|
|
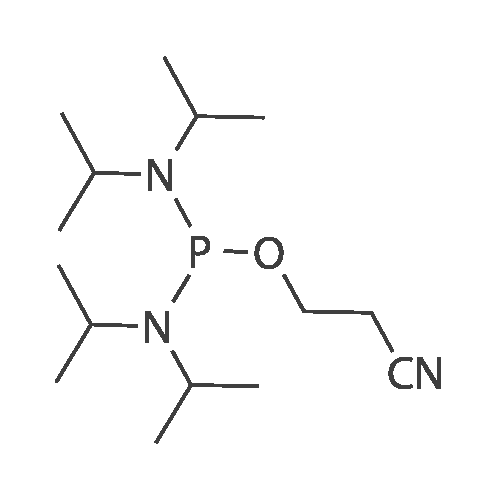
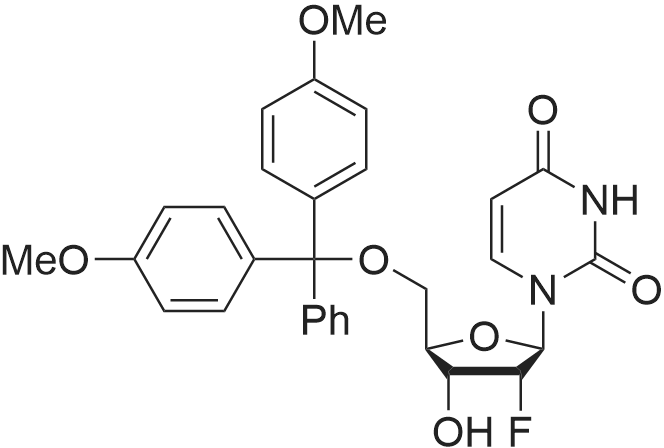
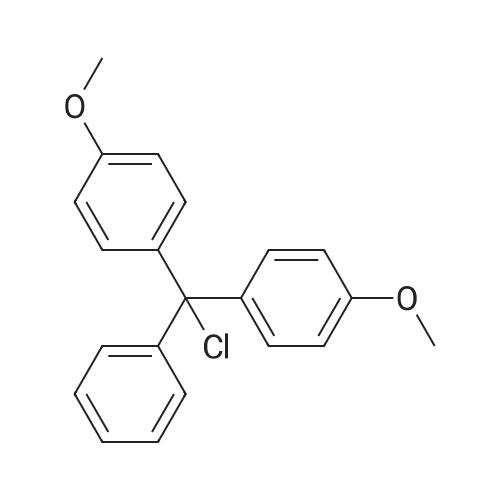
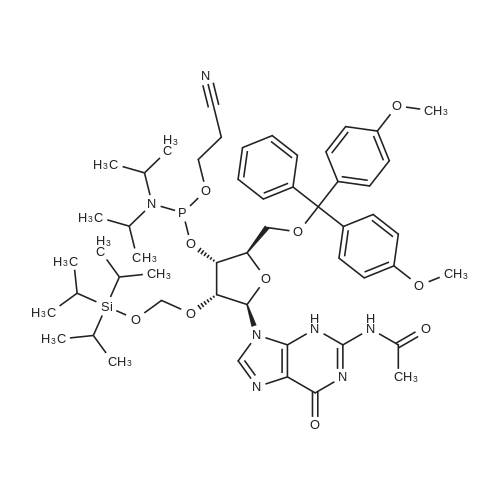
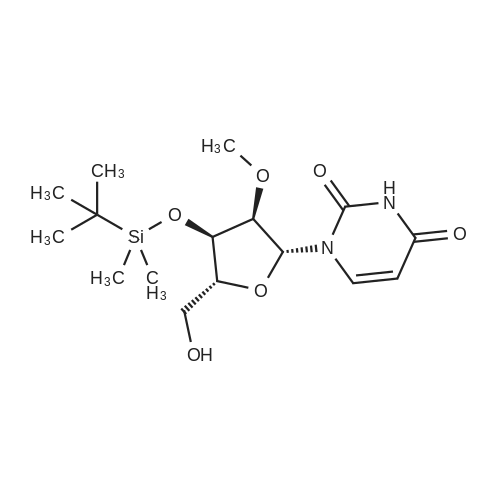
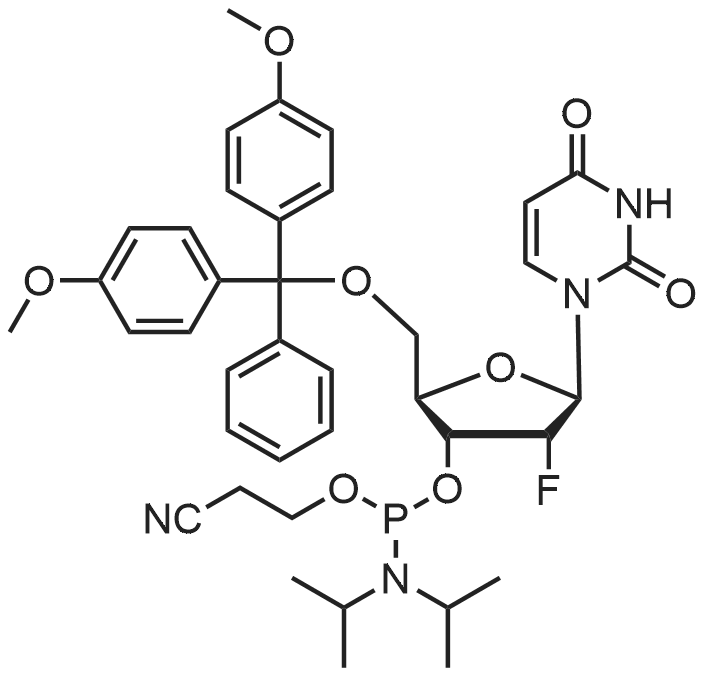
Compound 17: Compound 15 (11.01 g, 14.7 mmol) and compound 16 (5.74 g, 15.4 mmol, 1.05 eq.) were dried under high vacuum and dissolved in 100 ml dichloromethane (dry). To the solution was added 117 ml ETT (3.83 g, 29.4 mmol, 2 eq.), and the reaction mixture was stirred for 2.5 hours. PADS (6.35 g, 22.05 mmol, 1.5 eq.) and 2.52 ml 2.6-lutidine (2.36 g, 22.05 mmol, 1.5 eq.) were added, and the reaction mixture was stirred overnight. The reaction mixture was diluted with 800 ml dichloromethane and washed with 0 (300 ml) twice. The organic layer was dried over anhydrous Na2SO i and concentrated. The diastereomers were separated over CombiFlash column (330 g column, EtOAc: n-hexane (4: 1)) to yield 17a (top spot: 7.25 g 47%) and 17b (lower spot: 7.29 g, 47%)). In EtOAc: n- hexane (4: 1), 17a Rf = 0.23 and 17b Rf = 0.18. (0825) 17a NMR (400 MHz, DMS0 ) d 11.43 (s, 1H), 11.33 (s, 1H), 7.77 (d, J= 8.0 Hz, 1H), 7.57 (d, J= 8.1 Hz, 1H), 7.42 - 7.36 (m, 2H), 7.33 - 7.20 (m, 7H), 6.91 - 6.84 (m, 4H), 5.92 (d, J= 21.4 Hz, 1H), 5.81 (d, = 4.5 Hz, 1H), 5.63 (dd, .7= 8.1, 1.8 Hz, 1H), 5.60 - 5.42 (m, 2H), 5.36 - 5.24 (m, 1H), 4.34 - 4.21 (m, 3H), 4.21 - 4.06 (m, 2H), 4.03 - 3.93 (m, 2H), 3.86 (t, J= 4.8 Hz, 1H), 3.73 (s, 6H), 3.38 (d , J= 9.5 Hz, 1H), 3.34 (s, 3H), 3.29 (dd, J= 11.0, 4.8 Hz, 1H), 2.83 - 2.73 (m, 2H), 0.87 (s, 9H), 0.08 (s, 5H). 13C NMR (126 MHz, DMSO) d (0826) 163.11, 162.92, 158.13, 158.11, 150.31, 150.02, 144.31, 142.07, 140.13, 135.08, 135.03, 129.77, 129.72, 127.81, 127.76, 126.78, 120.26, 117.70, 113.15, 102.12, 101.73, 91.63, (0827) 90.05, 90.03, 89.75, 87.12, 85.97, 81.69, 81.62, 81.24, 79.61, 79.54, 73.12, 73.01, 69.64, 67.08, 67.06, 63.26, 63.23, 61.58, 57.61, 55.00, 25.54, 18.77, 18.70, 17.68, -4.88, -5.14. 31P NMR (202 MHz, DMSO) d 68.19. 19F NMR (376 MHz, DMSO) d -203.64, -203.70, -203.75, -203.78, -203.83, -203.89. Molecular weight for C49H59FN5O14PSS1 (M+Na): calculated 1074.368, found 1074.3170. (0828) 17b NMR (400 MHz, DMSO-ufe) d 11.51 - 11.46 (m, 1H), 11.43 - 11.38 (m, 1H), 7.80 (d, J= 8.1 Hz, 1H), 7.56 (d, j= 8.2 Hz, 1H), 7.38 (d, J= 7.5 Hz, 2H), 7.31 - 7.20 (m, 7H), 6.85 (dd, J= 8.9, 2.1 Hz, 4H), 5.91 (d, j= 21.8 Hz, 1H), 5.78 (d, J= 4.0 Hz, 1H), 5.63 - 5.46 (m, 2H), 5.41 (dd, J= 8.0, 2.0 Hz, 1H), 5.38 - 5.25 (m, 1H), 4.27 - 4.15 (m, 4H), 4.15 - 4.06 (m, 2H), 3.96 - 3.90 (m, 1H), 3.86 - 3.81 (m, 1H), 3.72 (s, 6H), 3 41 - 3.36 (m, 1H), 3.35 (s, 3H), 3.29 - 3.24 (m, 1H), 2.92 (t, j= 5.8 Hz, 2H), 0.85 (s, 9H), 0.07 (s, 3H), 0.06 (s, 3H). 13C NMR (126 MHz, DMSO) d 163.12, 162.94, 158.11, 156.61, 150.25, 150.00, 144.34, 142.06, 140.09, 137.28, 135.07, 135.00, 129.75, 129.73, 127.77, 127.75, 126.72, 120.32, 117.92, (0829) 113.11, 101.99, 101.69, 91.76, 90.27, 90.06, 89.78, 87.33, 85.98, 81.43, 81.40, 81.37, 79.48, 79.41, 72.88, 72.74, 69.40, 66.71, 63.59, 63.55, 61.33, 57.62, 54.95, 25.54, 18.84, 18.77, 17.67, -4.86, -5.13. 31P NMR (202 MHz, DMSO) d 68.34. 19F NMR (376 MHz, DMSO) d - 203.36, -203.42, -203.47, -203.50, -203.56, -203.61. Molecular weight for (0830) C49H59FN5Oi4PSSi (M+Na): calculated 1074.368, found 1074.3198. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping