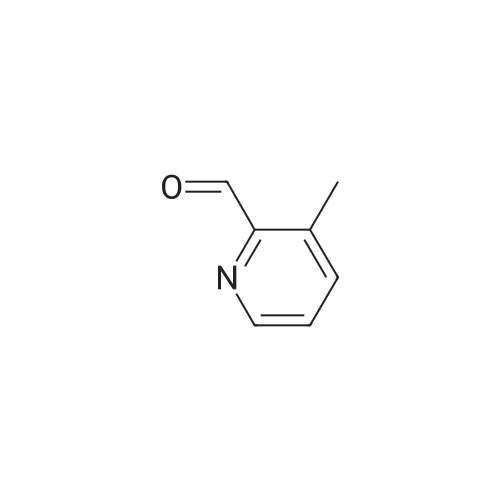
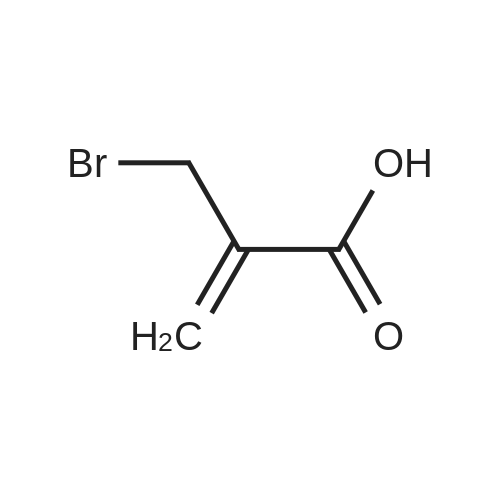
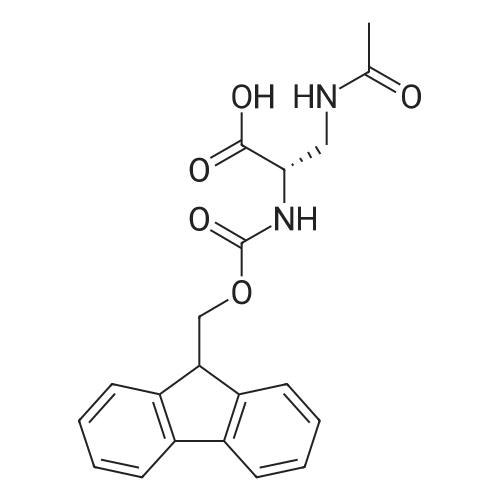
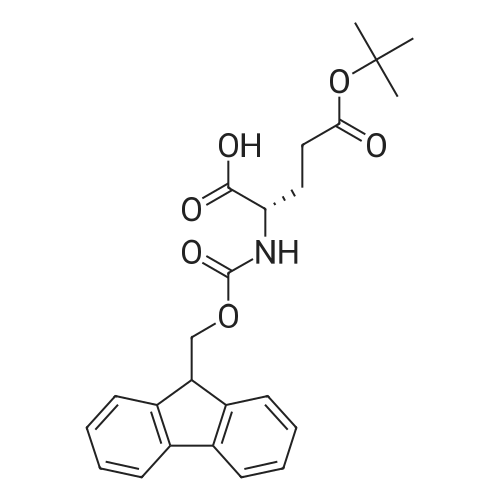
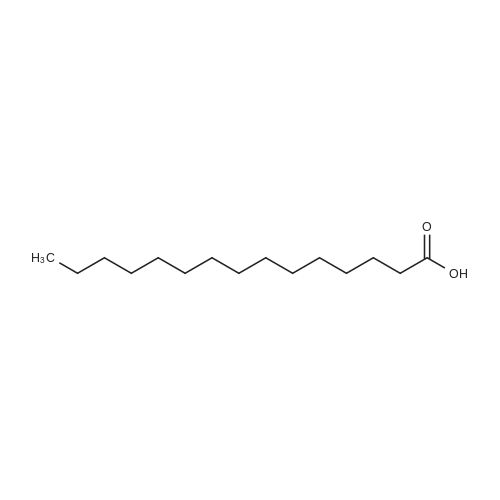
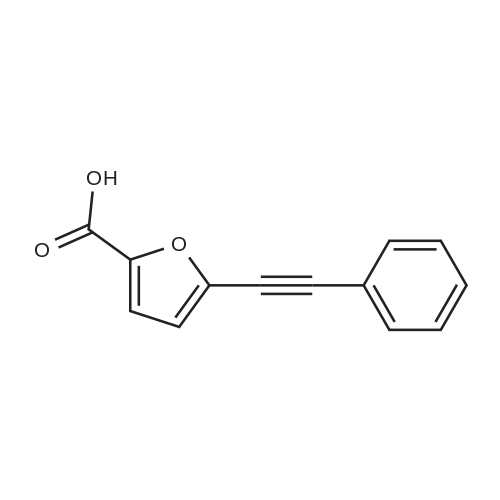
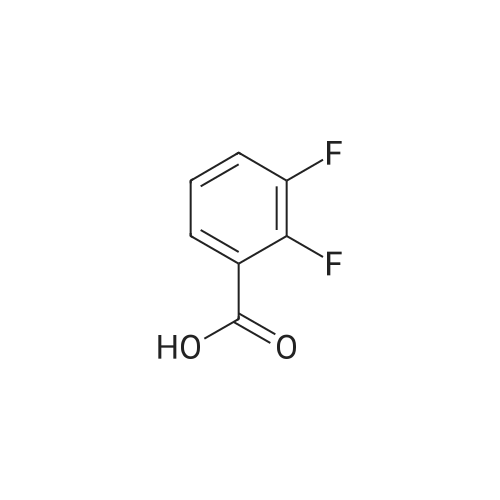
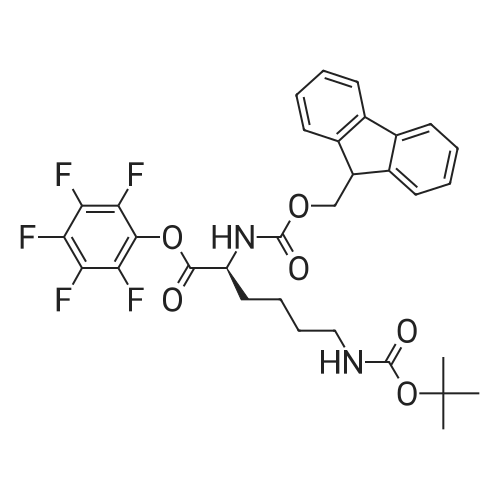
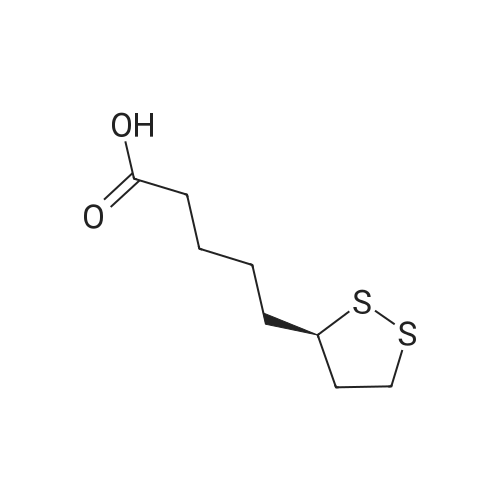
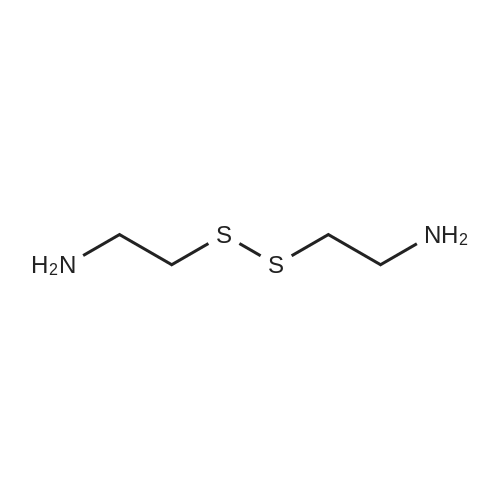
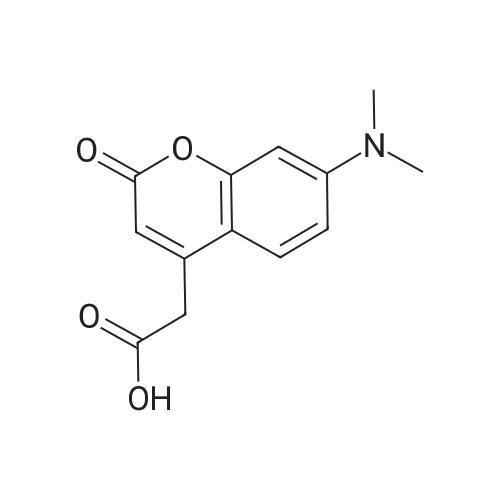
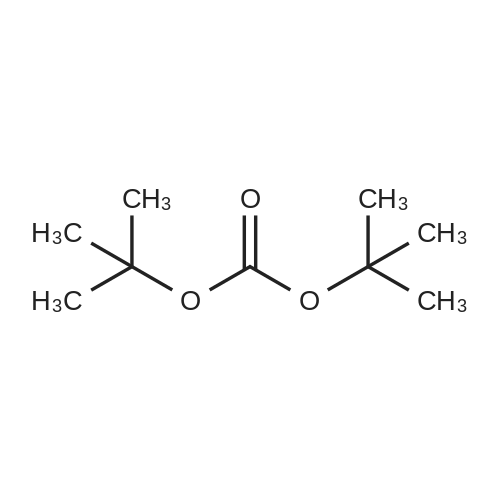
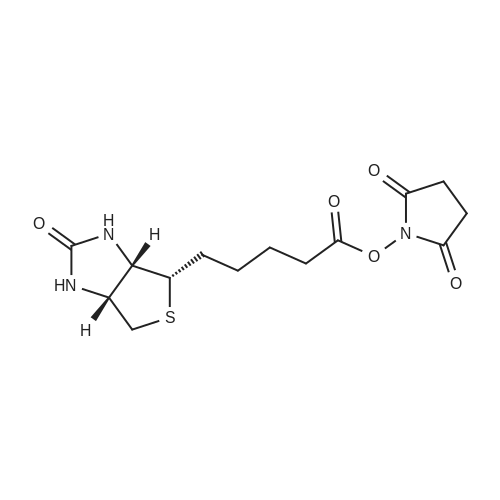
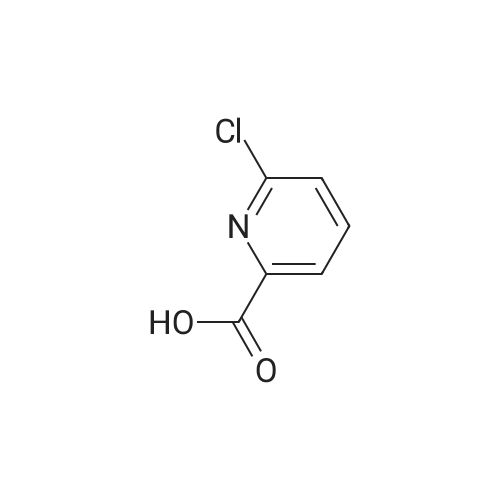
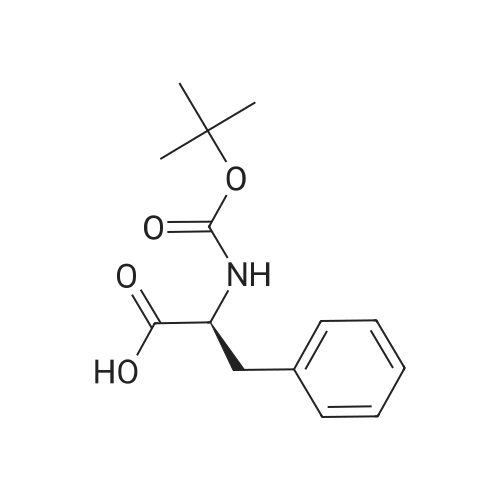
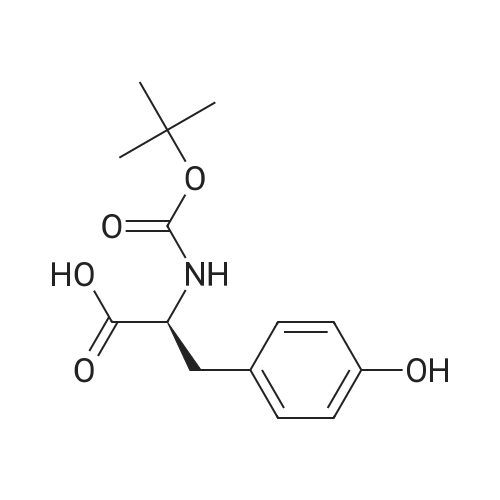
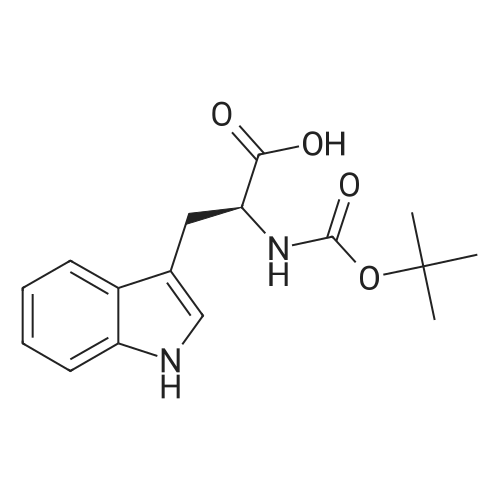
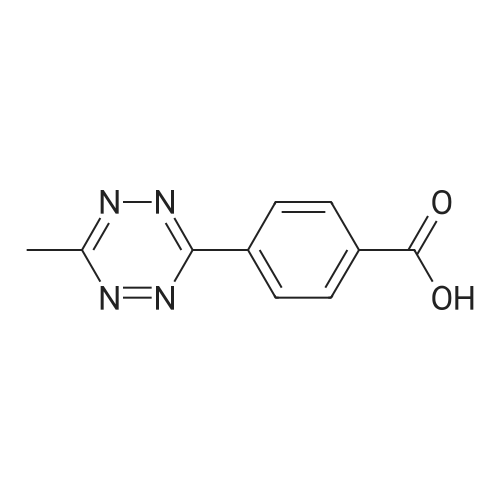
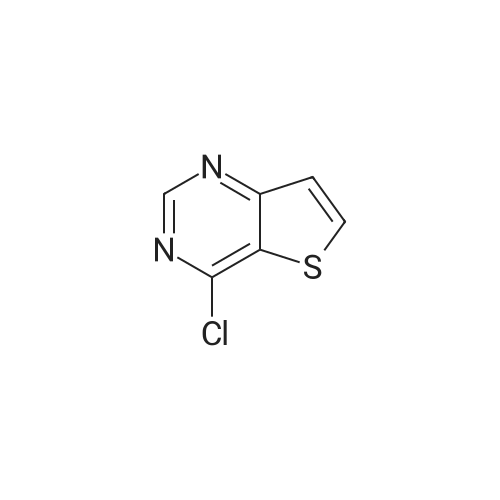
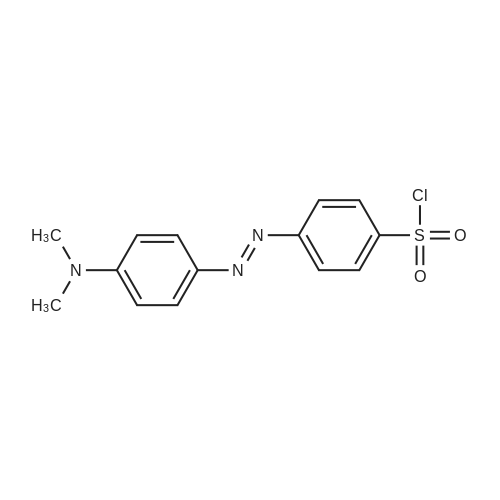
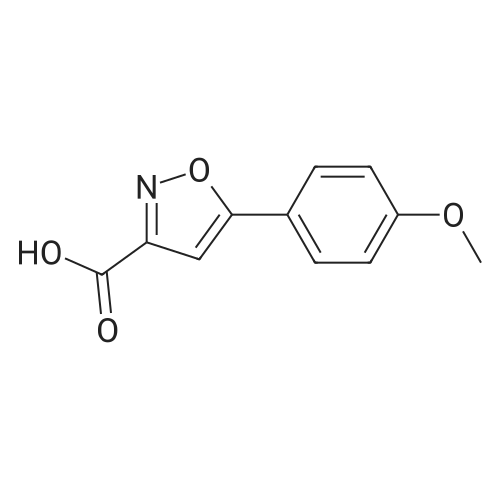
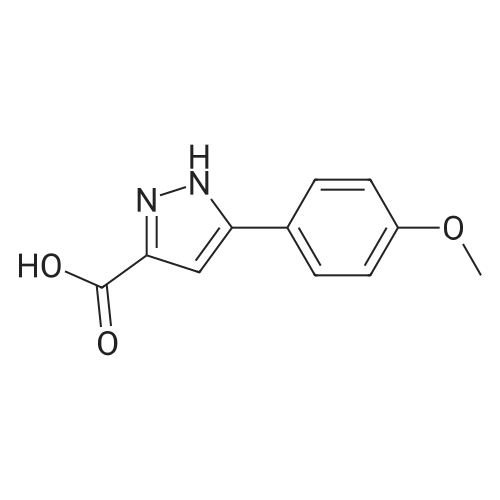
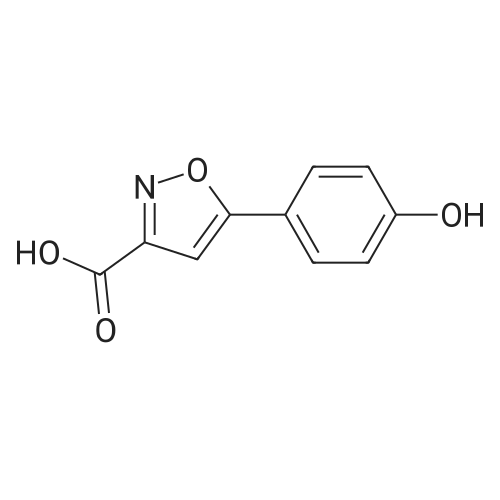
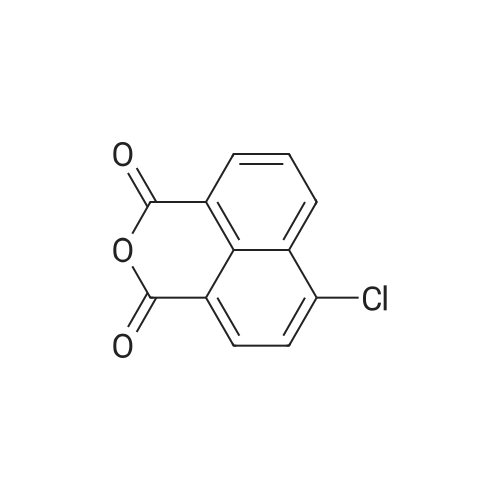
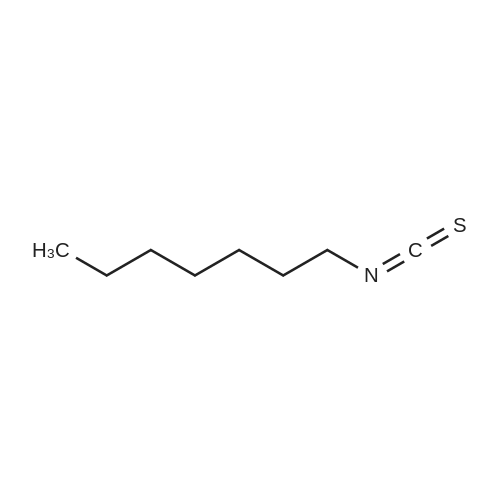
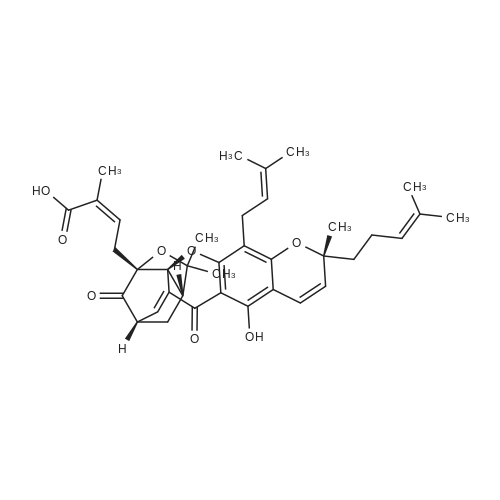
| 44% |
With triethylamine; In methanol; at 20℃; |
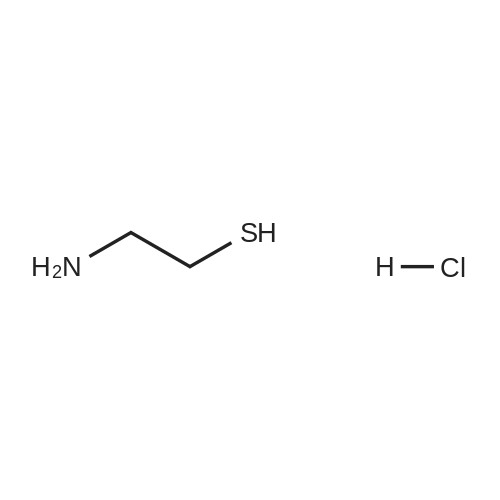
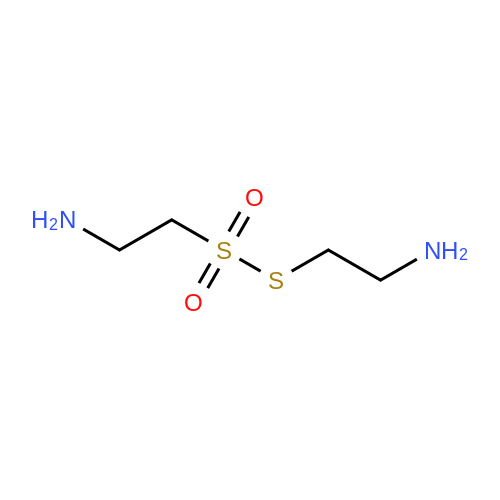
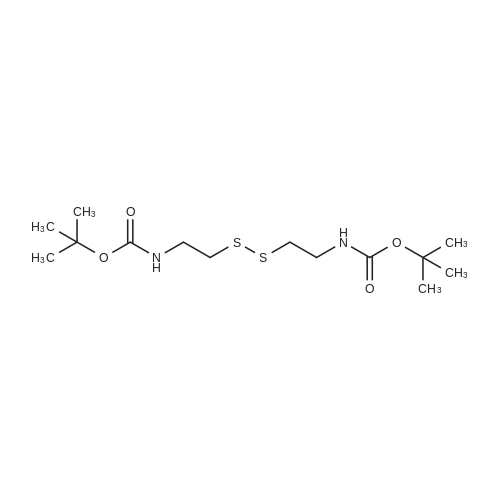
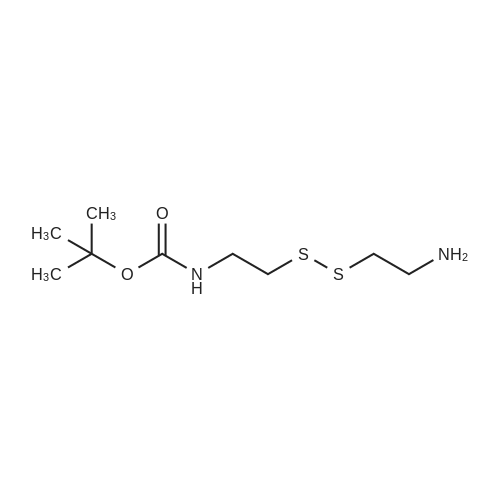
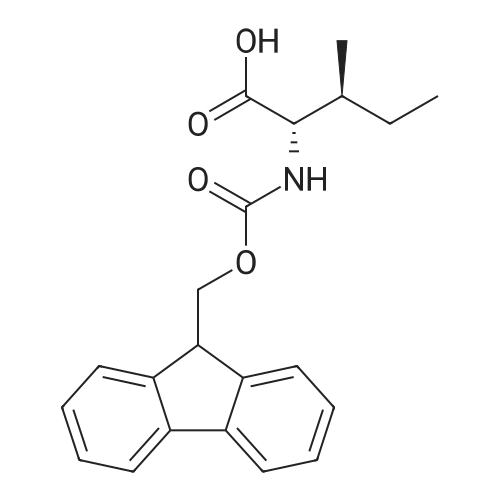
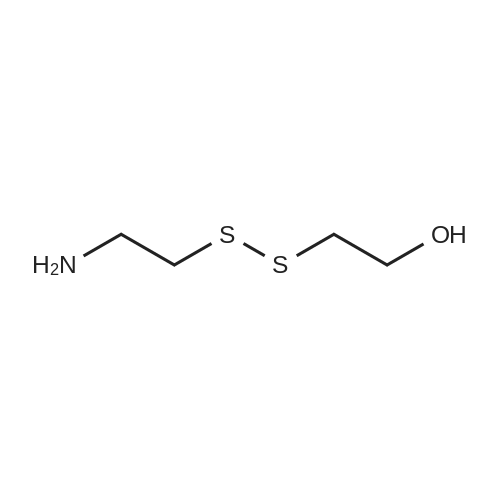
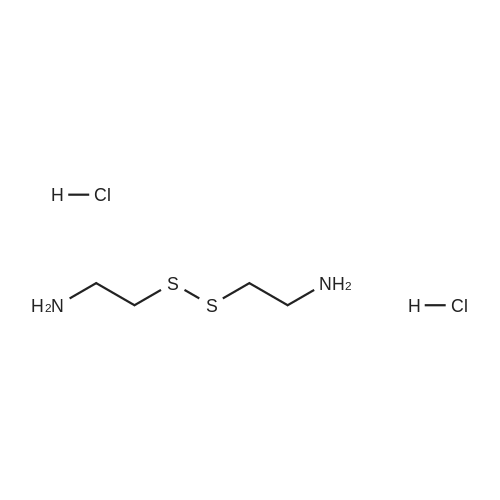
Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL) Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%).Separately, nicotinic acid (246 mg, 2.0 mmol) was taken up in CH3CN (10 mL) along with tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (503 mg, 2.0 mmol), EDCI (422 mg, 2.2 mmol). The resulting reaction mixture was stirred at room temperature for 4 h and then diluted with EtOAc. The organic layer was washed with dilute aqueous NaHCO3, brine, dried over Na2SO4, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (CH2Cl2) afforded tert-butyl 2-(2-(2-(nicotinamido)ethyl)disulfanyl)ethylcarbamate (400 mg, 56%).tert-Butyl 2-(2-(2-(nicotinamido)ethyl)disulfanyl)ethylcarbamate (200 mg, 0.56 mmol) was taken up in 25% TFA in CH2Cl2 solution (5 mL) and allowed to stand at room temperature for 4 h. The reaction mixture was then concentrated under reduced pressure to afford the TFA salt of N-(2-(2-(2-aminoethyl)disulfanyl)ethyl)nicotinamide. This material was taken up in CH3CN (10 mL) along with (4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19-hexaenoic acid (184 mg, 0.56 mmol), HATU (234 mg, 0.62 mmol) and DIEA (0.30 mL). The resulting reaction mixture was stirred at room temperature for 2 h. It was then diluted with EtOAc and washed successively with saturated aqueous NaHCO3 and brine. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (5% MeOH-CH2Cl2) afforded (N-(2-(2-(2-(4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19-hexaenamidoethyl)disulfanyl)ethyl)nicotinamide (300 mg, 86%). MS calculated for C32H45N3O2: 567.3; found: [M+H]+568. |
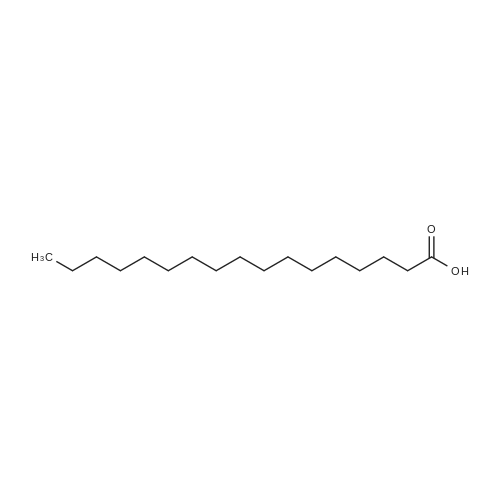
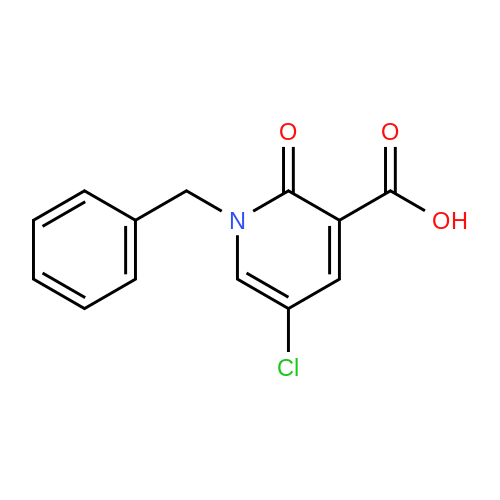
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
Example 7 Preparation of N-(2-(2-(2-(5Z,8Z,11Z,14Z,17Z)-eicosa-5,8,11,14,17-pentaenamidoethyl)disulfanyl)ethyl)-5-methyl-4-oxo-5-phenyl-4,5-dihydrofuran-2-carboxamide (I-4) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers are washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%). |
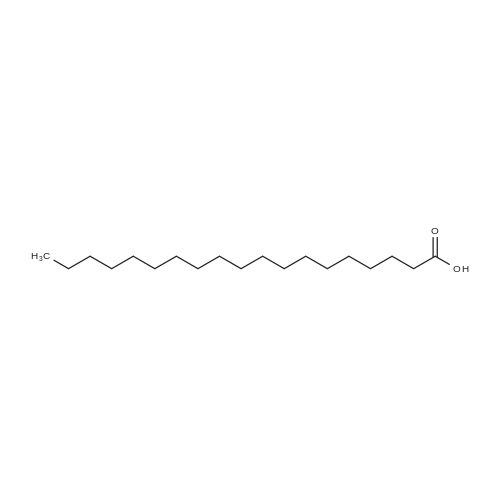
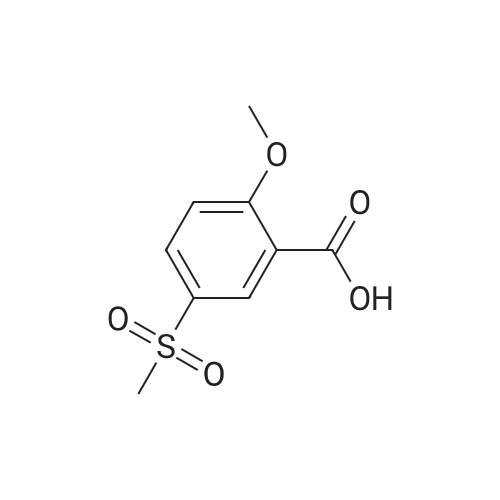
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
Example 7 Preparation of (4Z,7Z,10Z,13Z,16Z,19Z)-N-(2-(2-(2-(2-(4-(4-chlorobenzoyl)phenoxy)-2-methylpropanamido)ethyl)disulfanyl)ethyl)docosa-4,7,10,13,16,19-hexaenamide (Ia-3) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford of tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%). |
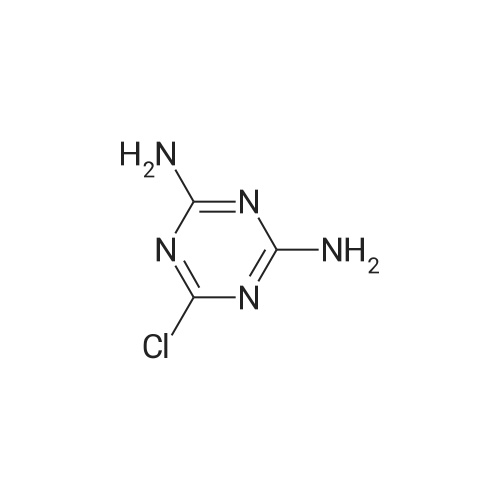
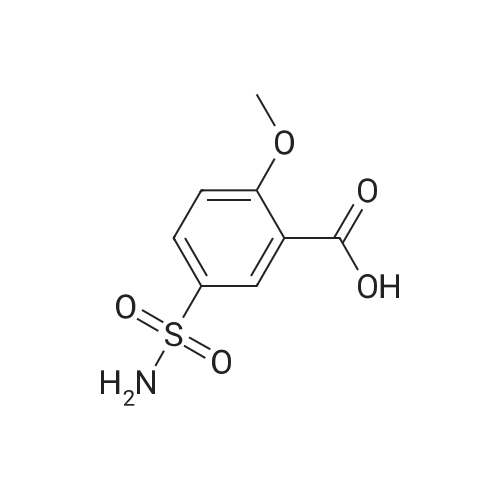
| 44% |
With triethylamine; In methanol; at 20℃; |
Example 6 Preparation of 5-((2-((2-((4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19-hexaenamido)ethyl)disulfanyl)ethyl)carbamoyl)-2-methylpyrazine 1-oxide (I-3) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in of MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%). |
| 44% |
|
Example 12 Preparation of (E)-methyl 4-(2-(2-(2-(4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19-hexaenamidoethyl)disulfanyl)ethylamino)-4-oxobut-2-enoate (Compound I-5) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in 50 mL of MeOH. Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in 5 mL of MeOH. The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 20 mL of 1M NaH2PO4. The aqueous layer was washed with 10 mL of a 1:1 solution of pentane/EtOAc, basified to pH 9 with 1 M NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried (Na2SO4) and concentrated under reduced pressure to afford 500 mg of tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (44% yield). |
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
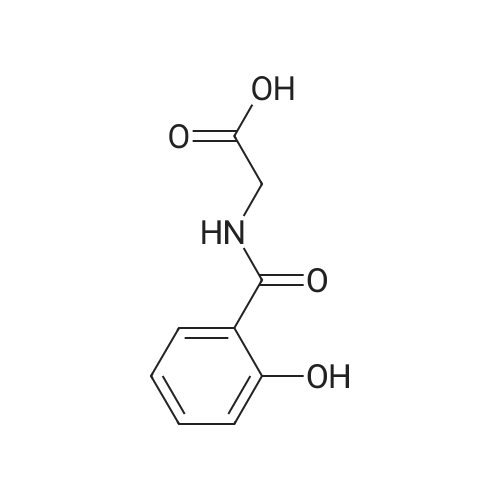
Example 5 Preparation of (S)-N-(2-(2-(2-(5-(1,2-dithiolan-3-yl)pentanamido)ethyl)disulfanypethyl)-2-hydroxybenzamide (I-2) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in 5 mL of MeOH The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M NaH2PO4 (20 mL). The aqueous layer was washed with 10 mL of a 1:1 solution of pentane/EtOAc, basified to pH 9 with 1M NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4 and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%). |
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
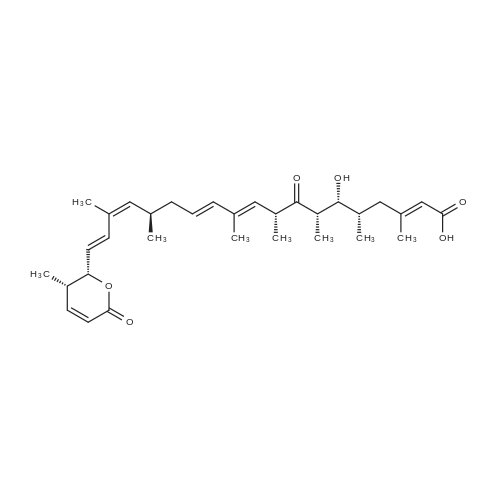
Example 5; Preparation of (4Z,7Z,10Z,13Z,16Z,19Z)-N-(2-(2-(2-((E)-6-(4-hydroxy-6-methoxy-7- methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4- enamido)ethyl)disulfanyl)ethyl)docosa-4,7,10,13,16,19-hexaenamide (I-4); Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer is washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44%).tert-Butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (1 2 mmol) was taken up in CH3CN (5 mL) along with mycophenolic acid (1.2 mmol) and EDCI (1.3 mmol). The resulting reaction mixture was stirred at room temperature for 18 h and diluted with EtOAc. The organic layer was washed with saturated aqueous NaHCO3, brine, dried over Na2SO4, filtered and concentrated under reduced pressure. The resulting residue was purified by silica gel chromatography (CH2Cl2) to afford (E)-tert-butyl 2-(2-(2-(6-(4-hydroxy-6-methoxy-7- methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4- enamido)ethyl)disulfanyl)ethylcarbamate.(E)-tert-butyl 2-(2-(2-(6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3- dihydroisobenzofuran-5-yl)-4-methylhex-4-enamido)ethyl)disulfanyl)ethylcarbamate (0.56 mmol) was taken up in 5 mL of 4 M HCl in dioxane and allowed to stand at room temperature for 2 h. The resulting reaction mixture is concentrated under reduced pressure to afford the HCl salt of (E)-N-(2-(2-(2-aminoethyl)disulfanyl)ethyl)-6-(4-hydroxy-6-methoxy- 7-methyl-3-oxo-1,3-dihydroisobenzofuran-5-yl)-4-methylhex-4-enamide. This material is taken up in CH3CN (5 mL) along with (4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19- hexaenoic acid (DHA, 183 mg, 0.56 mmol), HATU (234 mg, 0.62 mmol) and DIEA (290 1.7 mmol). The resulting reaction mixture was stirred at room temperature for 2 h. It was then diluted with EtOAc and washed successively with saturated aqueous NaHCO3 and brine. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (5% MeOH-CH2Cl2) afforded 200 mg of (4Z,7Z,10Z,13Z,16Z,19Z)-N-(2-(2-(2-9(E)-6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,3- dihydroisobenzofuran-5-yl)-4-methylhex-4-enamido)ethyl)disulfanyl)ethyl)docosa- 4,7,10,13,16,19-hexaenamide (47% yield). MS (EI) calcd for C43H60N2O6S2: 764.39; found 765 (M+1). |
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
[0238j Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2504, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44 %). |
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
[0326] Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc20 (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h. It was then concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2P04 (20 mL). The aqueous layer was washed with a 1 : 1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2S04, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44 %). |
| 44% |
With triethylamine; In methanol; at 20℃; for 3h; |
Example 8 Preparation of (4Z,7Z,10Z,13Z,16Z,19Z)-N-(2-(2-(2-(2-(1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)acetamido)ethyl)disulfanyl)ethyl)docosa-4,7,10,13,16,19-hexaenamide (I-15) (0380) Cystamine dihydrochloride (1.0 g, 4.44 mmol) was dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) was added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture was stirred at room temperature for 3 h, concentrated under reduced pressure and the resulting residue was taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer was washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers were washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (500 mg, 44% yield). (0381) tert-Butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate (150 mg, 0.595 mmol) was taken up in CH2C22 (10 mL) along with indomethacin (213 mg, 0.595 mmol) and EDCI (125 mg, 0.65 mmol). The resulting reaction mixture was stirred at room temperature for 18 h and diluted with CH2Cl2. The organic layer was washed with saturated aqueous NaHCO3, brine, dried over Na2SO4, filtered and concentrated under reduced pressure. The resulting residue was purified by chromatography (CH2Cl2) to afford 312 mg of tert-butyl 2-(2-(2-(2-(1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)acetamido)ethyl)disulfanyl)ethylcarbamate (89% yield). (0382) tert-Butyl 2-(2-(2-(2-(1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)acetamido)ethyl)disulfanyl)ethylcarbamate (312 mg, 0.528 mmol) was taken up in 6 mL of 4 M HCl in dioxane and allowed to stir at room temperature for 2 h. The resulting reaction mixture was concentrated under reduced pressure to afford the HCl salt of N-(2-(2-(2-aminoethyl)disulfanyl)ethyl)-2-(1-(4-chlorobenzo yl)-5-methoxy-2-methyl-1H-indol-3-yl)acetamide. This material was taken up in CH3CN (5 mL) along with (4Z,7Z,10Z,13Z,16Z,19Z)-docosa-4,7,10,13,16,19-hexaenoic acid (173 mg, 0.528 mmol), HATU (220 mg, 0.58 mmol) and DIEA (275 muL, 1.6 mmol). The resulting reaction mixture was stirred at room temperature for 2 h, diluted with EtOAc and washed successively with saturated aqueous NaHCO3 and brine. The organic layer was dried over Na2SO4, filtered and concentrated under reduced pressure. Purification by silica gel chromatography (5% MeOH-CH2Cl2) afforded 220 mg of (4Z,7Z,10Z,13Z,16Z,19Z)-N-(2-(2-(2-(2-(1-(4-chlorobenzoyl)-5-methoxy-2-methyl-1H-indol-3-yl)acetamido)ethyl)disulfanyl)ethyl)docosa-4,7,10,13,16,19-hexaenamide (52% yield). MS (EI) calcd for C45H56ClN3O4S2: 801.34; found 802 (M+1). |
| 32% |
With triethylamine; In methanol; at 20℃; for 18h;Reflux; Inert atmosphere; |
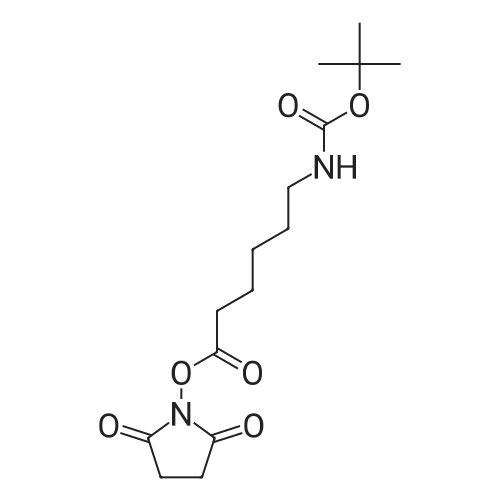
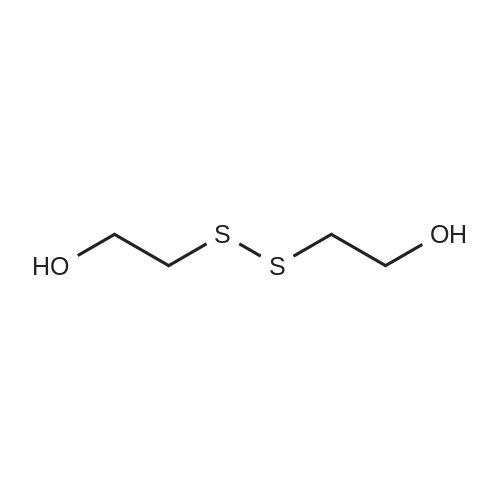
(0171) Preparation of [2-(2-Amino-ethyldisulfanyl)-ethyl]-carbamic acid tert-butyl ester: Cysteamine dihydrochloride L6 (168.75 g, 750 mmol) was dissolved in a solution of 23% TEA in CH3OH (1600 mL). A solution of di-tert- butyldicarbonate (66 g, 300 mmol) in methanol (150 mL) was added to this mixture with vigorous stirring. The mixture was refluxed for 2 hr and then left to stir at RT for 16 hr. The methanol and TEA were removed in vacuo, and water was added into the mixture. Aqueous NaOH solution (4.0 M) was added slowly to adjust the pH to 4-5 and the filtrate collected by filtration. The precipitate was washed with aqueous HCl (1.0 M, 50 mL). The acidic layer was extracted with EtOAc (250 mL). The pH value was adjusted to 5 with NaOH (4.0 M) and extracted with EtOAc (250 mL), followed by a final extraction (250 mL EtOAc) at pH 11. The combined organic layers were washed carefully with saturated aqueous NaHCO3 solution (2×200 mL), brine (2×200 mL), dried over Na2SO4 and filtered, evaporated to give the product L8 as an orange oil (60 g, 32%). 1H NMR (300 MHz, CDCl3): delta 1.50 (s, 9H), 2.77-2.83 (m, 4H), 3.04 (t, J = 6 Hz, 2H), 3.74-3.49 (m, 2H), 4.97 (m, 1H); LC-MS: (M+H)+ 253; HPLC >95%. |
|
With triethylamine; In methanol; at 20℃; for 3h; |
Cystamine dihydrochloride (1.0 g, 4.44 mmol) is dissolved in MeOH (50 mL). Triethylamine (1.85 mL, 3 eq) is added at room temperature, followed by dropwise addition of Boc2O (0.97 g, 4.44 mmol) as a solution in MeOH (5 mL). The resulting reaction mixture is stirred at room temperature for 3 h. It is then concentrated under reduced pressure and the resulting residue is taken up in 1M aqueous NaH2PO4 (20 mL). The aqueous layer is washed with a 1:1 solution of pentane/EtOAc (10 mL), basified to pH 9 with 1M aqueous NaOH, and extracted with EtOAc. The combined organic layers are washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to afford tert-butyl 2-(2-(2-aminoethyl)disulfanyl)ethylcarbamate. |
|
With triethylamine; In methanol; at 20℃; for 0.5h; |
200 mg of cystamine dihydrochloride (labeled as cystamine) dissolve in 10 ml of methanol, and then 386 mul of triethylamine was added; the 192.5 mg of di-tert-butyl dicarbonate(BOC anhydride) dissolve in 2 ml of methanol added dropwise to the solution of cystamine dihydrochloride in methanol, the reaction was stirred at room temperature for 30 minutes, and the methanol was evaporated to dryness by rotary evaporation. Continue to add 50ml of sodium dihydrogen phosphate solution (NaH2PO4, 1M), then washed twice with ether to remove the product with BOC anhydride attached to both ends. The pH of the solution was adjusted to 9 using NaOH (1M), extracted with ethyl acetate to obtain cystamine protected by BOC at one end, and the ethyl acetate layer was evaporated to dryness to obtain a white product (labeled as cystamine-BOC). |
|
With triethylamine; In methanol; at 20℃; for 0.5h; |
1) Dissolve 200 mg of cystamine dihydrochloride (labeled cystamine) in 10 ml of methanol.And add 386mul triethylamine;192.5 mg di-tert-butyl dicarbonate (BOC anhydride)Dissolved in 2 ml of methanol,Add dropwise to the cystamine dihydrochloride methanol solution,Stir the reaction at room temperature for 30 minutes and rotovap to dry the methanol.Continue to add 50ml of sodium dihydrogen phosphate solution (NaH2PO4, 1M),Wash twice with ether to remove the product with BOC anhydride attached to both ends.Using NaOH (1M) to adjust the pH of the solution to 9, extracting with ethyl acetate to obtain one-side BOC-protected cystamine, and evaporating to dry the ethyl acetate layer,A white product was obtained (labeled cystamine-BOC). |
|
With triethylamine; In methanol; at 20℃; for 105h; |
Cystamine dihydrochloride3.38gDispersed in 300 ml of methanol, this is liquid A,Dicarbonyl di-tert-butyl ester 3.27gAnd 6.3 mL of triethylamine was dissolved in 25 ml of methanol, which was B solution, and B solution was added dropwise to the solution A in 45 minutes. After the dropwise addition was completed, the reaction was further stirred at normal temperature for 60 minutes.After completion of the reaction, the solvent was removed by vacuum evaporation at 50 C, and 35 ml of sodium dihydrogen phosphate solution 1 M was added thereto, and the mixture was washed three times with 45 ml of diethyl ether, and the solution was adjusted to basic pH = 9 with sodium hydroxide solution 1 M. The mixture was extracted 3 times, and the organic phase was combined, dried over anhydrous sodium sulfate, and evaporated to give a pale yellow liquid by rotary evaporation at 60 C, and dried at 50 C to give the final product. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping