Alternatived Products of [ 13511-11-0 ]
Product Details of [ 13511-11-0 ]
| CAS No. : | 13511-11-0 |
MDL No. : | MFCD23115649 |
| Formula : |
C20H19N
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | ZCAWQFNYHFHEPZ-UHFFFAOYSA-N |
| M.W : |
273.37
|
Pubchem ID : | 18326012 |
| Synonyms : |
|
Application In Synthesis of [ 13511-11-0 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 13511-11-0 ]
- 1
-
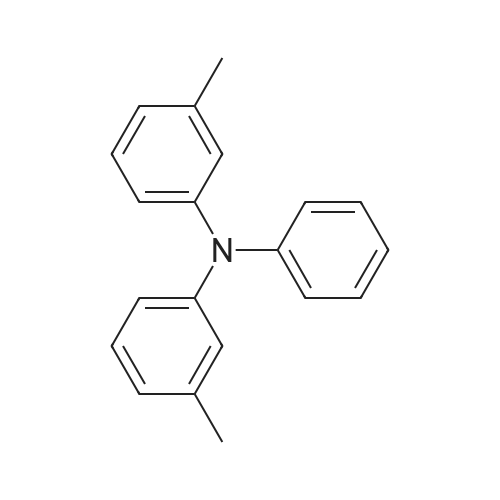 [ 13511-11-0 ]
[ 13511-11-0 ]

-
3,3'-dimethyltriphenylamine radical cation
[ No CAS ]

-
Phenyl-di-m-tolyl-amine; compound with phenyl-di-m-tolyl-amine
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
|
In 1,2-dichloro-ethane |
|
- 2
-
 [ 13511-11-0 ]
[ 13511-11-0 ]

-
C40H36N2
[ No CAS ]

-
C40H36N2
[ No CAS ]

-
N4,N4,N4',N4'-tetra-m-tolyl-[1,1'-biphenyl]-4,4'-diamine
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 13% |
Stage #1: 3-methyl-N-phenyl-N-(m-tolyl)aniline With methanesulfonic acid In dichloromethane at 0℃;
Stage #2: With chloranil In dichloromethane at 0 - 20℃; for 0.0166667h; |
|
- 3
-
 [ 1416276-98-6 ]
[ 1416276-98-6 ]

-
 [ 62-53-3 ]
[ 62-53-3 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 54% |
Stage #1: bis(3-methylphenyl)iodonium trifluoromethanesulfonate; aniline With copper(l) iodide; 2,6-di-tert-butyl-pyridine In toluene at 20℃; for 12h; Schlenk technique; Inert atmosphere;
Stage #2: With copper(l) iodide; 1,10-Phenanthroline; potassium <i>tert</i>-butylate In toluene at 120℃; for 24h; Schlenk technique; Inert atmosphere; |
|
- 4
-
 [ 108-41-8 ]
[ 108-41-8 ]

-
 [ 62-53-3 ]
[ 62-53-3 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 92% |
With bis-triphenylphosphine-palladium(II) chloride; (E)-2-(((2,5-bis(trifluoromethyl)phenyl)imino)methyl)phenol; sodium t-butanolate In o-xylene at 145℃; for 12h; Schlenk technique; Inert atmosphere; |
|
Reference:
[1]Tao, Xiaochun; Li, Lei; Zhou, Yu; Qian, Xuanying; Zhao, Min; Cai, Liangzhen; Xie, Xiaomin
[Chinese Journal of Chemistry, 2017, vol. 35, # 11, p. 1749 - 1754]
- 5
-
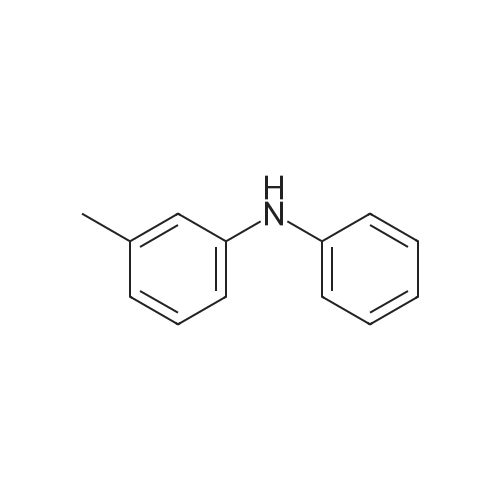 [ 1205-64-7 ]
[ 1205-64-7 ]

-
 [ 625-96-7 ]
[ 625-96-7 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 70% |
With palladium on activated charcoal; toluene-4-sulfonic acid In 1,3,5-trimethyl-benzene at 160℃; for 2h; Inert atmosphere; Schlenk technique; |
|
Reference:
[1]Jin, Xiongjie; Koizumi, Yu; Mizuno, Noritaka; Nozaki, Kyoko; Takayama, Satoshi; Yamaguchi, Kazuya; Yatabe, Takafumi
[Chemical Science, 2020, vol. 11, # 16, p. 4074 - 4084]
- 6
-
 [ 108-44-1 ]
[ 108-44-1 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 2 steps
1: palladium on activated charcoal; toluene-4-sulfonic acid / 1,3,5-trimethyl-benzene / 18 h / 160 °C / 760.05 Torr / Inert atmosphere; Schlenk technique
2: palladium on activated charcoal; toluene-4-sulfonic acid / 1,3,5-trimethyl-benzene / 2 h / 160 °C / 760.05 Torr / Inert atmosphere; Schlenk technique |
|
Reference:
[1]Jin, Xiongjie; Koizumi, Yu; Mizuno, Noritaka; Nozaki, Kyoko; Takayama, Satoshi; Yamaguchi, Kazuya; Yatabe, Takafumi
[Chemical Science, 2020, vol. 11, # 16, p. 4074 - 4084]
- 7
-
 [ 108-94-1 ]
[ 108-94-1 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
|
Multi-step reaction with 2 steps
1: palladium on activated charcoal; toluene-4-sulfonic acid / 1,3,5-trimethyl-benzene / 18 h / 160 °C / 760.05 Torr / Inert atmosphere; Schlenk technique
2: palladium on activated charcoal; toluene-4-sulfonic acid / 1,3,5-trimethyl-benzene / 2 h / 160 °C / 760.05 Torr / Inert atmosphere; Schlenk technique |
|
Reference:
[1]Jin, Xiongjie; Koizumi, Yu; Mizuno, Noritaka; Nozaki, Kyoko; Takayama, Satoshi; Yamaguchi, Kazuya; Yatabe, Takafumi
[Chemical Science, 2020, vol. 11, # 16, p. 4074 - 4084]
- 8
-
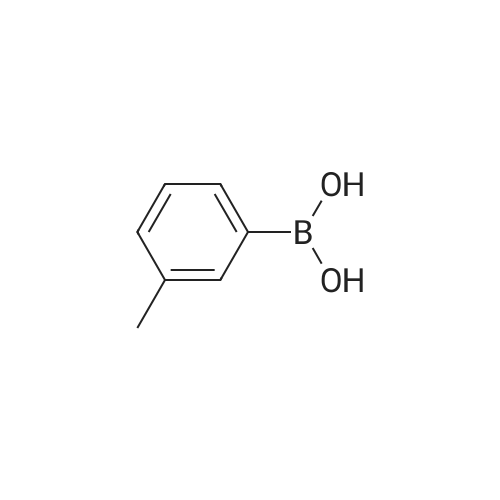 [ 17933-03-8 ]
[ 17933-03-8 ]

-
 [ 62-53-3 ]
[ 62-53-3 ]

-
 [ 1205-64-7 ]
[ 1205-64-7 ]

-
 [ 13511-11-0 ]
[ 13511-11-0 ]
| Yield | Reaction Conditions | Operation in experiment |
| 1: 83%
2: 17% |
With iron(III) trichloride hexahydrate; triphenylphosphine; sodium hydroxide In lithium hydroxide monohydrate; N,N-dimethyl-formamide at 20℃; for 0.75h; Sonication; Green chemistry; |
Method C:
General procedure: The ultrasonic probe was immersed directlyin the reactor. An ultrasonic generator (Sonics VC 505 300W) emitted sound vibration into the reaction mixture. Sonificationwas achieved at a low frequency of 20 kHz (50% amplitude) at room temperature for 45 min. The aryl halideor arylboronic acids (4.5 mmol), the aniline compound (2.25mmol), the ligand (0.05 mmol) and the catalyst FeCl3.6H2O,Ag, CuO or ZrO2) (0.025 mmol) were placed in a reactor. 3mL of solvent were added. After the reaction, the mixturewas extracted (three times) with diethyl ether. The latter wasdried on MgSO4 and the solvent removed under vacuum.The coupling product was finally isolated with silica gelchromatography. The reaction yields were determined withgas chromatography on a Shimadzu 2014-GC chromatograph. The capillary column was DB-5 and the carrier gaswas helium. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping







