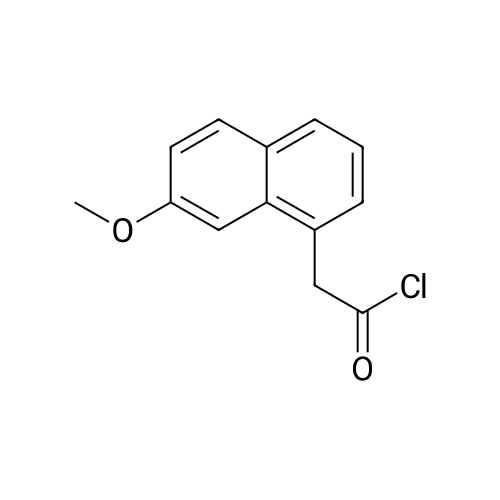
| 93% |
With oxygen; 2,3-dicyano-5,6-dichloro-p-benzoquinone; In dichloromethane; at 20℃; under 15.0015 Torr; for 5h; |
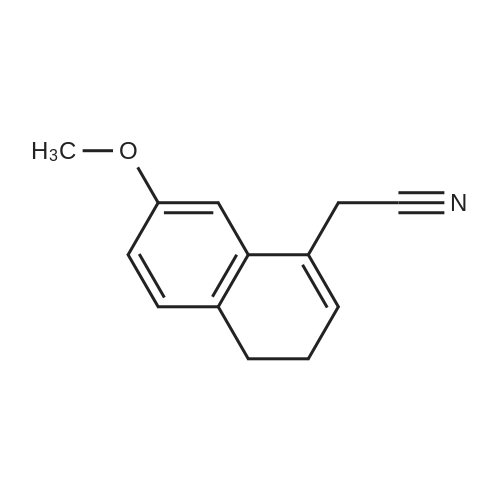
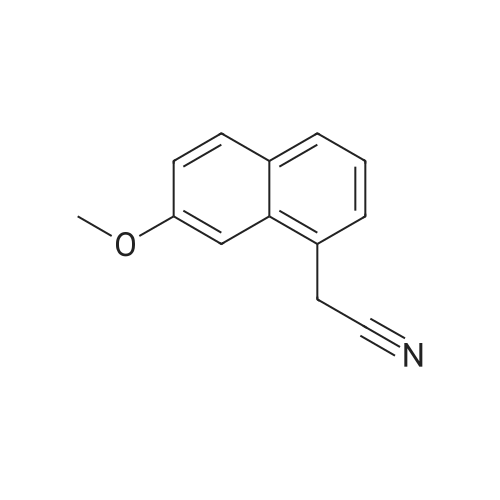
A method for synthesizing 7-methoxynaphthalene acetonitrile as shown in FIG. 2. In a 500 ml pressure cooker was added 150 ml of methylene chloride,19.9 g (0.1 mol) of 7-methoxy-3,4-dihydronaphthaleneacetonitrile,1.15 grams DDQ (0.005 mole),Into the 2kPa pressure of oxygen,The reaction was stirred at room temperature for about 5 hours,The reaction did not further absorb oxygen, the reaction was stopped,The reaction solution was poured into 100 ml of saturated sodium bicarbonate solution, the organic layer was washed with 100 ml of water,The organic layer was concentrated and the residue was recrystallized from 50 ml of ethanol-water = 4: 1,The product 7-methoxy-naphthalene acetonitrile 18.3 g, purity 98.5%, yield 93%. |
| 91% |
With allyl methacrylate;5%-palladium/activated carbon; In toluene;Heating / reflux; |
There are introduced into a 670 litre reactor 12.6 kg of 5% palladium-on-carbon in toluene, which is heated at reflux; then 96.1 kg of (7-methoxy-3,4-dihydro-1-naphthalenyl)-acetonitrile dissolved in toluene are added as well as 63.7 kg of allyl methacrylate. The reaction is continued at reflux and is followed by vapour phase chromatography. When all the starting substrate has disappeared, the reaction mixture is cooled to ambient temperature and then filtered. After removal of the toluene by evaporation, the resulting solid residue is recrystallized from an ethanol/water (80/20) mixture to give the title product in a yield of 91% and with a chemical purity exceeding 99%. Melting point: 83 C. |
| 91% |
With allyl methacrylate;palladium on activated charcoal; In toluene;Heating / reflux; |
There are introduced into a 670 litre reactor 12.6 kg of 5% palladium-on-carbon in toluene, which is heated at reflux; then 96.1 kg of (7-methoxy-3,4-dihydro-1-naphthalenyl)-acetonitrile dissolved in toluene are added as well as 63.7 kg of allyl methacrylate. The reaction is continued at reflux and is followed by vapour phase chromatography. When all the starting substrate has disappeared, the reaction mixture is cooled to ambient temperature and then filtered. After removal of the toluene by evaporation, the resulting solid residue is recrystallized from an ethanol/water (80/20) mixture to give the title product in a yield of 91% and with a chemical purity exceeding 99%. Melting point: 83 C. |
| 91% |
With allyl methacrylate;5%-palladium/activated carbon; In toluene;Heating / reflux; |
There are introduced into a 670 litre reactor 12.6 kg of 5% palladium-on-carbon in toluene, which is heated at reflux; then 96.1 kg of (7-methoxy-3,4-dihydro-1-naphthalenyl)-acetonitrile dissolved in toluene are added as well as 63.7 kg of allyl methacrylate. The reaction is continued at reflux and is followed by vapour phase chromatography. When all the starting substrate has disappeared, the reaction mixture is cooled to ambient temperature and then filtered. After removal of the toluene by evaporation, the resulting solid residue is recrystallized from an ethanol/water (80/20) mixture to give the title product in a yield of 91% and with a chemical purity exceeding 99%. Melting point: 83 C. |
| 91% |
With allyl methacrylate;5% Pd(II)/C(eggshell); In toluene;Reflux; Industry scale; |
Step B: (7-Methoxy-1-naphthyl)acetonitrile There are introduced into a 670 litre reactor 12.6 kg of 5% palladium-on-carbon in toluene, which is heated at reflux; then 96.1 kg of (7-methoxy-3,4-dihydro-1-naphthyl)acetonitrile dissolved in toluene are added and also 63.7 kg of allyl methacrylate. The reaction is continued at reflux and is monitored by vapour phase chromatography. When all the starting substrate has disappeared, the reaction mixture is cooled to ambient temperature and then filtered. After evaporating off the toluene, the solid residue obtained is recrystallized from an ethanol/water (80/20) mixture to yield the title product in a yield of 91% and with a chemical purity of more than 99%. Melting point: 83 C. |
| 91% |
With 2,3-dicyano-5,6-dichloro-p-benzoquinone; for 2h;Reflux;Product distribution / selectivity; |
Example-3: 5 g. (0.02 mol.) of (7-Methoxy-3,4-dihydro-l-naphthyl)acetonitrile, 7.5 g. (0.03 mol.) of 2,3-Dichloro-5,6-dicyano-benzoquinone (DDQ) and 200 ml. of O-xylene or toluene were charged in a clean and dry 4 neck R.B. Flask and heated to reflux temperature for about 2 hrs. The resultant reaction mixture was cooled to about 30C and filtered. The filtrate was washed with 3x25 ml. of 10% w/v sodium hydroxide solution and 2x25 ml. of water. The solvent was distilled completely under vacuum and the resulted solid residue was recrystallized from ethanol/ and water mixture in a ratio of 8:2 to yield 4.5 gr. (91%) of the title compound with a chemical purity 99%. |
| 65.8% |
With 2,3-dicyano-5,6-dichloro-p-benzoquinone; In dichloromethane; at 25℃; |
2-(7-methoxy-3,4-dihydronaphth-1-yl)acetonitrile(5.44g, 27.30mmol) and DDQ (7.44g, 32.76mmol) in dichloromethane (30 mL) at 25 in The reaction overnight, diluted with 60mL of ethyl acetate, filtered and the filtrate washed with saturated sodium bicarbonate solution (40 mL) and saturated brine (40 mL), dried the organic phase after separation was dried over anhydrous sodium sulfate. Filtered, the filtrate was spin dry, purified by column chromatography (petroleum ether / ethyl acetate (v / v) = 60/1) to give the title compound as a yellow solid (3.54g, 65.8%). |
|
With sulfur; at 175℃; for 0.166667h;Product distribution / selectivity; |
Example 21: Preparation of 2-(7-methoxy-l-naphthyl)acetonitrile (IVa)IV IVa(i) Taken 60gr. of (7-methoxy-3,4-dihydro-l-naphthyl)acetonitrile (IV) in a Hit. RBF. Added 24gr. of sulphur. Then heated to about 175C and stirred for lOmin. at the same temperature. Then cooled to 50-60C.Added 430ml. of 20% aq.ethanol solution and heated to 70-75C.6gr. of carbon treatment was given to the solution. Taken the filterate, cooled to 5- 10C and maintained for 30min. at the same temperature. Filtered the solid and washed with 30ml. of chilled 20%aq.ethanol and dried to furnish 20gr. of the title compound. |
|
With 2,3-dicyano-5,6-dichloro-p-benzoquinone; In toluene; at 10 - 20℃; for 1h; |
Example 1: Preparation of (7-methoxy-l-naphthyl)acetonitrile of Formula (C) 7-methoxy- l-tetralone of Formula (A) (200 g), toluene (1000 ml), cyanoacetic acid ( 140 g), benzyl amine (30 g) and formic acid ( 13 g) were added at 25C to 35C and stirred for 20 minutes. The reaction mass was heated to 1 10C to 120C and maintained for 30 to 36 hours. The reaction mass was treated with Sodium hydroxide solution (500 ml) and organic layer (7-Methoxy-3,4-dihydro- l -naphthalenyl)acetonitrile of Formula (B) was taken for the next step. Toluene (1000 ml) and 2,3-Dichloro-5,6-dicyano 1 ,4- benzoquinone (240 g) was added at 25C to 35C. The reaction mass was cooled to 10C to 20C and separated organic layer of compound of Formula (B) was added and maintained for 1 hour. The reaction mass was washed with Toluene (200.0 mL) and treated with Sodium bicarbonate solution (500 ml). The organic layer was treated with charcoal (10 g) and finally treated with ethanol (300 ml) at 25-30C. The reaction mass was heated at 50C to 55C followed by addition of water (200 ml) and cooled to 25C to 30C. The reaction mass was filtered and washed with chilled ethanol (2x50ml) afforded (7-methoxy- 1 -naphthyl)acetonitrile of Formula (C). Yield - 76% |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping