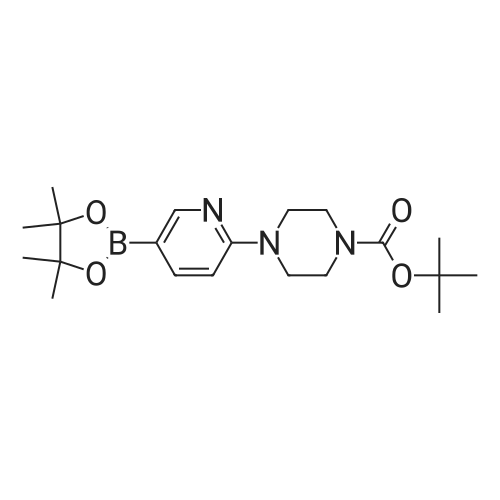
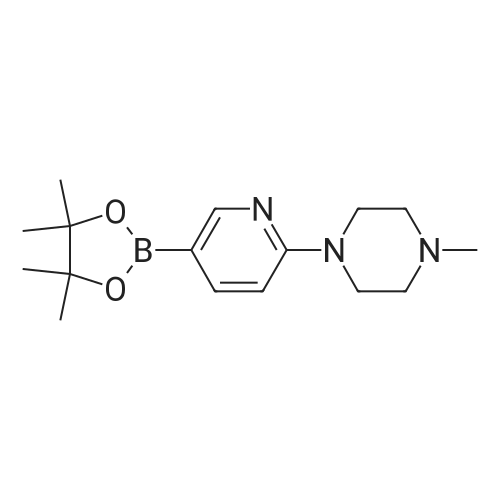
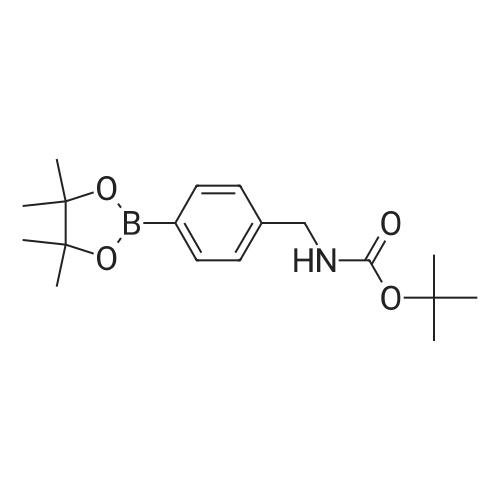
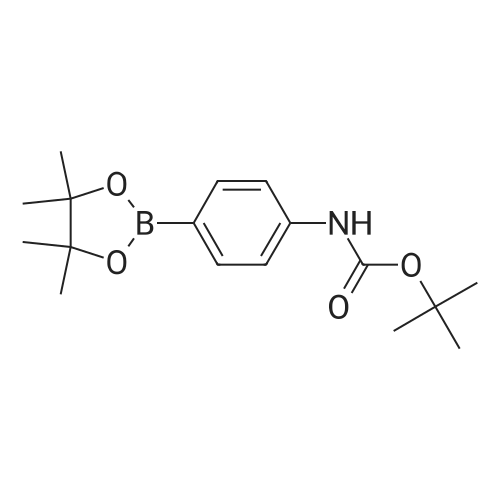
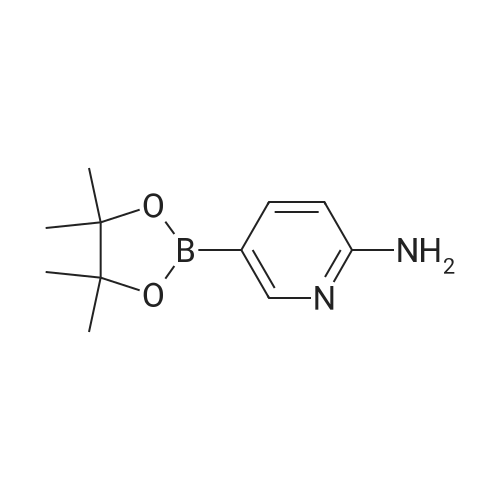
| 53% |
With potassium acetate;(1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; In N,N-dimethyl-formamide; at 80℃; for 2h;Inert atmosphere; |
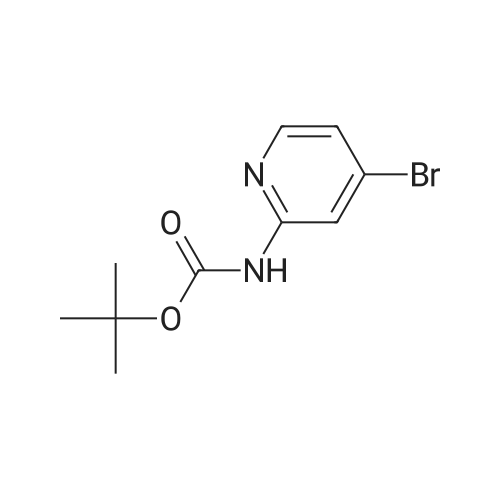
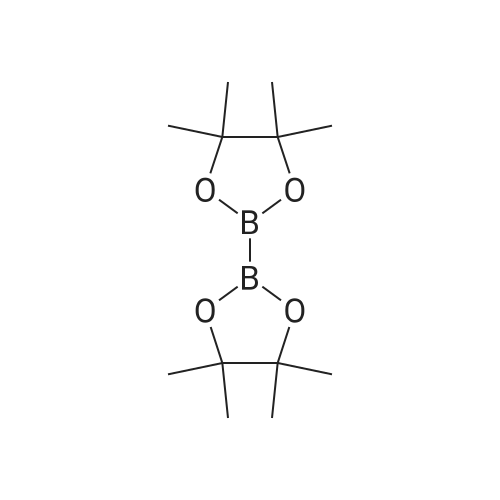
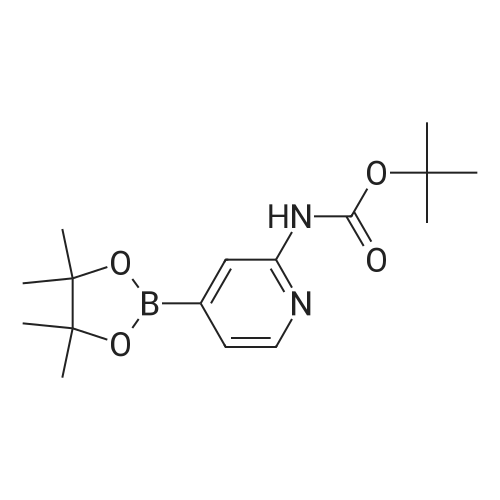
Step 6.1. tert-Butyl [4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrid-2-yl]carbamate To a solution of 6.76 (24.8 mmol) of <strong>[207799-10-8]tert-butyl (4-bromopyrid-2-yl)carbamate</strong> (Deady, Leslie W.; Korytsky, Olga L.; Rowe, Jeffrey E.; Aust. J. Chem.; 35; 10; 1982; 2025-2034) in 150 mL of dimethylformamide are added 8.0 g (81 mmol) of potassium acetate predried at 130 C. and 6.9 g (27 mmol) of bis(pinacolato)diboron. A stream of argon is then sparged through for a few moments, and 1.2 g (1.5 mmol) of [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II) are added. The mixture is stirred at 80 C. under argon for 2 hours and then poured into saturated aqueous ammonium chloride solution. The product is extracted with ethyl acetate, the organic phase is dried over sodium sulfate and the solvent is stripped off under reduced pressure. The residue is triturated in 300 mL of refluxing diisopropyl ether and the insoluble matter is separated out by filtration. The filtrate is cooled and partially concentrated under reduced pressure. After adding 70 mL of hexane, the precipitate formed is isolated by filtration to give 4.2 g of an orange-colored solid after drying. Yield: 53% m.p.: 188-193 C. 1H NMR (CDCl3) delta: 8.15 (m, 2H), 7.65 (broad s, 1H), 7.15 (d, 1H), 1.40 (s, 9H), 1.20 (s, 12H) ppm. |
| 46% |
With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium acetate; In 1,4-dioxane; at 90℃; for 3h;Inert atmosphere; |
A 20 mL Biotage microwave vial loaded with <strong>[207799-10-8]tert-butyl (4-bromopyridin-2-yl)carbamate</strong> D-1 (1 g, 3.66 mmol), 4,4,4?,4?,5,5,5?,5?-octamethyl-2,2?-bi(1,3,2-dioxaborolane) (1.3 g, 5.13 mmol), PdCl2(dppf) (215 mg, 0.293 mmol), and KOAc (1.15 g, 11.72 mmol) was capped, purged with argon, then injected with degassed dioxane (8.14 mL), and heated to 90 C. for 3 h in an oil bath. The reaction was cooled, diluted with EtOAC, poured into a separatory funnel containing EtOAc, saturated ammonium chloride solution, the mixture was shaken, and filtered through celite. After multiple extractions with EtOAc, the organics were washed with saturated ammonium chloride solution and brine. After concentration, the mixture was dissolved in Et2O and then heated/cooled repeatedly with Et2O, filtered, and washed with Et2O affording tert-butyl (4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-2-yl)carbamate D-2 White solid (540 mg, 1.686 mmol, 46% yield). 1H NMR (DMSO) delta: 9.76 (s, 1H), 8.27 (dd, J=4.7, 1.0 Hz, 1H), 8.09 (s, 1H), 7.19 (dd, J=4.8, 0.9 Hz, 1H), 1.49 (s, 9H), 1.33 (s, 12H). |
|
With potassium acetate;(1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; In DMF (N,N-dimethyl-formamide); at 80℃; for 2.16667h; |
To a mixture of compound 1 (301 mg, 1.10 mmol), bis (pinacolato) diboron (309 mg, 1.22 mmol), potassium acetate (356 mg, 3.63 mmol) and [1,1'- bis (diphenylphosphino) ferrocene] dichloropalladium II (48.0 mg, 0.0656 mmol), degassed and sealed under nitrogen, was added dry dimethylformamide (7.5 mL). Following further nitrogen purging, the mixture was stirred at 80 C for 130 min. After cooling to room temperature, compound 2 (208 mg, 0.734 mmol), [1, 1'- bis (diphenylphosphino) ferrocene] dichloropalladium II (48. 2 mg, 0.0659 mmol) and 2M aqueous sodium carbonate (2.75 mL, 5.5 mmol) were added, and then the mixture was degassed, sealed under nitrogen, and stirred at 85 C for 4 h. After cooling to room temperature, the reaction mixture was added to aqueous sodium bicarbonate (100 mL) and extracted with ethyl acetate (2x100 mL), 10% methanol in dichloromethane (3x100 mL), and then further ethyl acetate (3x100 mL). The combined extracts were concentrated under reduced pressure. The crude product was purified by silica gel chromatography (gradient elution 100% dichloromethane to 2.5% methanol/dichloromethane) to provide (3) {4- [2- (3- Ethyl-ureido) -imidazo [1, 2-a] pyridin-7-yl]-pyridin-2-yl}-carbamic acid tert-butyl ester (122 mg). (MeOH/CH2CI2/hexane) LCMS (APCI+) 397.2 (100%, MH). |
|
With potassium acetate;(1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; In N,N-dimethyl-formamide; at 80℃; for 2h;Inert atmosphere; |
Step 5.1.: tert-Butyl [4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-2-yl]carbamate A solution of 6.76 g (24.8 mmol) of <strong>[207799-10-8]tert-butyl (4-bromopyridin-2-yl)carbamate</strong> (Deady, Leslie W.; Korytsky, Olga L.; Rowe, Jeffrey E.; Aust. J. Chem.; 35; 10; 1982; 2025-2034) in 150 ml of dimethylformamide is admixed with 8.0 g (81 mmol) of potassium acetate, dried at 130 C. beforehand, and with 6.9 g (27 mmol) of bis(pinacolato)diboron. Subsequently a stream of argon is bubbled in for a few moments, and 1.2 g (1.5 mmol) of 1,1'-bis(diphenylphosphino)ferrocenedichloropalladium(II) are added. The mixture is stirred at 80 C. under argon for 2 hours and then poured into saturated aqueous ammonium chloride solution. The product is extracted with ethyl acetate, the organic phase is dried over sodium sulphate and the solvent is stripped off under reduced pressure. The residue is triturated in 300 ml of diisopropyl ether at reflux and the insoluble matter is separated by filtration.The filtrate is cooled and partially concentrated under reduced pressure. Following addition of 70 ml of hexane, the precipitate formed is isolated by filtration, to give 4.2 g of an orange solid after drying.m.p.: 188-193 C.1H NMR (CDCl3) delta: 8.15 (m, 2H), 7.85 (broad s, 1H), 7.15 (d, 1H), 1.40 (s, 9H), 1.20 (s, 12H) ppm. |
|
With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium acetate; In dimethyl sulfoxide; at 85℃; for 1h;Inert atmosphere; Sealed tube; |
fe/f-butyl (4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)pyridin-2-yl)carbamate (i6): To a stirred suspension of ie f-butyl (4-bromopyridin-2-yl)carbamate (i5) (0.5 g, 1.8 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1 ,3,2-dioxaborolane) (1 .4 g, 5.5 mmol), potassium acetate (0.541 g, 5.5 mmol) in DMSO (8 ml_), argon was purged for 15 min and PdCI2 (dppf) (0.15 g, 0.18 mmol) was added. The reaction was heated at 85C for 1 h in a sealed tube. The progress of the reaction was monitored by TLC. After completion, the reaction mixture was diluted with ethyl acetate washed with 0.2 M HCI solution. The organic layer was dried over sodium sulphate and concentrated under reduced pressure to afford ie f-butyl (4-(4,4,5,5-tetramethyl-1 ,3,2- dioxaborolan-2-yl)pyridin-2-yl)carbamate (i6) (1 .5 g, crude), compound was used as such in the next reaction. |
|
With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; potassium acetate; In N,N-dimethyl-formamide; at 80℃; for 3h;Inert atmosphere; |
A mixture of ferf-butyl 4-bromopyridin-2-ylcarbamate (0.301 g, 1 .10 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1 ,3,2-dioxaborolane) (0.309 g, 1 .22 mmol), potassium acetate (0.356 g, 3.36 mmol) and [1 ,1 ,-b/s(diphenylphosphino)ferrocene]dichloropalladium(ll)-dichloromethane (0.048 g, 0.066 mmol) in dry A/,A/-dimethylformamide (7.5 ml_) was stirred at 80 C for 3 h under nitrogen. After being cooled to room temperature, 1 -(bromomethyl)-3-chlorobenzene (0.1 50 g, 0.73 mmol, [1 ,1 ,-b/s(diphenylphosphino)ferrocene]dichloropalladium(ll)-dichloromethane (0.048 g, 0.066 mmol), sodium carbonate (0.0583 g, 5.5 mmol) and water (2.5 ml_) were added. The mixture was stirred at 85 C for 2 h under nitrogen atmosphere. The reaction was concentrated, under reduced pressure, and the residue was purified by column chromatography (silica gel, petroleum ether/ethyl acetate = 1 /1 ) to obtain ferf-butyl 4-(3-chlorobenzyl)pyridin-2-ylcarbamate (0.036 g, 0.1 1 mmol, 10.3% for 2 steps) as a white solid. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping