|
With 4-methoxy-phenol; hydroquinone; In tert-butyl alcohol; at 180 - 190℃; for 6h;Product distribution / selectivity; |
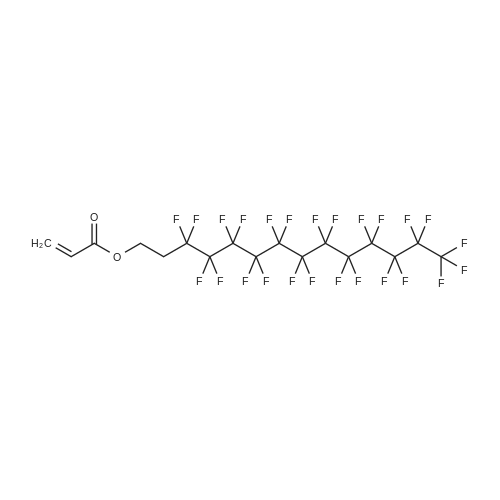
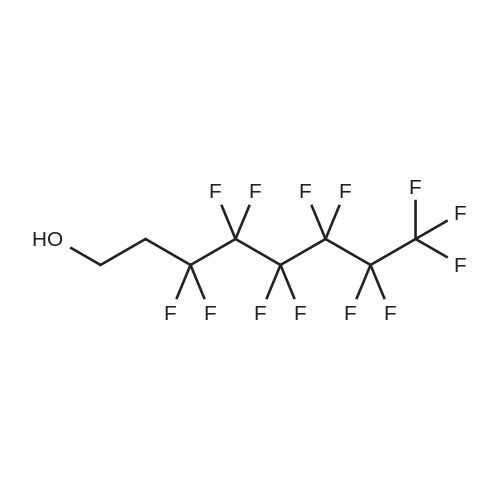
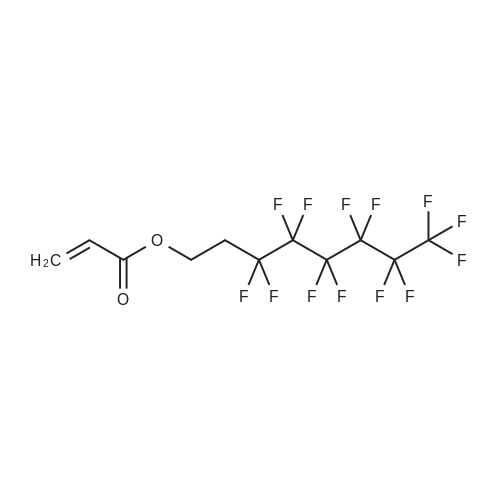
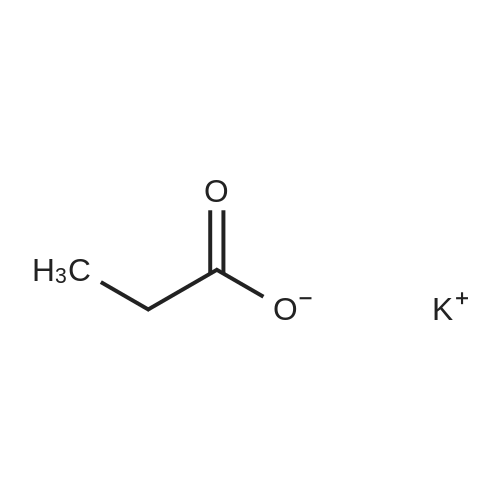
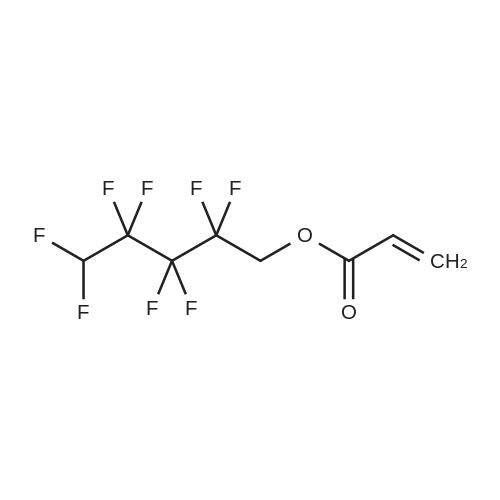
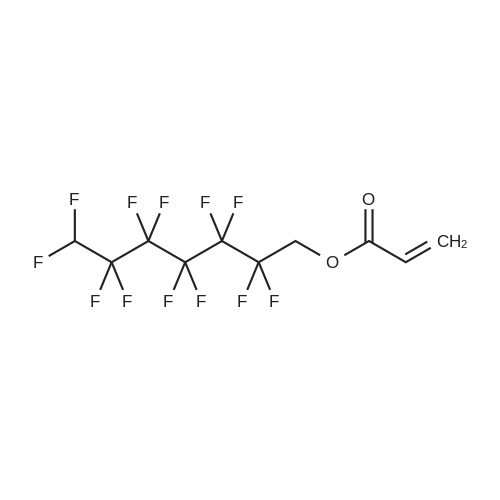
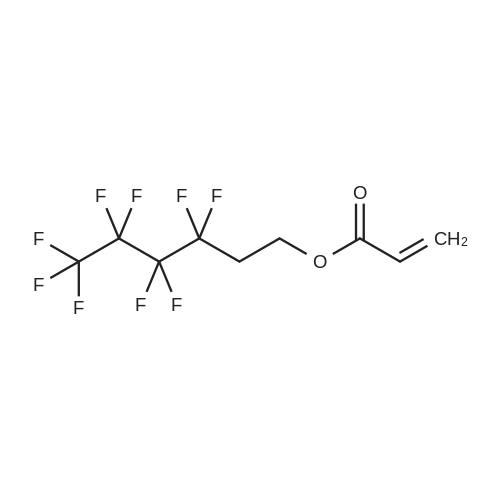
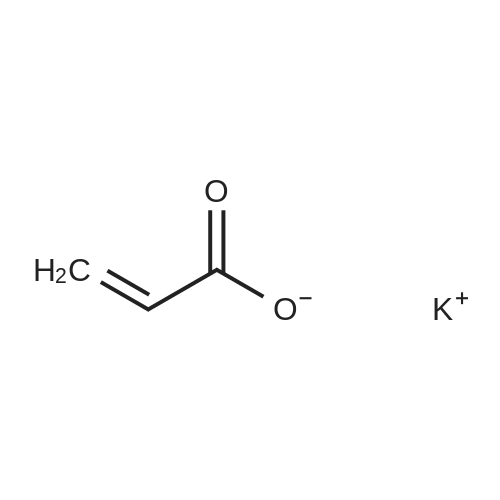
(Example 3) A mixture containing the fluorine-containing acrylic esters represented by the formula (1) was obtained by obtaining a mixture of the ethylene adducts of the fluoroalkyl iodides, which adducts were represented by C8F17CH2CH2I (n=7) and then reacting this mixture with the acrylic acid compound. Specifically, 1533g (2.67 mol) of the ethylene adducts of fluoroiodides, 320g (2.90 mol) of potassium acrylate, 680mL of tert-butyl alcohol, and 1.8g of hydroquinone and 0.32g of hydroquinone monomethylether as inhibitors of polymerization were charged into an autoclave with a volume of 3L and heated to 180C to 190C and reacted for 6 hours. After the reaction, a reaction mixture was cooled. Next, KI as a byproduct was removed by filtration. Thereafter, a filtrate was subjected to distillation to remove tert-butyl alcohol and then 887g of a reaction mixture having a composition shown in Table 1 was obtained. 500g of this reaction mixture was measure and charged into a still and subjected to distillation for 6 hours at a pressure in a rectifier of 0.9kPa and a still temperature of 160C, introducing nitrogen into the still at a flow rate of 20 ml/min such that the oxygen concentration was 0 mol% relative to the total moles of gas-phase components in the distillation column. Further, the distillation column used in this example was the same as that used in Example 1. As a result of distillation, 272g of C8F17CH2CH2OCOCH=CH2 having a composition as shown in Table 1 was obtained as a distillate a distillate liquid (a recovery rate based on the fluorine-containing acrylic esters was 59%). In Table 1, the compositions of the reaction mixture and the mixture after distillation were determined by gas chromatography. Table 3 Component Reaction mixture (mol%) Mixture after distillation (mol%)C8F17CH=CH2 7.04 0C8F17CH2CH2OH 1.79 0.32C8F17CH2CH2OCOCH=CH2 88.79 97.18 As shown in Table 3, a highly-pure mixture of the fluorine-containing acrylic esters with less impurities was obtained also in Example 3, similarly to Examples 1 and 2. Specifically, the mixture contained, as the impurity, C8F17CH2CH2OH (n=7) in an amount of 0.32 mol%.(Comparative Example 2) The ethylene adducts of the fluoroalkyl iodides, which adduct was represented by C8F17CH2CH2I (n=7) was obtained and then this mixture was reacted with the acid ester compound to give a mixture containing the fluorine-containing acrylic esters represented by the formula (1). Specifically, 1533g (2.67 mol) of the ethylene adduct of the fluoroiodide and 320g (2.90 mol) of acrylate potassium, 680mL of tert-butyl alcohol, 1.8g of hydroquinone and 0.32g of hydroquinone monomethylether as the inhibitors of polymerization were firstly charged into an autoclave with a volume of 3L and heated to 180C to 190C and reacted for 6 hours. After the reaction, a reaction mixture was cooled. Next, KI as a byproduct was removed by filtration. Thereafter, a filtrate was subjected to distillation to remove tert-butyl alcohol and then 1360g of a reaction mixture having a composition shown in Table 1 was obtained. 500g of this reaction mixture was measured and charged into a still and subjected to distillation at a pressure in a distillation column of 0.9kPa and a still temperature of 160C, introducing air into the still at a flow rate of 20 ml/min such that the oxygen concentration in the distillation column was 1 mol% relative to the total moles of gas-phase components in the distillation column. Further, the continuous distillation was carried out using an oldershaw-type distillation column with 20 plates which is made of iron as a whole (including the internal structure). As a result of the distillation, 412g of C8F17CH2CH2OCOCH=CH2 having a composition as shown in Table 1 was obtained as a distillate liquid (a recovery rate based on the fluorine-containing acrylic esters was 89%). In Table 1, the compositions of the reaction mixture and the mixture after distillation were determined by gas chromatography. Table 4 Component Reaction mixture (mol%) Mixture after distillation (mol%)C8F17CH=CH2 7.04 NDC8F17CH2CH2OH 1.79 NDC8F17CH2CH2OCOCH=CH2 88.79 97.44 Comparative Example 2 is an example wherein the internal structure of the distillation column was made of iron having a low electrode potential. As shown in Table 4, a highly-pure mixture of the fluorine-containing acrylic esters with less impurities was obtained also in Comparative Example 2, similarly to Examples 1, 2 and 3. However, the recovery rate was low and the resultant distillate liquid was colored brownish yellow which could not be used as a product. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping