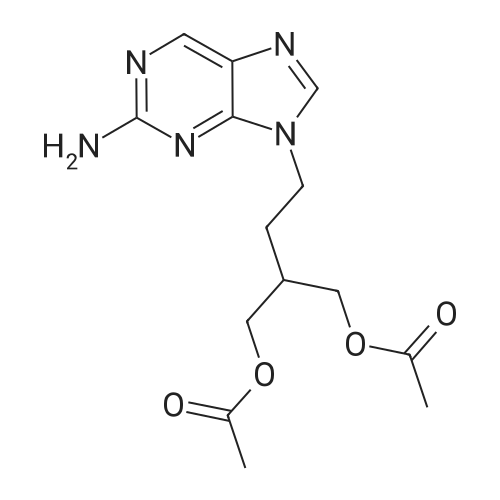 [ 104227-87-4 ]
[ 104227-87-4 ]
- 4
-
 [ 17495-12-4 ]
[ 17495-12-4 ]

-
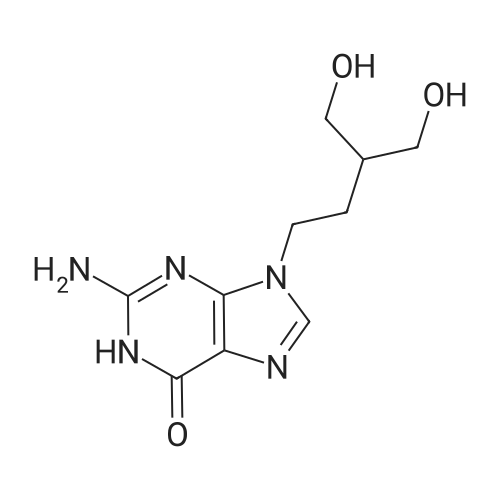 [ 39809-25-1 ]
[ 39809-25-1 ]
- 5
-
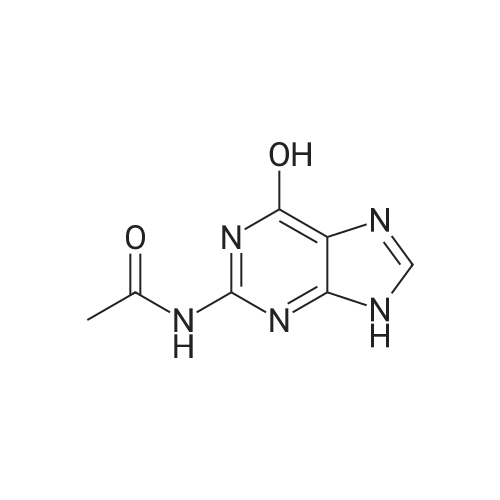 [ 19962-37-9 ]
[ 19962-37-9 ]

-
 [ 17495-12-4 ]
[ 17495-12-4 ]
- 6
-
 [ 17495-12-4 ]
[ 17495-12-4 ]

-
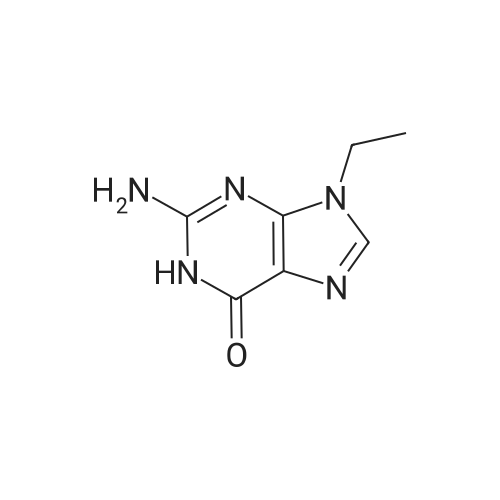 [ 879-08-3 ]
[ 879-08-3 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 2 steps
1: 90 percent / 2 h / 135 - 145 °C
2: 53 percent / H2 / Pd/C / ethanol / 75 - 80 °C |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping











