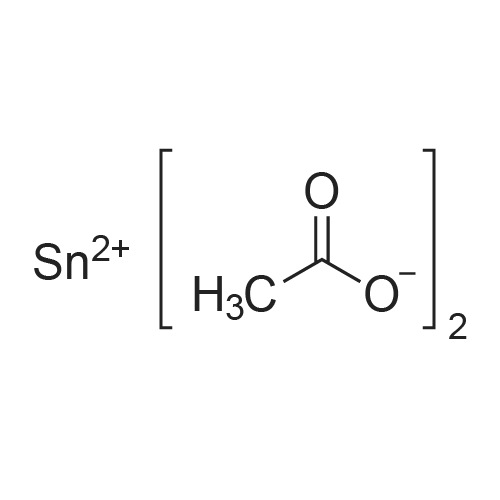
| 98.8 - 100% |
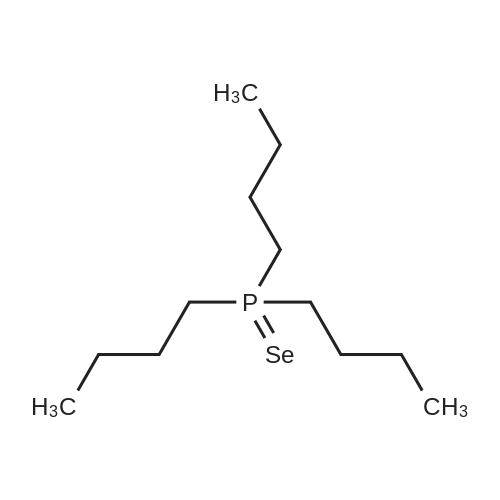
With Tergitol 15-S-7; zinc(II) oxide;phosphoric acid; In water; at 55 - 73℃; for 1h; |
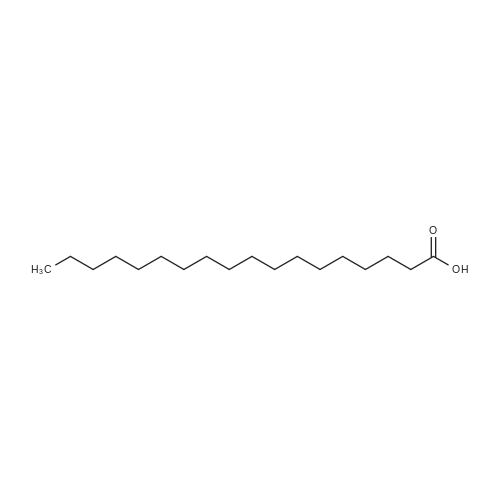
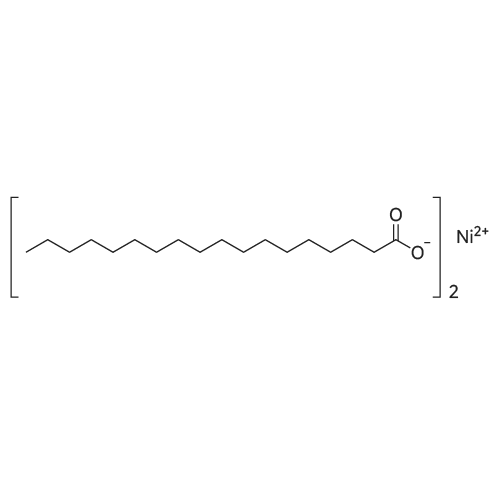
A molten stearic acid (29.7 grams, Industrene 7018) was added gradually to a stirred slurry of zinc oxide (4.86 grams), phosphoric acid (0.13 gram, 85% solution in water), Tergitol 15-S-7 (0.8 gram), and water (65 grams) at 65 C. The mixture was stirred at 55-73 C. for one hour to give a quantitative yield of zinc stearate as shown by the infrared spectrum of the final product. Example 10. A molten stearic acid (29.5 grams, Hystrene 7018) was added gradually to a stirred mixture of zinc oxide (4.9 grams), phosphoric acid (0.1 gram), Tergitol 15-S-7 (0.2 gram), and water (66 grams) at 65 C. The reaction mixture was stirred at 60-70 C. for one hour, cooled to 25 C., filtered, and dried to afford 32.4 grams (98.8% yield) of zinc stearate white powder having 0.1% water, 14.3% ash, 0.44% by weight of free fatty acid, and a melting point of 126 C. |
| 50% |
With Tergitol 15-S-7; zinc(II) oxide; hydroxypropyl methylcellulose; In water; at 55 - 70℃; for 1h; |
A molten stearic acid (22 grams, Industrene 7018) was added gradually to a stirred slurry comprising zinc oxide (3.6 grams), Methocel F50 (0.2 gram), Tergitol 15-S-7 (0.2 gram), and water (70 grams) at 55 C. The mixture was stirred at 65-70 C. for 1 hour to produce a yield of zinc stearate of about 50% by its infrared spectrum. |
| 10% |
With Tergitol 15-S-7; zinc(II) oxide; In water; at 55 - 75℃; for 3h; |
Zinc oxide (6.6 grams), Tergitol 15-S-7 (2 grams), and water (70 grams) were placed in a beaker and heated to 55 C. with stirring to form a slurry mixture. Stearic acid (43 grams of Industrene 7018, titer=58-62 C.) was heated to about 75 C. and the resulting molten stearic acid was added gradually to the slurry mixture. The reaction mixture was stirred at 60-65 C. for three hours. A sample was drawn from the reaction mixture, dried, and analyzed by infrared spectrum to show about a 10% yield of zinc stearate. |
|
With zinc(II) oxide; at 124℃; under 5414.51 Torr;Neat (no solvent);Product distribution / selectivity; |
Example 1 Molten stearic acid at ~170F (77C) was pumped from storage through an in-line continuous mixer. Solid zinc oxide at ambient temperature was metered continuously from a super sack into the same continuous mixer at a rate so as to achieve approximate stoichiometric ratios of the two reactants. The slurry of zinc oxide in stearic acid exiting the mixer was then continuously preheated to ~255F (124C) in a shell-and-tube heat exchanger prior to entering the high shear, continuous reactor. A temperature rise of ~30F (17C) was noted in the high shear reactor. The continuous discharge from the reactor was routed through a standard pipe to a receiver. A backpressure of 90 psig was maintained on the pipe and reactor during the reaction. The reaction mass discharged continuously from the pipe into a receiver where byproduct steam was flashed to the vent system. Total residence time required for the reaction, i.e. from the high shear reactor through the pipe to the receiver, was ?40 seconds. The combined feed rate of zinc oxide solids and stearic acid was maintained at 3,000 pounds per hour (1,361 Kg/hr) until 950 pounds (431 Kg) of ZnO and 6,515 pounds (2,892 Kg) of stearic acid were fed and the resulting zinc stearate product was collected in the receiver. The molten zinc stearate product had the following analysis: clear appearance, ash content 13.5%, free fatty acid 0.12%, water content 0.22%, and melt point of 250F (121C). The molten product was maintained under a nitrogen blanket to preserve color prior to being prilled (sprayed) to obtain the desired particle size, which in one embodiment, is about less than 100 microns. The characteristics of the zinc stearate were as shown in Table I. [Table I] ParameterPrior ArtNew processYellowness Index13.2-0.1% Transmittance34.673.3Color (Gardner Index)4.1 G< 1 G. The Prior Art product was made in a batch mode by introducing stearic acid into a 1000 gallon stainless steel reactor having four longitudinally-directed spaced-apart baffles positioned abot a periphery of the reactor which was further equipped with two vertically spaced-apart rotating impellers, each having three angled paddles, on a shaft rotating at about 88 revolutions per minute, tip speed 481 ft/min (147 m/min), the reactor having an internal working pressure of 50 psig maximum at 350 F (177 C). After adding the requisite amount of stearic acid, an equimolar amount of solid zinc oxide is added into the reactor with agitation followed by heating the reactor to about 250 F (121 C). Allow the reaction to proceed for about 30 min followed by reducing the pressure followed by a nitrogen purge and collection of zinc stearate from the reactor. |
|
With carbon dioxide; ammonia; zinc(II) oxide; In water; at 65 - 100℃;pH 9.27 - 11.56; |
Bubble 63 grams of ammonia gas and 29 grams of carbon dioxide gas into 386 grams of water, then add 65 grams of zinc oxide into the solution. Stir well and the zinc oxide completely dissolves into the solution. Analysis shows that the solution contains 9.56 wt. % zinc with a pH of 11.56. This solution is called solution A. 450 grams of stearic acid is added to solution A and stirred well. The stearic acid completely dissolves in the solution. Heat the solution in a laboratory reactor with agitation and a water cooled condenser to recover the evaporated ammonia and carbon dioxide. The solution boils at around 65 C. and as it is boiling, white gelatinous precipitates form. As water level drops, 1,500 ml more of water is added to the mixture. When most of the ammonia is boiled out the temperature rises to 100 C. and the pH drops to 9.27. Cool the mixture to 25 C. to precipitate other slightly soluble organic zinc salts which may be in the solution, then filter and predry the white precipitate at 90 C. for 2 hours to remove most of the water. Then further dry the precipitate at 105 C. for one hour, and mill it to obtain zinc stearate A.Using the conventional fusion method, zinc stearate B was prepared. Following are properties of both samples: Zinc Stearate A Zinc Stearate B Zinc (wt. %) 10.17 10.22 Acid Value (mg KOH/g) 18.43 96 Melting point ( C.) 112 116 pH 8.09 6.35 Bulk density (g/cm3) 0.36 0.45 Other water insoluble or sparingly soluble organic zinc salts or mixtures of organic zinc salts like zinc resinate, zinc laurate, zinc oleate, and zinc salts of coconut fatty acids can also be made according to the method described for the preparation of zinc stearate A. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping