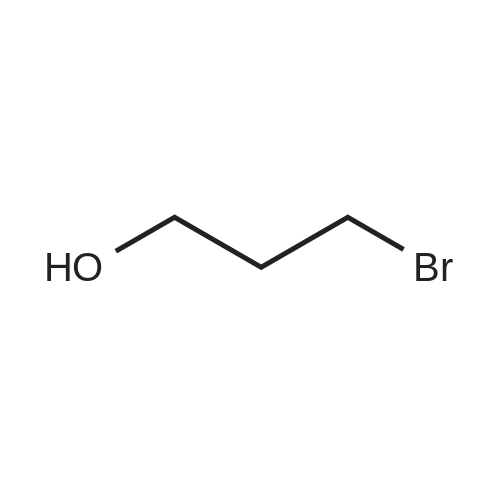
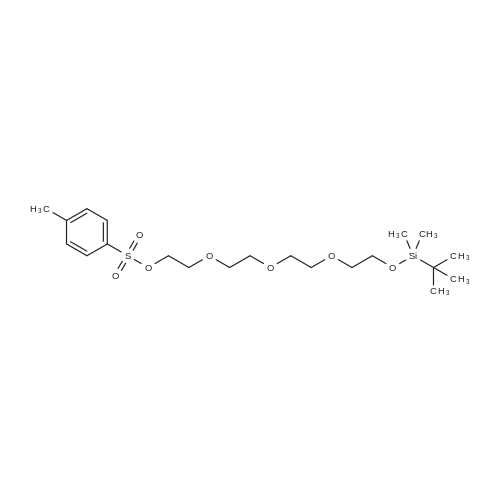
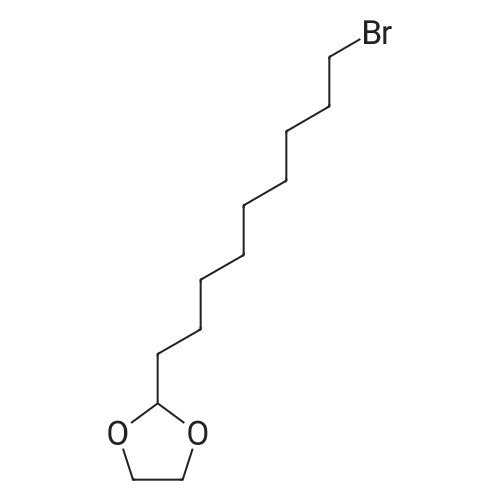
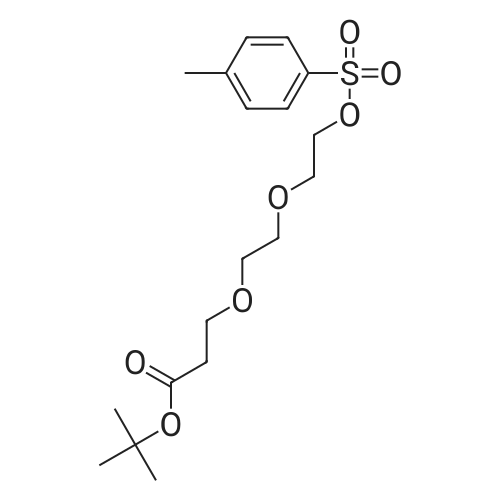
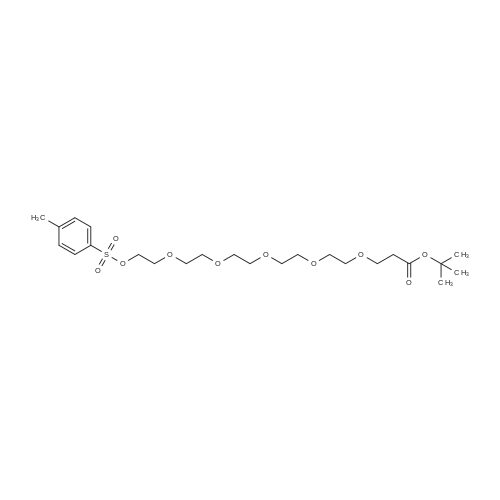
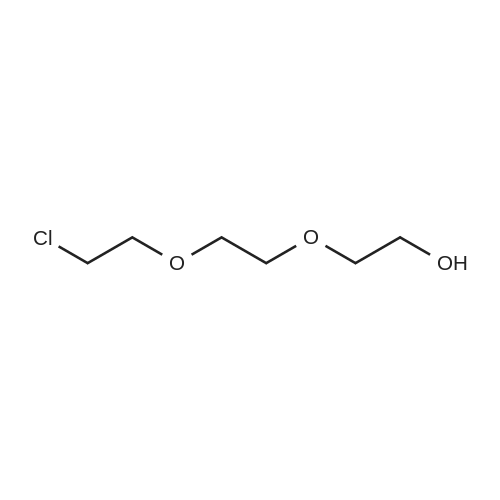
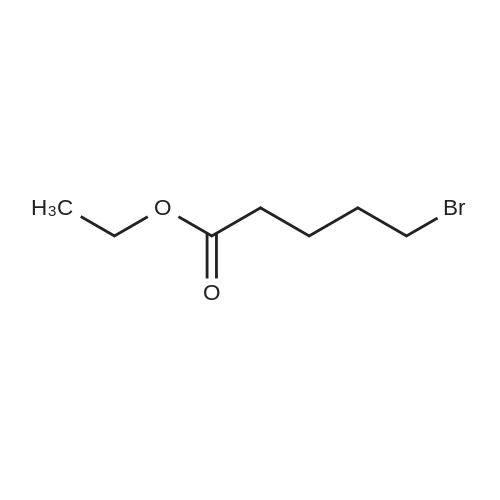
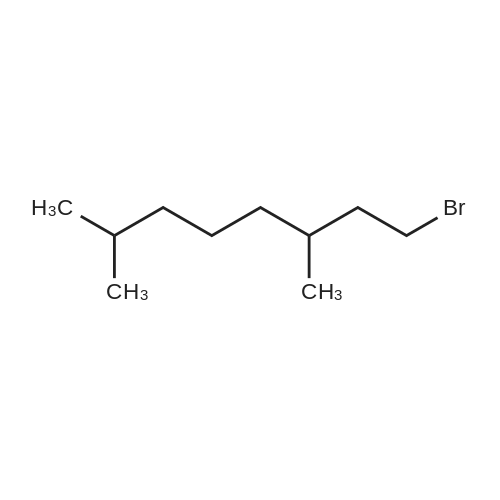
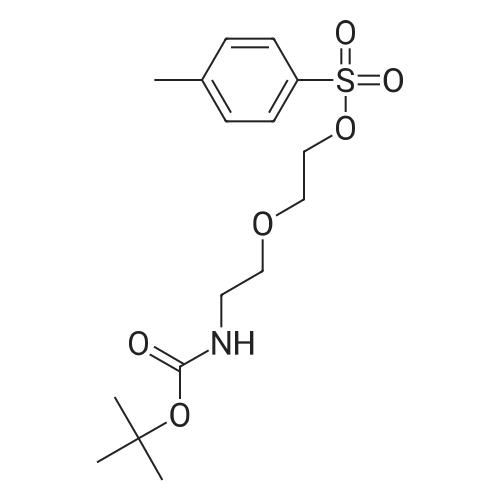
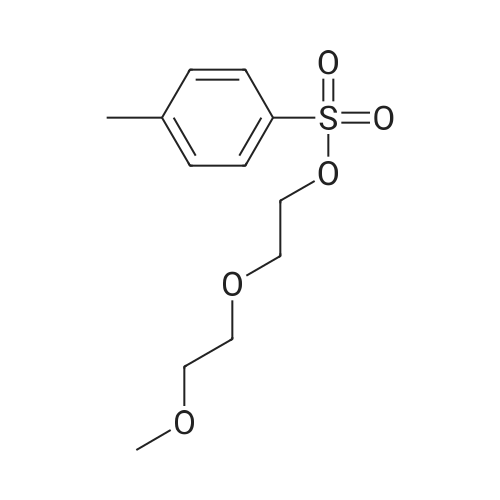
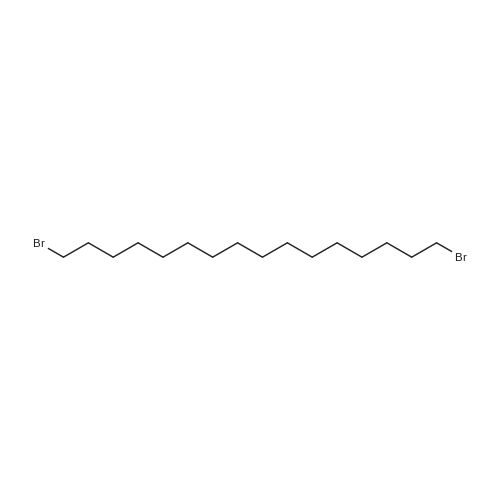
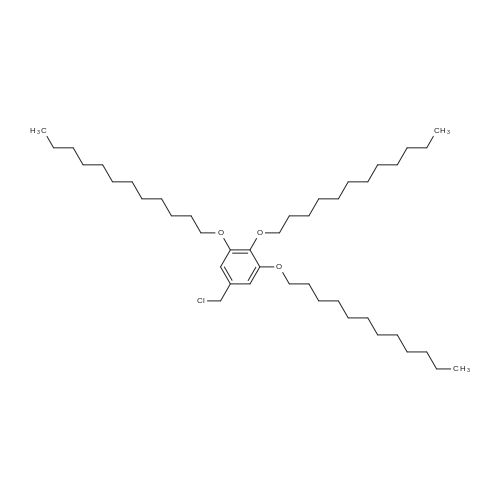
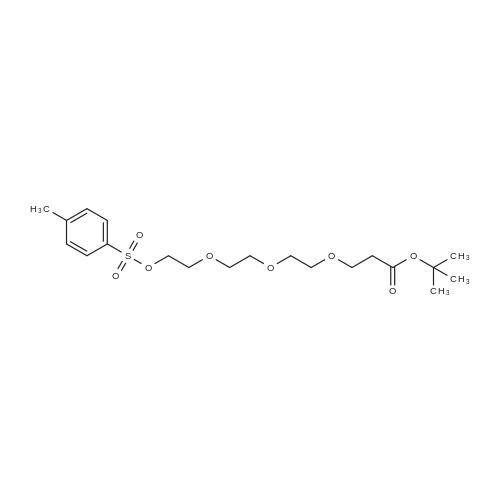
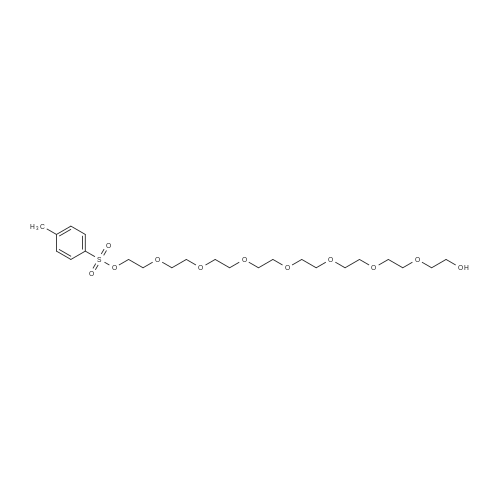
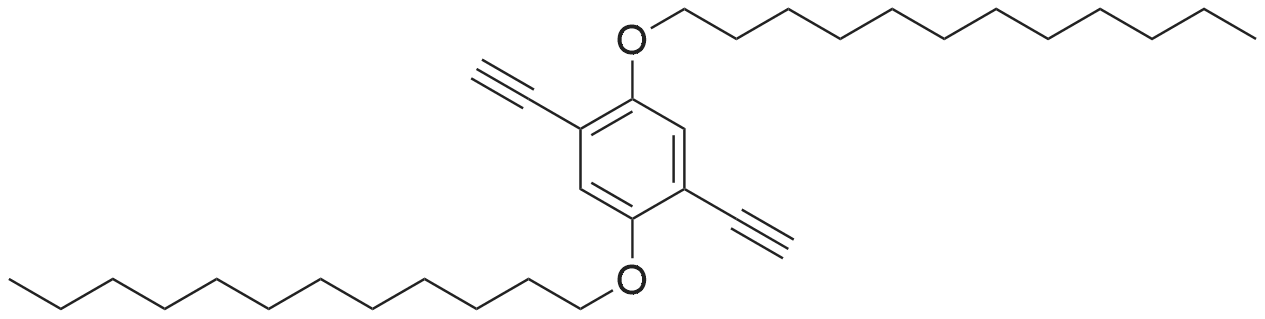
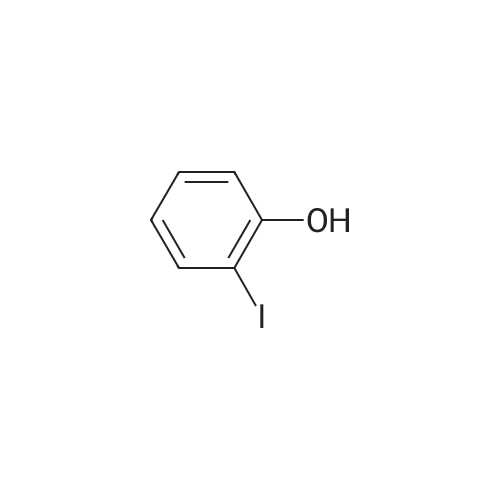
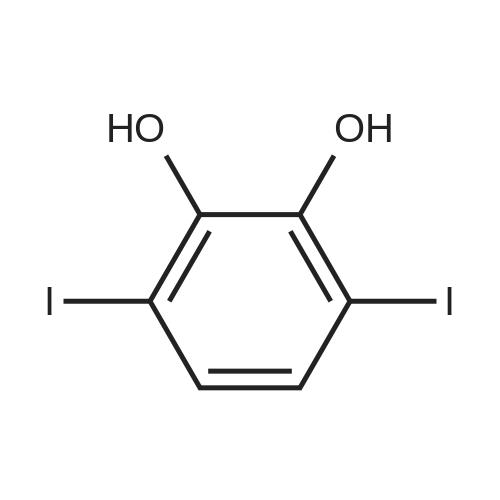
|
With boron tribromide; In dichloromethane; at -78 - 20℃; |
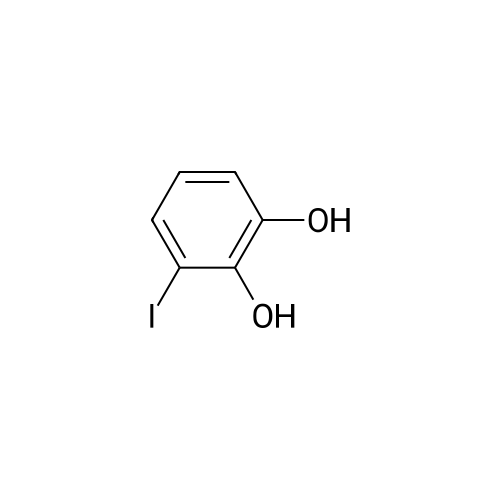
Compound b is dissolved in non-water, methylene chloride, and for cooling to -78 degrees, by adding dropwise boron tribromide (in pre-dissolved in methylene chloride), in the charging process keeping the temperature at -78 degrees. After adds the bromide boron, the room temperature gradually return to the reaction system, and continuing to stir, the pink reaction liquid. Under stirring conditions, the pink solution is slowly dropped into ice water, to produce a solid immediately, filtering and collecting the solid, to obtain gray solid, compound c is obtained after drying under vacuum. |
|
With boron tribromide; In dichloromethane; at -78 - 20℃; for 16h;Inert atmosphere; |
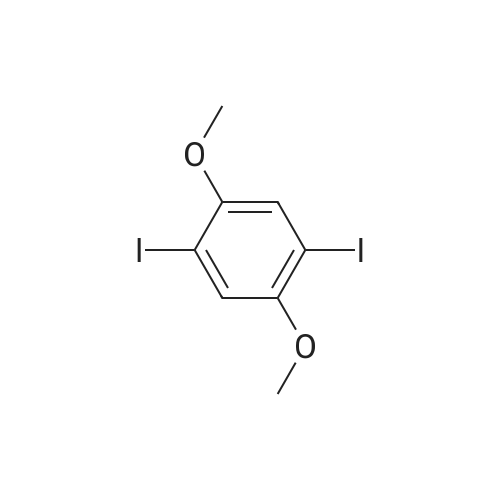
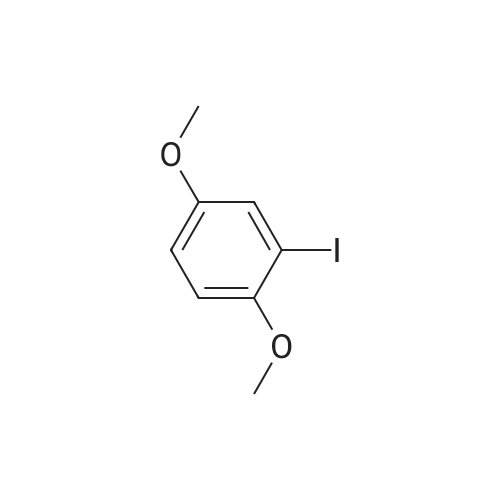
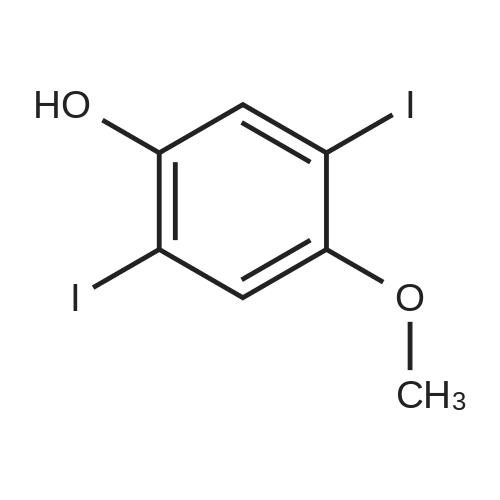
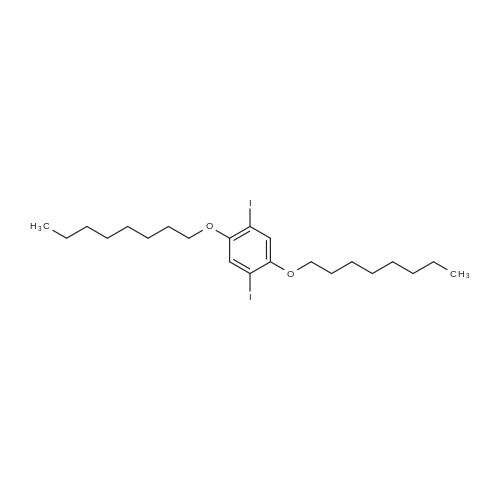
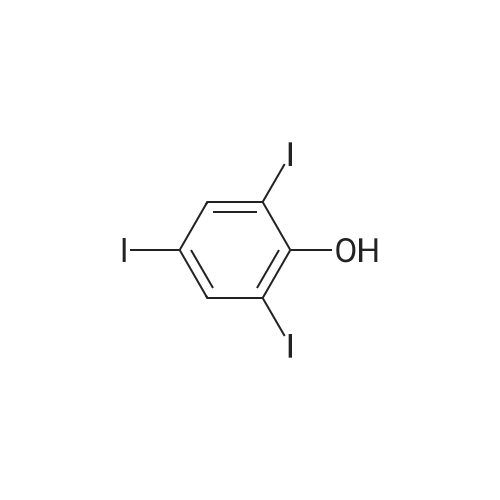
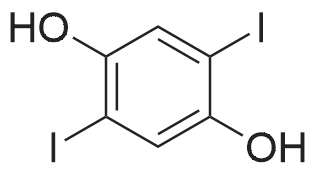
Under nitrogen, the 7.8g 2,5-diiodo-1,4-methoxybenzene was dissolved in 50mL of dichloromethane, and at -78 condition, to the solution was added dropwise 5.58mL three boron bromide. After completion of the dropwise addition, stirred for 16 hours at room temperature, the solid was suction filtered to produce a large number, and dried in vacuo to give a white solid which is 2,5-diiodo-1,4-bis(hydroxy)benzene. Among these, 2,5-diiodo-1,4-di(methoxybenzene, boron tribromide in methylenechloride and the molar ratio of 1: 3:40; |
|
With boron tribromide; In dichloromethane; at -78℃; for 16h;Inert atmosphere; |
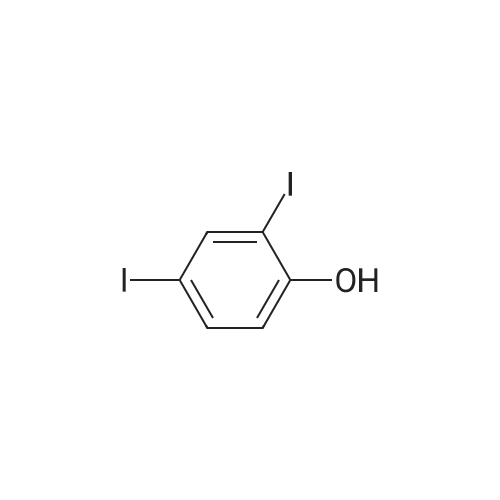
Under an argon atmosphere, 15.6 g of compound 1 was dissolved in 100 mL of dichloromethane,And 8.18 mL of boron tribromide was added dropwise to the solution at -78 C.Wherein the molar ratio of compound 1, boron tribromide and methylene chloride is 1: 2.2: 40. The reaction was stirred for 16 hours,After completion of the reaction, the reaction solution was transferred to an ice-water mixture and stirred, suction-filtered and dried in vacuo to give a white solid. |
|
With boron tribromide; In dichloromethane; for 96h;Cooling with acetone-dry ice; |
Compound 4-1 (10.06 g, 25.8 mmol) was dissolved in 100 mL of dry dichloromethane,The reaction flask was then cooled in a dry ice acetone bath,A solution of 1 mol / L boron tribromide in dichloromethane (56.8 mL, 56.8 mmol) was slowly added dropwise,Natural warmingreaction4 days.100 mL of ice water was allowed to cool in an ice-water bath, and then the reaction solution was gradually dropped into it with stirring,Produce a lot of solid, standing. Filtered with a Buchner funnel, the solid was washed three times with ice water, dried in vacuo,To give white solid compound 5-1 (8.71 g, 24.1 mmol)Yield: 93%; characterization: white solid; |
|
With boron tribromide; In dichloromethane; at -78 - 20℃; |
The compound was synthesized following the method described in Zhao and Schanze, Chem. Commun 2010, 46, 6075-6077, but using compound 6 as the amine. NMR (CD Cl 3, 300 MHz, 6ppm): 7.14 (s, 2H), 6.55 (br, s, 2H), 4.66 (br, s, 6H), 4.34 (s, 4H), 3.03 (m, 12H), 1.76 (m, 12H), 1.45 (m; 12H), 1.43 (s, 54H). 13C NMR (CDCh, 75 MHz, 5ppm): 166.0, 156.2, 151.7 122.7, 86.5, 79.4, 68.7, 58.9, 40.9, 32.9, 28.7, 24.2. HRMS: (ESI+) m/z calcd for CeoHiiuHNsOie [(M + H)]+ 1447.5732, found 1447.5706. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping