| 93.3 % |
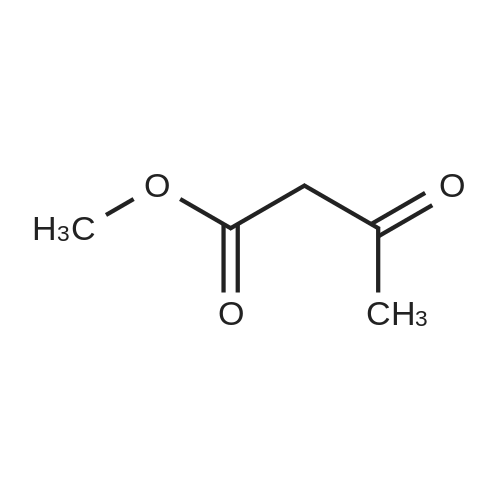
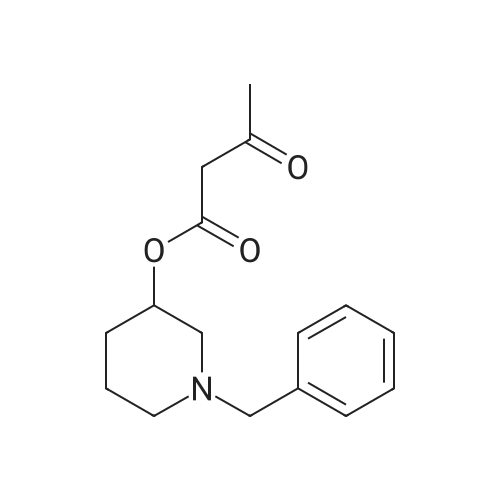
Stage #1: methyl (Z)-2-(3-nitrobenzylidene)-3-oxobutanoate; 1-benzylpiperidin-3-yl acetoacetate With ammonium acetate In methanol at 50 - 60℃; Large scale;
Stage #2: With hydrogenchloride In water at 20 - 30℃; Large scale; |
3; 4; 5; 6; 2; 3; 4 Example 3 Preparation of benidipine hydrochloride crude product:
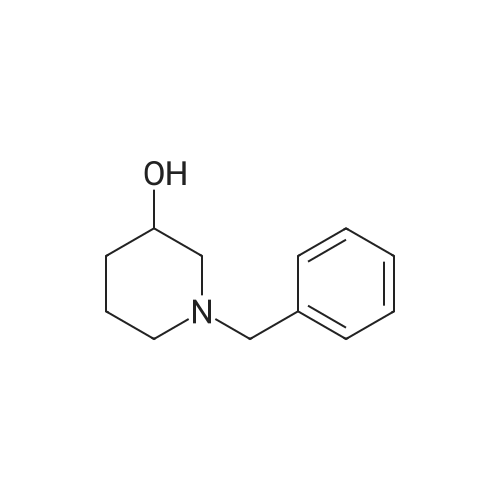
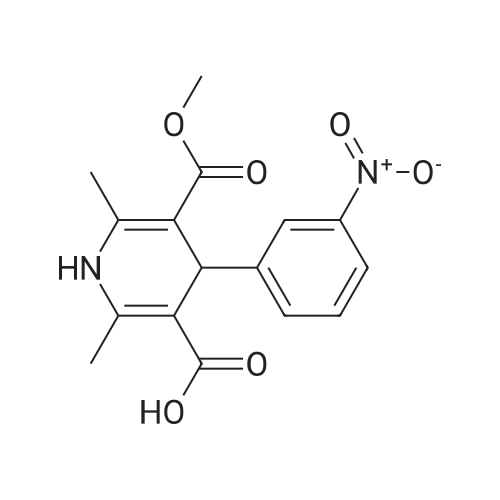
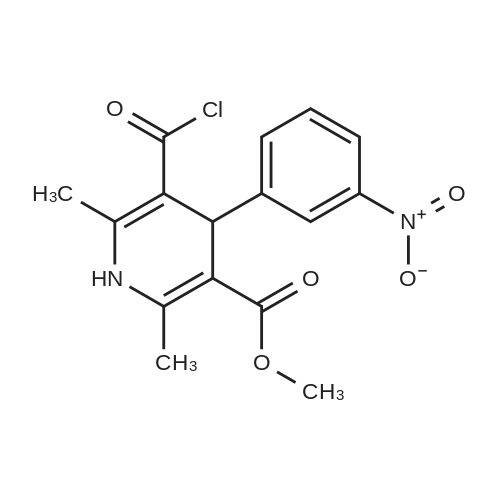
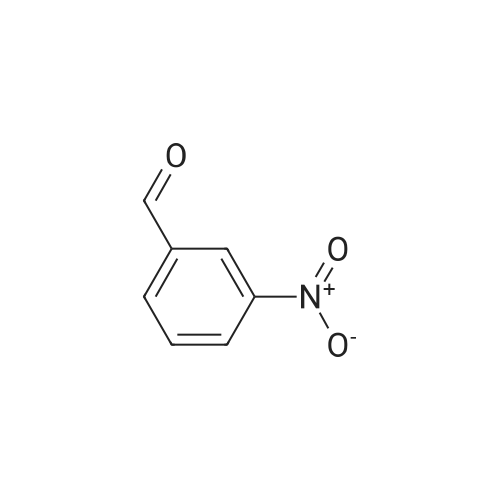
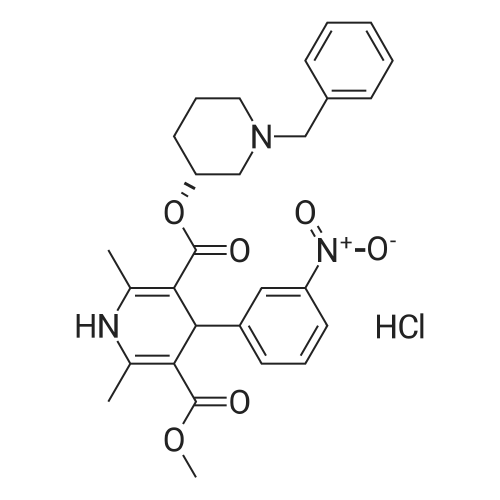
80.0kg methanol was put into the reactor, stirring was started, then 41.6kg intermediate P1 (2 equivalents, from Example 1), 12.9kg ammonium acetate (2 equivalents) and 23.0kg intermediate P2 (1 equivalents, from Example 1) were added respectively. Example 2); 5060 of stirring reaction 6 hours, cool down to 25 , filter, filter cake is washed with 24.0kg methanol, filtrate sampling, HPLC detection result is shown in Figure 3; The pressure is -0.09-0.07MPa) until no fraction flows out; 90.0kg of methylene chloride and 45.0kg of purified water are added to the reactor, stirred for 5 minutes, left standing for 30 minutes, layered, and the lower organic phase is collected; Add 45.0kg of methylene chloride, stir for 5 minutes, leave standstill for 30 minutes, layer by layer, collect the lower organic phase; the lower organic phase and the organic phase of the previous step are combined; slowly add 12.0kg of carbonic acid to the combined organic phase The mixed solution of sodium hydrogen and 45.0kg purified water was turned on and stirred for 5 minutes, left standstill for 30 minutes, layered, and the lower organic phase was collected; 30.0 kg of purified water and 15.0 kg of concentrated hydrochloric acid were added to the lower organic phase, at 2030 Stir for 30 minutes, let stand for 30 minutes, separate layers, and collect the lower organic phase; add 5.0 kg of ammonium chloride and 45.0 kg of purified aqueous solution to the lower organic phase, stir for 5 minutes, and let stand for 30 minutes; layer and collect Lower organic phase; open stirring, add 8.0kg anhydrous sodium sulfate to the organic phase, stir for 30 minutes; filter, remove anhydrous sodium sulfate, and the filter cake is washed once with 23.0kg methylene chloride; the gained filtrate is concentrated under reduced pressure ( T=≤45, the vacuum pressure is ≤-0.05MPa) until no fraction flows out, add 23.0kg acetone to the reaction kettle, after stirring evenly, continue to concentrate under reduced pressure (T=1545, the vacuum pressure is ≤-0.05 MPa) until no fraction flows out; add 23.0kg acetone to the reactor again, after stirring, continue to concentrate under reduced pressure (T=1545 , vacuum pressure is ≤-0.05MPa) until no fraction flows out; in the reactor Add 230.0kg of acetone, stir at 30-50°C for 30 minutes to fully dissolve the material; cool the material to 20-30°C, and stir until a small amount of solid is precipitated; when a small amount of solid appears, continue stirring at 25°C for 24h ; Filtration, the filter cake is beaten with 23.0kg acetone, drained, and discharged; vacuum drying (T=5565, vacuum pressure≤-0.05Mpa) to loss on drying≤0.5%, discharging to obtain 15.0 kg of benidipine hydrochloride crude product, the molar yield is 33.1%, Add 150.0kg of absolute ethanol to the reaction kettle, start stirring, add 15.0kg of benidipine hydrochloride crude product (from embodiment 3); the reaction solution is heated to 6065 , all solids are dissolved; Heat filtration, wash the reactor and filter with 16.0kg of absolute ethanol, and transfer the filtrate to the crystallization tank; reduce the temperature of the feed liquid in the crystallization tank to 20-30°C; control the temperature of the feed liquid in the crystallization tank Stir at 20-30°C for 3 hours; collect the solid by filtration and discharge to obtain a wet product of ethanol, take a sample at 40-45°C and vacuum-dry to constant weight for IR detection; put the wet product of ethanol into a crystallization bottle , add 124.0kg of acetone, and stir at 20-30°C until the system turns bright yellow, which takes about 24 hours; collect the solid by filtration, wash the solid with 12.4kg of acetone, and discharge the material to obtain acetone wet product; put the wet product into vacuum drying Oven drying (T=5560 DEG C), be dried to loss on drying≤0.5%; Dry product is packed in the high-efficiency pulverizer feeding bin and pulverized, screened, discharged; Obtain benidipine hydrochloride finished product 14.0kg, The molar yield is 93.3%. The HPLC detection result is shown in FIG. 6, and the HPLC purity is 99.93%. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping