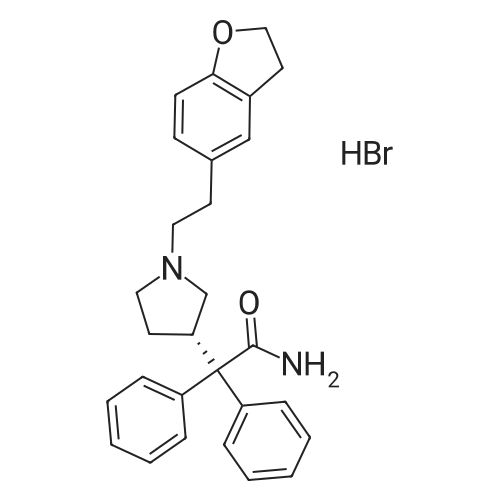
|
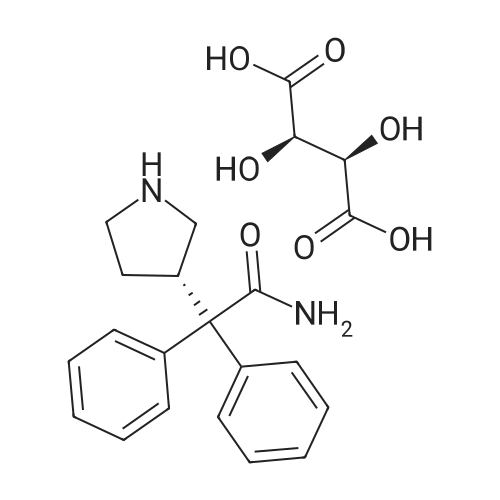
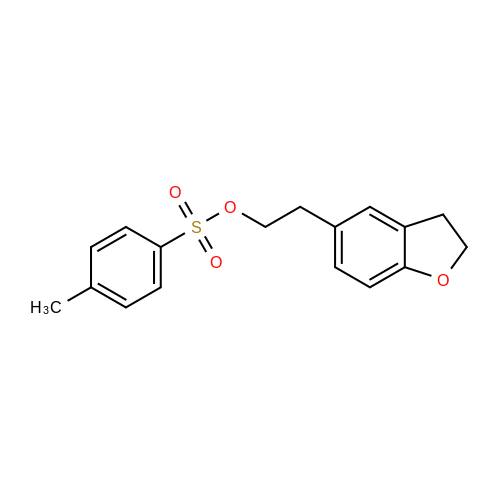
Stage #1: (S)-2,2-diphenyl-2-(pyrrolidin-3-yl)-acetamide L-tartrate; 2-(2,3-dihidrobenzofuran-5-yl)ethyl tosylate With potassium carbonate In acetonitrile at 25 - 70℃; Inert atmosphere;
Stage #2: With hydrogen bromide In dichloromethane; water at 5 - 30℃; |
1.2; 3; 4; 5; 6; 7; 8
Stage-2:PREPARATION OF DARIFENACIN HYDROBROMIDE3-(S)-(-)-(l -Carbamoyl- l , l -diphenylmethyl)pyrrolidine L-(+)-tartrate (10 g, 0.02 mol), anhydrous potassium carbonate (22.50 g, 0.16 mol) and 5-(2-tosyloxyethyl)- 2,3-dihydrobenzofuran (7 g, 0.02 mol) were suspended in anhydrous acetonitrile ( 100 ml) under nitrogen atmosphere at 25 +/- 2°C. The reaction suspension was heated to 70 +/- 2 °C and stirred for 4 h. Reaction progress was monitored by HPLC. The reaction mass was cooled to 30 + 2°C, the salts were filtered and washed with acetonitrile (10 ml). The filtrate was concentrated under reduced pressure at 50 +/- 2 °C. The residue was dissolved in dichloromethane (50 ml), water (50 ml) was added and the pH was adjusted to 2 +/- 0.1 with 24% w/w aqueous hydrobromic acid at 25- 30°C. The layers were separated and the aqueous layer was extracted with aqueous dichloromethane (20 ml). Water (50 ml) was added to the combined dichloromethane layer and pH was adjusted to 9 +/- 0.1 with 25% w/w aqueous potassium carbonate solution at 25 +/- 2°C. The layers were separated and concentrated under reduced pressure at 35-40°C. The residue was dissolved in acetone (50 ml), cooled to 5-10°C and the pH was adjusted to acidic with 48% w/w aqueous hydrobromic acid at 5-10°C. The residue was stirred for 2 h at 20-25°C, cooled to 0-5°C and stirred for 1 h at 0-5°C. The product was filtered, washed with chilled acetone (10 ml) and dried at 50-55°C.Yield: 9.4 gChromatographic purity (By HPLC): 98.2%.5 -(2-Tosy loxyethy l)-2, 3 -dihydrobenzofuran : NilDarifenacin dimer impurity: 0.96%.; EXAMPLE - 3PURIFICATION OF DARIFENACIN HYDROBROMIDE Darifenacin hydrobromide (10 g) was suspended in acetic acid (15 g) at 25 +/- 2°C and heated to 65-70°C. Activated carbon (0.25 g) was added and stin-ed for 15 min at 65-70°C. Carbon was filtered off through hyflo and washed with hot acetic acid (5 g). Water (200 ml) was added to the filtrate slowly at 50-55°C, cooled to 45°C and Darifenacin hydrobromide seed (0.05 g) was added. The resulting solution was cooled to 20-25 °C and stin-ed for 1 h and further cooled to 0-5 °C and stirred for 1 h. The solid was filtered and washed with cold water (10 ml). The product was dried at 50-55°C.Yield: 7.6 gChromatographic purity (By HPLC): 99.52%5-(2-bromoethyl)-2,3-dihydrobenzofuran: Nil 5-(2-Tosyloxyethyl)-253-dihydrobenzofuran: NilDarifenacin dimer impurity: 0.20%.EXAMPLE - 4PURIFICATION OF DARIFENACIN HYDROBROMIDEDarifenacin hydrobromide (15 g) was suspended in a mixture of acetic acid (25 g) and water (25 ml) at 25 +/- 2°C and heated to 65-70°C. Activated carbon (0.75 g) was added and stirred for 15 min at 65-70°C. Carbon was filtered off through hyflo and washed with a mixture of acetic acid and DM water (10 g). Water (120 ml) was added to the filtrate slowly at 50-55°C, cooled to 45°C and Darifenacin hydrobromide seed (0.05 g) was added. The resulting solution was cooled to 20- 25°C and stirred for 1 h and further cooled to 0-5°C and stirred for 1 h. The solid was filtered and washed with cold water (30 ml). The product was dried at 50-55°C. Yield: 1 1.9 gChromatographic purity (By HPLC): 99.71 %5-(2-bromoethyl)-2,3-dihydrobenzofuran: Nil5-(2-Tosyloxyethyl)-2,3-dihydrobenzofuran: NilDarifenacin dimer impurity: 0.20%. EXAMPLE - 5PURIFICATION OF DARIFENACIN HYDROBROMIDEDarifenacin hydrobromide (9 g) was suspended in acetone (45 ml) at 25 +/- 2°C, heated to 55-60°C and stirred for 30 + 5 min at 55-60°C. The resulting solution was cooled to 20-25°C and stin-ed for 30 + 5 min, which is further cooled to 0-5°C and stirred for 1 h. The solid was filtered and washed with chilled acetone (9 ml). The product was dried at 50-55°C.Yield: 8.8 gChromatographic purity (By HPLC): 99.87%5-(2-bromoethyl)-2,3-dihydrobenzofuran: Nil5-(2-Tosyloxyethyl)-2,3-dihydrobenzofuran: NilDarifenacin dimer impurity: 0.08%. EXAMPLE - 6PURIFICATION OF DARIFENACIN HYDROBROMIDEDarifenacin hydrobromide (9 g) was suspended in a mixture of acetone (45 ml) and DM water (1.77 ml) at 25 +/- 2°C, heated to 55-60°C and stirred for 30 + 5 min at 55- 60°C. The resulting solution was cooled to 20-25°C and stirred for 30 + 5 min, which was further cooled to 0-5°C and stirred for 1 h. The product was filtered and washed with chilled acetone (9 ml). The product was dried at 50-55°C.Yield: 8.4 gChromatographic purity (By HPLC): 99.88%EXAMPLE - 7PURIFICATION OF DARIFENACIN HYDROBROMIDEDarifenacin hydrobromide (10 g) was suspended in a mixture of acetone (50 ml) and DM water (3.95 ml) at 25 +/- 2°C, heated to 55-58°C and stirred for 30 +/- 5 min. The resulting solution was cooled to 20-25°C and stirred for 30 +/- 5 min, which was further cooled to 0-5 °C and stirred for 1 hour. The product was filtered and washed with chilled acetone (10ml, 0-5°C). The product was dried at 50-55°C.Yield: 8.30gChromatographic Purity (By HPLC): 99.83 %Darifenacin dimmer: 0.10%EXAMPLE - 8PURIFICATION OF DARIFENACIN HYDROBROMIDEDarifenacin hydrobromide (10 g) was suspended in a mixture of acetone (50 ml) and DM water (7.9 ml) at 25 +/- 2°C, heated to 55-60°C and stirred for 30 + 5 min. The resulting solution was cooled to 20-25°C and stirred for 30 +/- 5 min, which was further cooled to 0-5°C and stirred for 1 hour. The product was filtered and washed with chilled acetone (10 ml, 0-5°C). The product was dried at 50-55°C.Yield: 6.70gChromatographic Purity (By HPLC): 99.94 %Darifenacin dimmer: Nil. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping