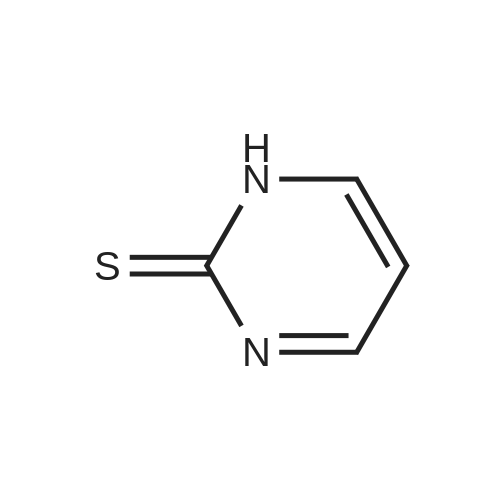
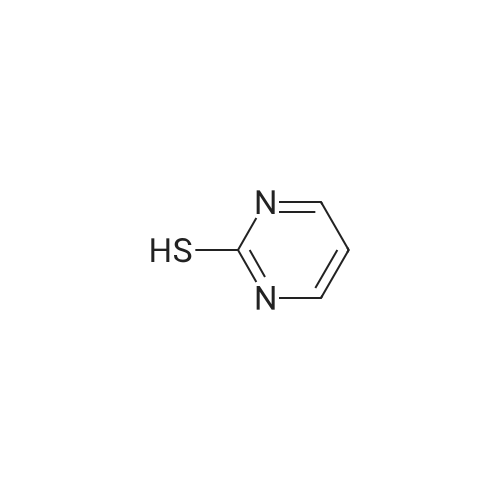
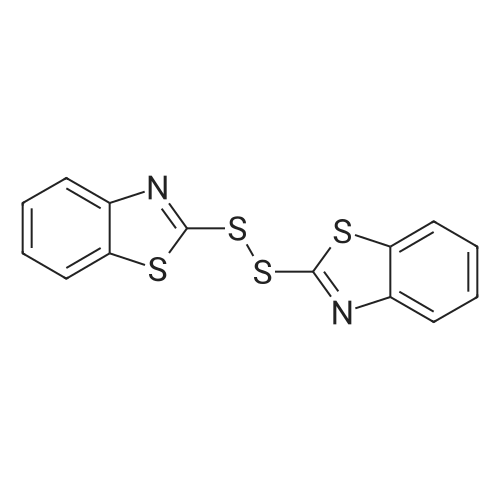
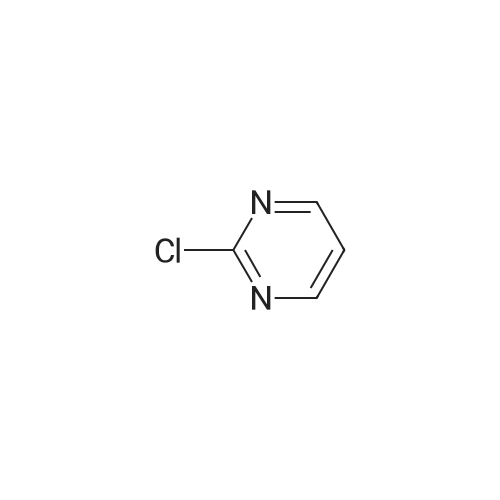
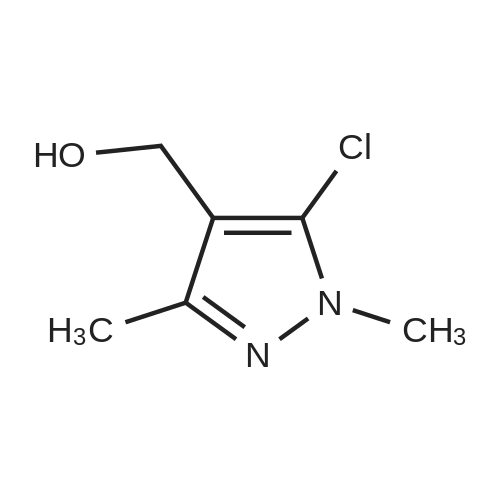
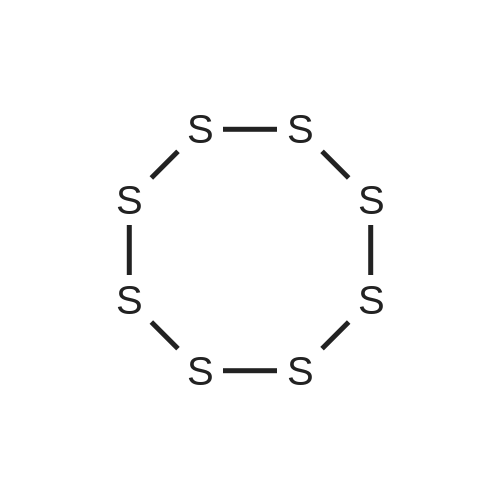
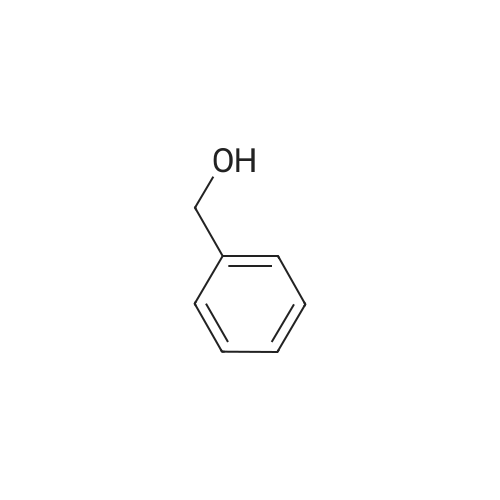
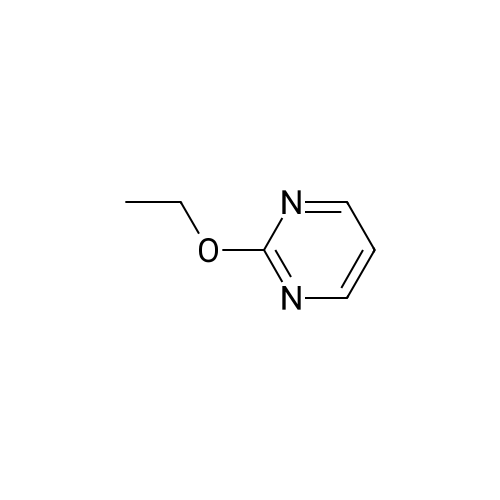
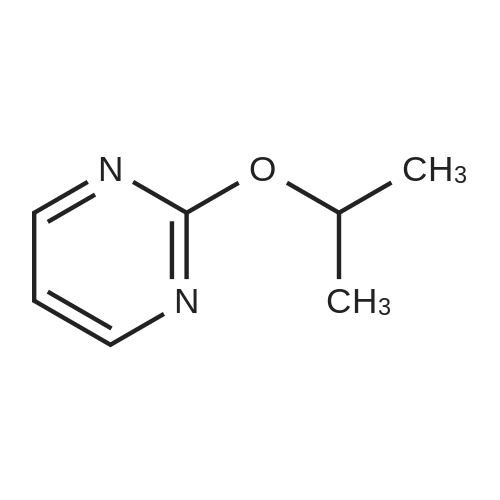
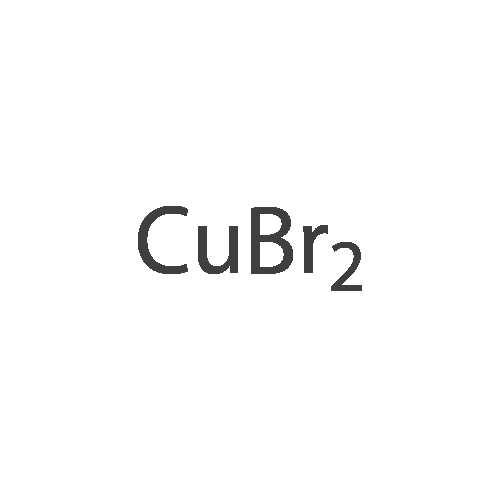| 97% |
With 1,4-dibenzyl-1,4-diazoniabicyclo[2.2.2]octane chlorochromate In acetonitrile for 0.25h; Heating; |
|
| 96% |
With water; dihydrogen peroxide In various solvent(s) at 20℃; for 0.166667h; |
|
| 95% |
With benzyltriphenylphosphonium peroxodisulfate In acetonitrile for 0.75h; Heating; |
|
| 95% |
With sodium periodate In water at 20℃; for 0.025h; |
|
| 94% |
With melamine hydrogen peroxide In acetonitrile for 0.5h; Reflux; |
|
| 93% |
With 1-benzyl-4-aza-1-azoniabiyclo<2.2.2>octane peroxodisulfate In acetonitrile for 0.5h; Heating; |
|
| 93% |
With ammonium peroxydisulfate at 20℃; for 0.25h; |
|
| 93% |
With butyltriphenylphosphonium dichromate In acetonitrile for 0.0333333h; Microwave irradiation; |
|
| 90% |
With zinc bismuthate In toluene for 1h; Heating; |
|
| 90% |
With butyltriphenylphosphonium dichromate In chloroform for 0.25h; Heating; |
|
| 90% |
With tungstate sulfuric acid; sodium nitrite In dichloromethane at 20℃; for 0.0833333h; |
|
| 90% |
With sodium nitrite In water at 20℃; for 0.0666667h; |
|
| 90% |
With [Mn(III)(BHBPDI)Cl]; oxygen In ethanol at 20℃; for 1.66h; |
|
| 90% |
With silica gel-supported iodine monochloride In chloroform at 20℃; for 1.16667h; |
|
| 90% |
With potassium bromate; hexaammonium heptamolybdate tetrahydrate In water; acetonitrile at 20℃; for 0.75h; |
Typical Procedure
General procedure: 4-chlorothiophenol (0.145 g, 1 mmol) was added to a heterogeneous mixture of KBrO3 (0.167 g, 1 mmol), (NH4)6Mo7O24·4H2O (0.124 g, 10 mmol%), and CH3CN/H2O (7:3) (5 mL) and the mixture was stirred under a hood for 4 min at ambient atmosphere and room temperature. The progress of the reaction was monitored by TLC (eluent: EtOAc/n-C6H14, 1/13). After completion, CH2Cl2 (20 mL) was added and the reaction mixture was filtered. The filtrate was washed with 5% NaOH, water, and dried over anhydrous MgSO4. Finally, evaporation of the solvent gave the product with sufficient purity formost purposes |
| 90% |
With laccase from Trametes versicolor; oxygen; 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt In methanol at 20℃; for 5h; Green chemistry; Enzymatic reaction; |
|
| 90% |
With Porcine Pancreas Lipase In water at 20℃; for 26h; Green chemistry; Enzymatic reaction; |
|
| 88% |
With silver bromate In diethyl ether; ethanol for 0.2h; |
|
| 88% |
With bovine serum albumin In water at 20℃; for 16h; Enzymatic reaction; |
2.3.1. General procedure for the synthesis of disulphides (1b-27b)
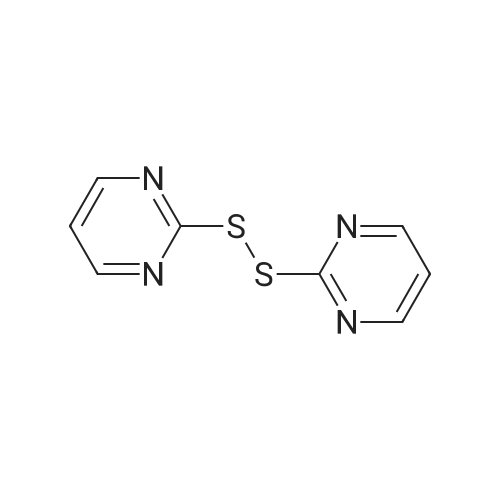
Thiophenol (0.25 mmol), BSA (50 mg) and deionized water(600 L) were stirred at room temperature for 12 h or till com-pletion of reaction (monitored by TLC). Then water (2 mL) wasadded to reaction mixture and extracted with ethyl acetate(3 mL 2), organic part dried over Na2SO4, ltered and then con-centrated under rotary evaporator and chromatographed oversilica gel by hexane/ethyl acetate (99:1). The obtained disulphides(1b-27b) were characterized and conrmed by comparing the1H NMR and 13C NMR data with those reported in the literature[38-51,108-116]. 2.4.21. Bis(pyrimidin-2-yl)disulphide (21b) [45]Yellow solid, mp: 144-145 C (lit [45] 143-145 C), 1H NMR(DMSO-d6 + CDCl3, 400 MHz): 8.47-8.46 (4H, m), 7.08-7.06 (2H,m); 13C NMR (DMSO-d6 + CDCl3, 100 MHz): 173.8, 163.0, 123.0 |
| 83% |
With (Bu4N)2S2O8 at 20℃; for 0.0833333h; |
|
| 83% |
With hydrogenchloride; tetrabutylammonium perchlorate; oxygen In water; acetonitrile for 0.25h; Electrochemical reaction; Flow reactor; Green chemistry; |
|
| 81% |
With rose bengal In isopropyl alcohol at 20℃; for 3h; Irradiation; |
4.2 General procedure A for synthesis of disulfides in iPrOH
General procedure: To a mixture of thiol (0.35mmol) in iPrOH in a 10mL vial, Rose Bengal (0.05equiv) were added and the reaction mixture was stirred at room temperature under white LED irradiation. The reaction mixture was quenched by addition of saturated aqueous NaOH (10mL), extracted with Et2O (3×10mL), dried over Na2SO4 and evaporated under reduced pressure to give the desired product. |
| 80% |
With 18-crown-6 ether; carbon tetrabromide; potassium carbonate In benzene at 80℃; for 5h; |
|
| 77% |
With manganese(II)carbonate; 3,4,5-trihydroxybenzoic acid; oxygen; sodium carbonate In water at 80℃; for 4h; Schlenk technique; Green chemistry; |
|
| 76% |
With maleic anhydride; urea-hydrogen peroxide In methanol at 0℃; for 1h; |
|
| 76% |
With 1H-imidazole; [N,N'-bis(salicylidene)-o-phenylenediaminato]manganese(III) chloride; urea hydrogen peroxide In methanol at 0℃; for 1h; |
|
| 75% |
With [bis(acetoxy)iodo]benzene In isopropyl alcohol at 20℃; for 0.0833333h; Green chemistry; |
|
| 72% |
With Fe(BTC); oxygen In acetonitrile at 70℃; for 2h; |
|
| 70% |
With pyridine; benzenesulfonyl chloride In dichloromethane Ambient temperature; |
|
| 69% |
With sulfuryl dichloride In dichloromethane for 0.5h; cooling; |
|
| 22% |
With oxygen Enzymatic reaction; |
|
|
With iodine In perchloric acid var. ionic strength; |
|
|
With hexachloroiridate(IV) |
|
|
With Ag(cyclam)(2+) at 25 - 35℃; |
|
|
With Ag(tmc)(2+) |
|
|
With Ag(<15>aneN4)(2+) |
|
|
With iodine In perchloric acid |
|
|
With hydrogen; water-d2 for 24h; Heating; |
|
|
With [K.18-crown-6][Br3]n In acetonitrile at 20℃; |
|
|
With iodine; iron(II) chloride In dichloromethane |
|
| 61 %Spectr. |
With L-arginine; oxygen In water at 50℃; for 1.5h; Green chemistry; chemoselective reaction; |
|
|
With propyl bromide; dimethyl sulfoxide at 25℃; for 24h; |
|
|
With iodine In water; acetonitrile at 0℃; for 1h; |
793.3 Step 3: i.2-Di(PYR1M1DIN-2-YL)DISULFANE
To a solution of 2-mercapto-pyrirnidine (2.0 g, 18 mrnoi) in acetonitril (80 mL) and water (16 mL) was added iodine (0.459 mL, 8.92 mrnol). The resultingmixture was stirred at r.t. for I h. After concentration water was added and it was extracted with DCM (3x120 mL). The combined organic phase was washed with 30 mL of sodium thiosulfate (1 M), brine and dried over anhydrous MgSO4. It was concentrated and the crude product was used for further reactions. |
|
With sodium hydroxide In methanol; water |
2.2.1 Preparation of Na[Au(SC4H3N2)2] (1)
A suspension of 2-pyrSH (1.1432g, 10.189mmol) in a 5:2 mixture of water and methanol (14mL) was treated with an aqueous solution of NaOH (0.45g, 11.3mmol) to produce a clear, yellow solution of 2-pyrS-. A solution of NaAuCl4·2H2O (1.00g, 2.519mmol) in water (10mL) was slowly poured into the 2-pyrS- solution. A brown/black precipitate immediately formed, but slowly decomposed over 5 minutes, resulting in a white solid suspended in a pale-yellow solution. The suspension was extracted with chloroform (2×25mL), resulting in the removal of the white solid (C4H3N2SSC4H3N2). A mixture of the yellow aqueous layer and 1-butanol (50mL) was placed in a round-bottomed flask and the water layer was removed under rapid stirring and reduced pressure, resulting in the formation NaCl crystals. The yellow 1-butanol solution was removed from the NaCl crystals, which were washed with cold 1-butanol (2×2mL). The combined 1-butanol solutions were reduced in volume to 25mL, and the yellow solid that formed upon cooling was isolated by vacuum filtration (0.94g, 85%). The golden yellow crystals slowly darken and decompose starting at 190.0°C. Anal. Found: C, 21.9; H, 1.4; N, 12.65; S, 14.7. Calc. for NaAuC8H6N4S2: C, 21.7; H, 1.4; N, 12.7; S, 14.5. IR: νmax/cm-1 1559m, 1539s, 1369s ν(CC, CN); 1203 w, 1178s ν(CN, NCS, and CS). δH (D2O): 6.912 (1H, t, J=4.8Hz, Hpyr-5), 8.185 (2H, d, J=5.2Hz, Hpyr-4, pyr-6). MS (ESI) m/z 464.9 ({Na2[Au(SC4H3N2)2]+, 100%). |
|
With core-shell Pd/Cu nanomaterial loaded on MoS2 nanoflowers In water; acetonitrile at 25℃; for 3h; Irradiation; |
2.4 Photocatalytic activity of PdCu/MoS2 for the oxidative coupling of thiols
General procedure: The reaction mixture consisted of thiol (0.1 mol), a catalyst based on Pd (0.5 mol%), and solvent (3 mL). Following the reaction, the catalyst was removed by centrifugation. Afterward, the disulfide product was obtained by adding ethyl acetate, separating the catalyst by centrifugation, dehydrating with MgSO4, and concentrating with a rotary evaporator. Then, the conversion of the product was measured by the GC-MS technique. Photocatalytic reactions were conducted under irradiation from a Xenon (Xe) lamp (300 W) with a 400-800 nm cut-off and an intensity of 0.1 W/cm2. The lamp is equipped with liquid filter (reduce heat), IR cut-off (block wavelength >800 nm), and specific long pass filter (cut-on 400, 455, 515, 610 nm) to block the transmission below specific wavelength. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping