Alternatived Products of [ 14852-31-4 ]
Product Details of [ 14852-31-4 ]
| CAS No. : | 14852-31-4 |
MDL No. : | MFCD00004553 |
| Formula : |
C16H34O
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | - |
| M.W : |
242.44
|
Pubchem ID : | - |
| Synonyms : |
|
Safety of [ 14852-31-4 ]
| Signal Word: | |
Class: | |
| Precautionary Statements: | |
UN#: | |
| Hazard Statements: | |
Packing Group: | |
Application In Synthesis of [ 14852-31-4 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Upstream synthesis route of [ 14852-31-4 ]
- Downstream synthetic route of [ 14852-31-4 ]
- 1
-
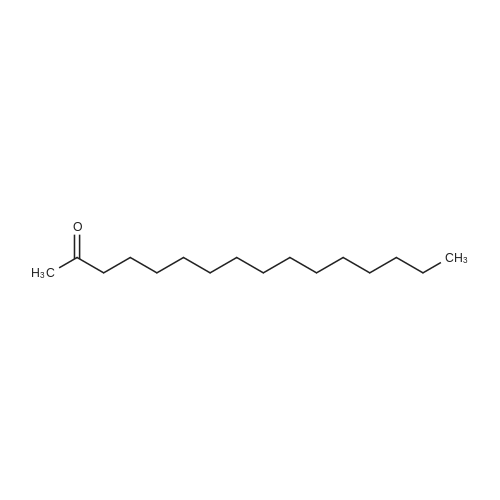 [ 18787-63-8 ]
[ 18787-63-8 ]

-
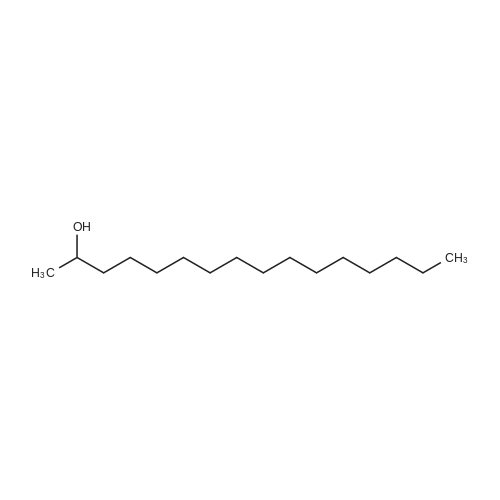 [ 14852-31-4 ]
[ 14852-31-4 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With cyclohexane; cobalt catalyst at 200℃; Hydrogenation; |
|
|
With sodium tetrahydroborate |
|
|
With lithium tri-t-butoxyaluminum hydride In tetrahydrofuran no long-chain inhibition compared with 2-hexanone; |
|
Reference:
[1]Asinger; Eckoldt
[Chemische Berichte, 1943, vol. 76, p. 591]
[2]Jones,D.F.; Howe,R.
[Journal of the Chemical Society C: Organic, 1968, p. 2801 - 2808]
[3]Menger; Nelson Jr., Kingsley H.
[Tetrahedron Letters, 1994, vol. 35, # 9, p. 1347 - 1348]
- 2
-
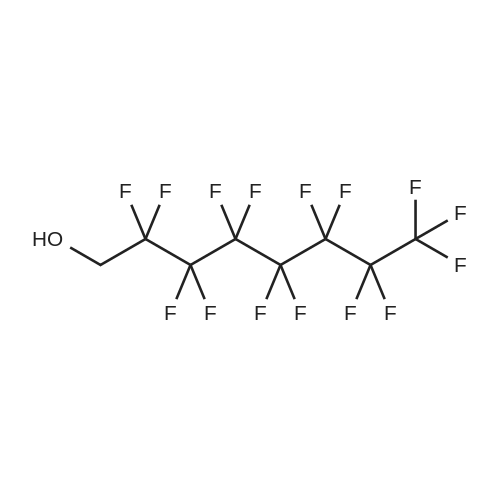 [ 307-30-2 ]
[ 307-30-2 ]

-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
2-(2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-Pentadecafluoro-octyloxy)-hexadecane
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 93% |
With tributylphosphine; 1,1'-azodicarbonyl-dipiperidine In benzene at 65℃; for 6h; |
|
- 3
-
 [ 18787-63-8 ]
[ 18787-63-8 ]

-
 [ 14852-31-4 ]
[ 14852-31-4 ]
| Yield | Reaction Conditions | Operation in experiment |
|
at 200℃; Hydrogenation; |
|
- 4
-
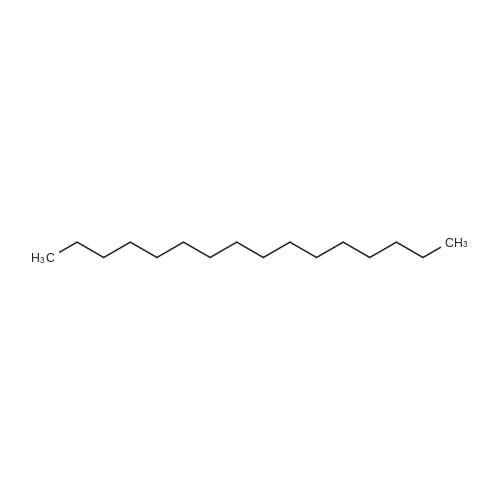 [ 544-76-3 ]
[ 544-76-3 ]

-
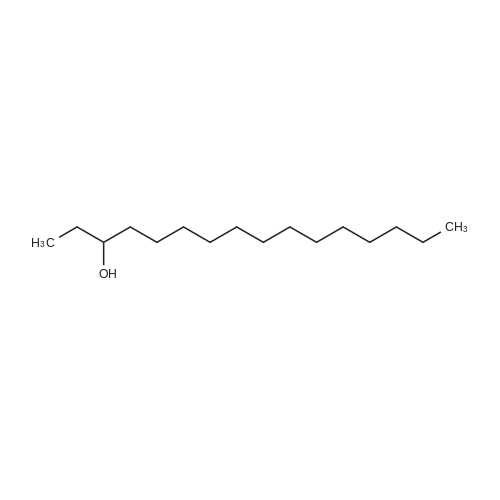 [ 593-03-3 ]
[ 593-03-3 ]

-
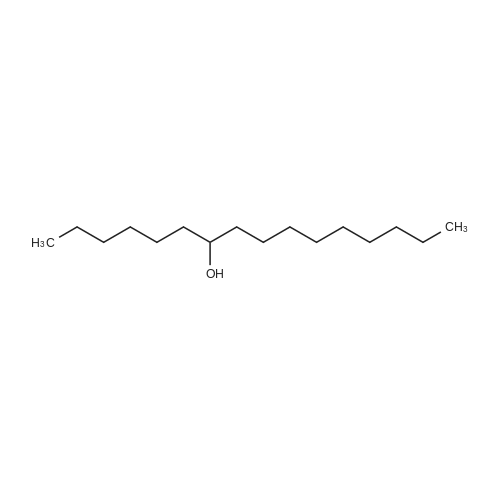 [ 591-72-0 ]
[ 591-72-0 ]

-
 [ 21078-87-5 ]
[ 21078-87-5 ]

-
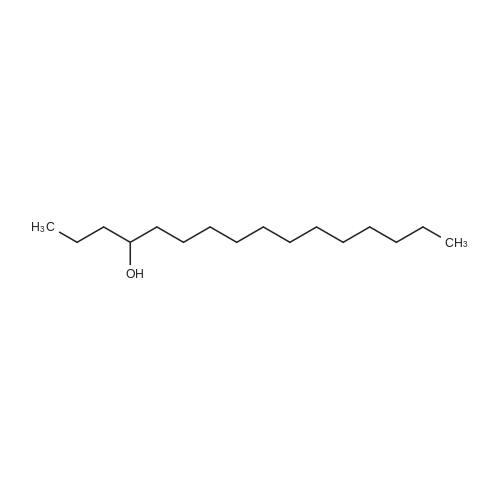 [ 19781-43-2 ]
[ 19781-43-2 ]

-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
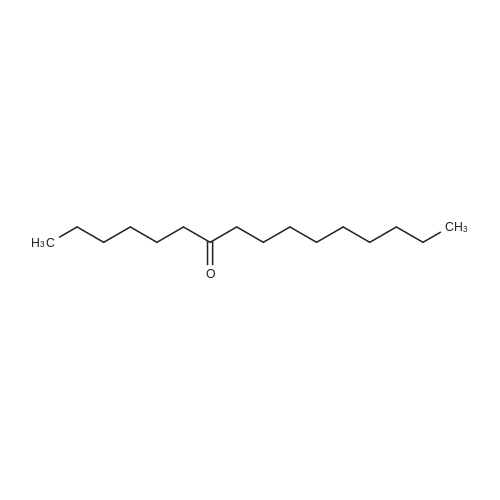 [ 45206-91-5 ]
[ 45206-91-5 ]

-
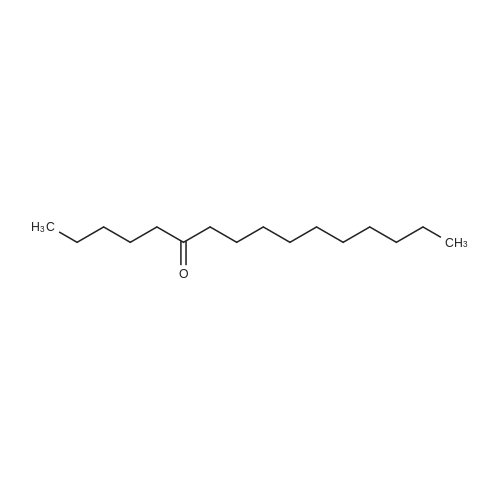 [ 57661-23-1 ]
[ 57661-23-1 ]

-
 [ 18787-64-9 ]
[ 18787-64-9 ]

-
 [ 18787-63-8 ]
[ 18787-63-8 ]

-
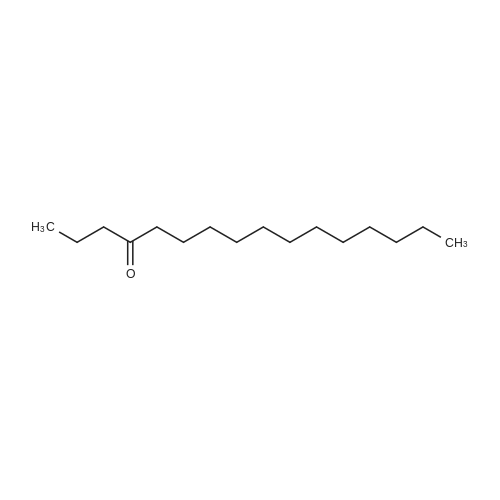 [ 18787-65-0 ]
[ 18787-65-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With 2percent Au/2percent TESPTS in mesoporous silica at 150℃; for 6h; Air; |
|
- 5
-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
 [ 18787-63-8 ]
[ 18787-63-8 ]
| Yield | Reaction Conditions | Operation in experiment |
| 70% |
With pyridine; tert.-butylhydroperoxide; [Fe[(S)-N-benzyl-2-phenyl-2-(pyridin-2-ylmethoxy)-N-(pyridin-2-ylmethyl)ethanamine](OTf)2] In acetone at 20℃; for 8h; Inert atmosphere; Green chemistry; |
7.4.2. Determination of isolated yields in acetone, typical procedure for 9H-fluoren-9-one
General procedure: The substrate 9H-fluorene (0.100 g, 0.601 mmol) and the catalyst [Fe(12)(OTf)2] (0.013 g, 0.018 mmol) were dissolved in acetone (2.0 mL). The oxidant tBuOOH (0.344 mL, 70 wt.% in H2O, 2.4 mmol) was added dropwise and the dark solution was shaken for 8 h at room temperature. The solvent was removed under vacuum. The product 9H-fluoren-9-one was isolated by column chromatography as an colorless oil (0.071 g, 0.394 mmol, 65%) using silica gel and CHCl3 as eluent. Analytical data for all oxidation products in Table 1 are given in the Supporting information |
| 98 %Chromat. |
With 5H3N*5H(1+)*IMo6O24(5-); oxygen; sodium acetate In water; acetonitrile at 70℃; for 20h; Green chemistry; |
|
Reference:
[1]Lenze, Matthew; Sedinkin, Sergey L.; Bauer, Eike B.
[Journal of Molecular Catalysis A: Chemical, 2013, vol. 373, p. 161 - 171]
[2]Zhang, Mengqi; Zhai, Yongyan; Ru, Shi; Zang, Dejin; Han, Sheng; Yu, Han; Wei, Yongge
[Chemical Communications, 2018, vol. 54, # 72, p. 10164 - 10167]
- 6
-
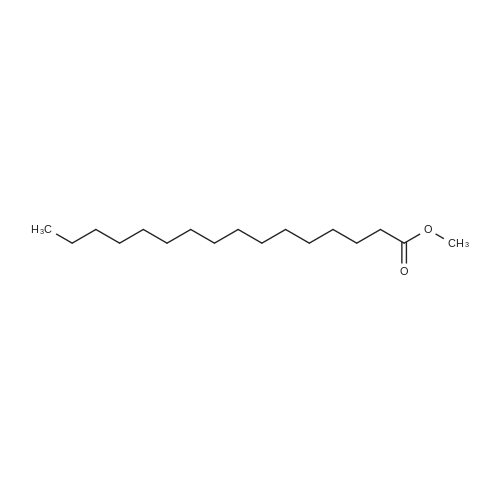 [ 112-39-0 ]
[ 112-39-0 ]

-
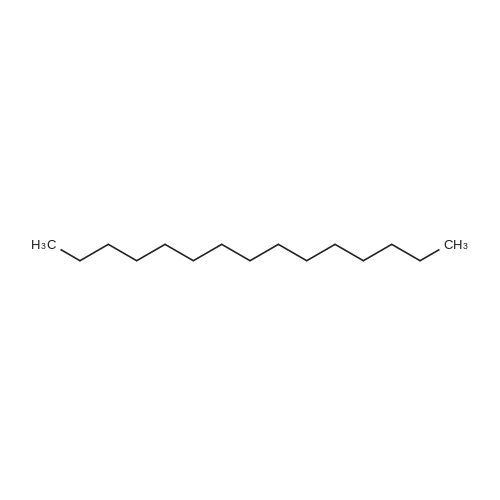 [ 629-62-9 ]
[ 629-62-9 ]

-
 [ 34557-54-5 ]
[ 34557-54-5 ]

-
 [ 544-76-3 ]
[ 544-76-3 ]

-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
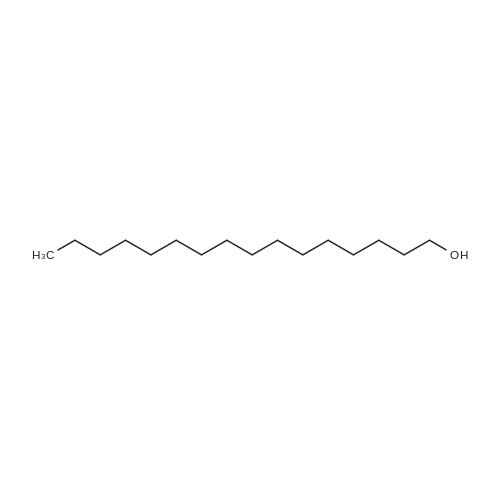 [ 36653-82-4 ]
[ 36653-82-4 ]

-
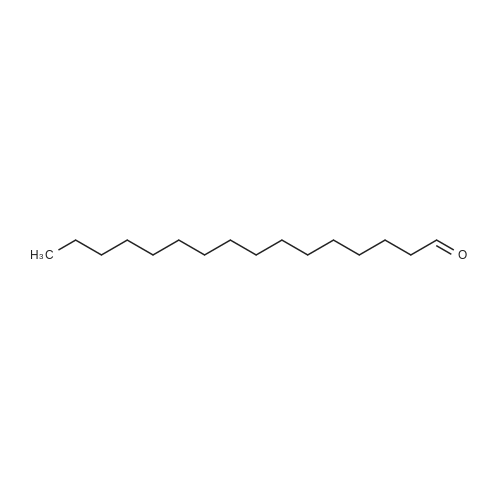 [ 629-80-1 ]
[ 629-80-1 ]

-
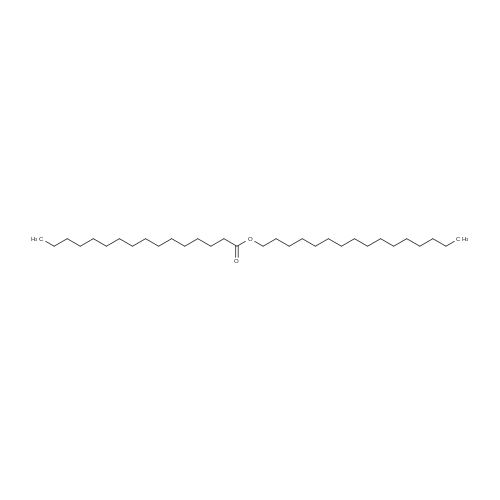 [ 540-10-3 ]
[ 540-10-3 ]

-
 [ 124-38-9 ]
[ 124-38-9 ]

-
 [ 201230-82-2 ]
[ 201230-82-2 ]

-
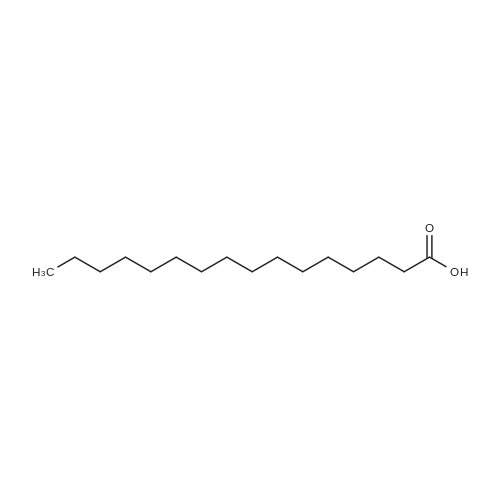 [ 57-10-3 ]
[ 57-10-3 ]
- 7
-
 [ 112-39-0 ]
[ 112-39-0 ]

-
 [ 629-62-9 ]
[ 629-62-9 ]

-
 [ 34557-54-5 ]
[ 34557-54-5 ]

-
 [ 544-76-3 ]
[ 544-76-3 ]

-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
 [ 36653-82-4 ]
[ 36653-82-4 ]

-
 [ 629-80-1 ]
[ 629-80-1 ]

-
 [ 540-10-3 ]
[ 540-10-3 ]

-
 [ 201230-82-2 ]
[ 201230-82-2 ]

-
 [ 57-10-3 ]
[ 57-10-3 ]
- 8
-
 [ 14852-31-4 ]
[ 14852-31-4 ]

-
 [ 629-62-9 ]
[ 629-62-9 ]

-
 [ 629-59-4 ]
[ 629-59-4 ]

-
 [ 544-76-3 ]
[ 544-76-3 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With hydrogen In n-heptane at 199.84℃; Autoclave; |
|
Reference:
[1]Di, Lu; Yao, Sikai; Li, Mengru; Wu, Guangjun; Dai, Weili; Wang, Guichang; Li, Landong; Guan, Naijia
[ACS Catalysis, 2015, vol. 5, # 12, p. 7199 - 7207]
Categories

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping




















