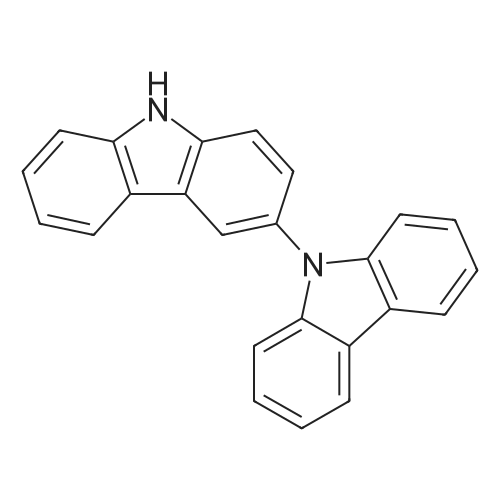
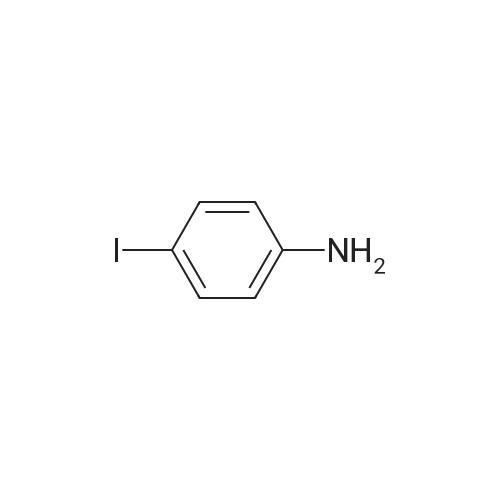
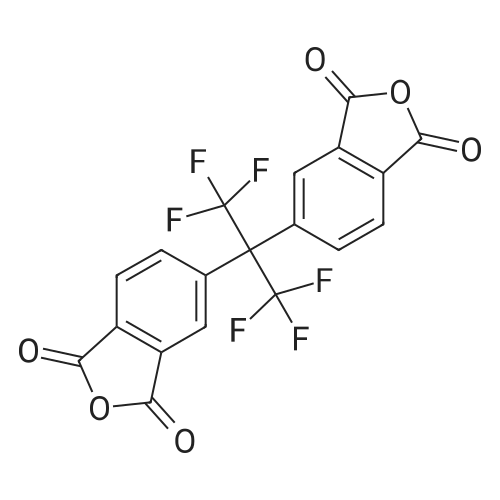
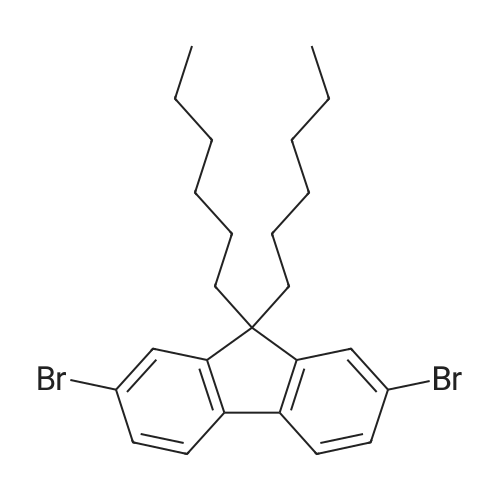
| 98% |
With palladium 10% on activated carbon; hydrazine hydrate In ethanol at 80℃; |
|
| 97.82% |
With iron; acetic acid at 20℃; for 16h; Cooling with ice; |
1 Synthesis of compound 2
In a 200mL single-neck flask, add compound 1 (4.00g, 13.87mmol) and 60mL glacial acetic acid successively, cool in an ice-water bath, add reduced iron powder (3.85g, 69.35mmol) in batches, magnetically stir for 10min, transfer to room temperature and continue stirring reaction for 16h. Stop the reaction, pour the reaction liquid into 150mL ice water, solids will precipitate out, filter with suction, and wash with distilled water. The solid was dissolved in DCM, filtered, and the remaining liquid was distilled under reduced pressure to remove the volatile solvent to obtain 3.50 g of viscous liquid (yield: 97.82%). |
| 96% |
With palladium on activated charcoal; hydrazine hydrate In ethanol for 4h; Reflux; |
|
| 95% |
With tin(ll) chloride In ethanol for 6h; Heating; |
|
| 95% |
With palladium on activated charcoal; hydrazine hydrate In ethanol at 90℃; for 12h; Inert atmosphere; |
|
| 95% |
With palladium 10% on activated carbon; hydrazine hydrate In ethanol at 80℃; for 24h; Inert atmosphere; |
2.2 (2) Synthesis of Intermediate 4- (9H-carbazol-9-yl) aniline
After 2.883g (0.01mol) 9- (4-nitrophenyl) -9H-carbazole was added to three 500ml flask, 300ml of absolute ethanol, a magnetic stirrer and argon gas, was heated to 80 deg.] C oil bath, was added 10% wt after the palladium on carbon 0.1g, was added and 10ml of hydrazine hydrate, the reaction was refluxed for 24h, the reaction solution was suction filtered and the filtrate was cold crystallization, the resulting cake was suction filtered and dried in vacuo 80 24h, to give the product 2.454g, 95% yield. This intermediate structure is as follows |
| 95% |
With sodium tetrahydroborate In ethanol at 20℃; for 2h; |
|
| 94% |
With palladium 10% on activated carbon; hydrazine hydrate In ethanol for 4h; Reflux; |
2.2.2. Synthesis of N-(4-aminophenyl) carbazole (2)
To a refluxed solution of compound 1 (1.44 g, 5.00 mol)and Pd/C (0.0720 g) in ethanol (10 mL) was added dropwise hydrazine monohydrate. The mixture was refluxedfor 4 h and cooled to room temperature. Pd/C was removedby filtration through Celite and the filtrate was concentrated to give compound 2 as a clear viscous liquid(1.24 g) in 94 % yield. IR (KBr): 3460, 3379 cm1 (NH2stretch). 1H NMR (500 MHz, DMSO-d6, d, ppm): 8.19(d, 2H), 7.40 (t, 2H), 7.27-7.21 (m, 4H), 7.18 (d, 2H), 6.81(d, 2H), 5.35 (s, 2H). |
| 90% |
With tin(II) chloride dihdyrate In ethanol at 70℃; for 1.66667h; Inert atmosphere; |
|
| 90% |
With tin(II) chloride dihdyrate In ethanol at 80℃; for 1.83333h; Inert atmosphere; |
1
Weigh 6.00g product NPC (22mmol),Add 30.2g of tin(II) chloride dihydrate (134mmol) and 35ml of ethanol into the mixed system, heat to 80°C and reflux,React for 110 min under nitrogen protection. After removing the solvent by distillation under reduced pressure,Place the product in a cold water bath,Add 25-30% NaOH solution under vigorous stirring,After the system becomes milky white liquid, the product is extracted with ether,After washing with saturated brine and fully drying with anhydrous magnesium sulfate,Filter and spin-evaporate to remove the solvent,Dry to get a white viscous productN-(4-anilino)carbazole(CzPA) 5.12g, the yield is 90% |
| 90% |
Stage #1: 9-(4-nitrophenyl)-9H-carbazole With palladium 10% on activated carbon; hydrazine hydrate In ethanol at 20℃; for 1h; Inert atmosphere;
Stage #2: at 90℃; for 12h; |
Step 2: synthesis of 9-(4-aminophenyl)-9H-carbazole
0.08 mol (23.05 g) of 9-(4-nitrophenyl)-9H-carbazole and 0.016mol (1.7 g) of 10% Pd/C were dispersed in 100mL of ethanol in a 250mL two-neck round-bottom flask equipped with a stirring hot plate, a condenser and an inlet of inert gas. The reaction mixture was stirred under nitrogen for 1 h at room temperature before 0.8mol (40 g) of hydrazine monohydrate was slowly added. The reaction mixture was refluxed at 90 C for 12 h. The reaction mixture was filtered to get rid of Pd/C, and then ethanol was removed by a vacuum evaporator to leave the white gel product (yield 90%) (Scheme 1). |
| 85% |
With tin(II) chloride dihdyrate In ethyl [2]alcohol for 24h; Reflux; |
2 Synthesis of APCB
2.3.2 Synthesis of N-(4-aminophenyl)carbazole (APCB) To a solution of 5.76 g (0.02 mol) of NPCB in 40 ml ethanol, 15.8 g (0.07 mol) of SnCl2·2H2O was added. After refluxing for 24 h, the reaction mixture was condensed under reduced pressure to distill off most of the ethanol followed by neutralization with 40 wt% aqueous NaOH solution until the mixture became alkaline. The resulting mixture was then extracted with toluene, dried over anhydrous magnesium sulfate, and evaporated under reduced pressure to get 4.5 g (85% in yield) of yellowish syrup. FT-IR (KBr, cm-1): 3375, 3459 (N-H stretch). 1H NMR (DMSO-d6, 400 MHz), δ (ppm): 8.19 (d, 2H, J = 7.67 Hz), 7.40 (t, 2H, J = 7.66 Hz), 7.26∼7.17 (m, 6H), 6.79 (d, 2H, J = 8.61 Hz), 5.43 (s, 2H). |
| 85% |
With 5%-palladium/activated carbon; hydrazine hydrate In ethanol at 80℃; |
|
| 80% |
With palladium 10% on activated carbon; hydrazine hydrate In ethanol for 10h; Reflux; |
4-(9H-Carbazol-9-yl)aniline (4b), compound 3b (0.09 mol), 10% Pd/C (0.103 g) and ethanol (20 mL) were added to a 50-mL round-bottom flask equipped with a stirring bar. The reaction mixture was heated under reflux. Hydrazine monohydrate (3 mL) was added slowly to the mixture, and then the solution was stirred under reflux for 10 h. The solution was cooled to room temperature, filtered to remove Pd/C, and then concentrated to afford a light brown viscous liquid in a yield of 80%. 1H NMR (500 MHz, CDCl3): δ 8.15 (d, J = 8.0 Hz, 2H), 7.41 (t, J = 7.5 Hz, 2H), 7.34-7.26 (m, 6H), 6.88- 6.87 (m, 2H), 3.83 (s, 2H). 4-(3,6-Dibromo-9H-carbazol-9-yl)aniline (4c), compound 4c was synthesized according the method used to prepare 4a with 3c instead of 3a to afford a white powder in a yield of 92%. 1H NMR (500 MHz, CD3CN): δ 8.34 (d, J = 2.0 Hz, 2H), 7.55 (d, J = 9.0, 2.0 Hz, 2H), 7.25-7.23 (m, 4H), 6.92-6.90 (m, 2H), 4.74 (-NH2, 2H). |
| 79.8% |
With palladium on activated carbon; hydrazine hydrate In ethanol at 20 - 60℃; |
3.2.2. Synthesis of N-(4-aminophenyl)carbazole (II)
The dried nitro compound N-(4-nitrophenyl)carbazole (monomer I in Scheme 1, 8.6 g, 30 mmol), along with the palladium carbon hydrogenation catalyst (Pd/C, 0.7 g, 10 wt%), and hydrazine hydrate (24 mL) were carefully weighed or measured. Monomer I and the Pd/C hydrogenation catalyst were then sequentially added to a 250 mL three-necked flask that was equipped with a stir bar. After the addition of ethanol (50 mL), this mixture was stirred at room temperature for 5-10 min until the reaction system was uniformly mixed, and it was then heated to 60 °C. After the temperature had become stable, the hydrazine hydrate was added dropwise to the reaction system through a constant pressure funnel at a controlled rate of 4-5 s per drop, so that the hydrazine hydrate was completely added within 2 h, and the reaction proceeded at a constant temperature for 12 h. After the reaction reached completion, Pd/C was removed via filtration through a sand core funnel. The filtrate was collected and the ethanol and hydrazine hydrate were distilled off via rotary evaporation, and the residual liquid was taken up in water to give a white precipitate. After washing with distilled water for 4-5 times, this precipitate was dried in a vacuum oven at 60 °C for 12 h. The crude product N-(4-aminophenyl)carbazole was thus obtained with a dried weight of 6.2 g, a theoretical yield of 7.7 g, and a percentage yield of 79.8%. The crude product was recrystallized from absolute ethanol under a nitrogen atmosphere at 85 °C, and the resultant crystals were dried in a vacuum oven at 60 °C for 12 h, thus yielding 5.2 g of the purified product as brown crystals.Monomer (II), brown crystals, the yield was 79.8%. Tm: 105-107 °C,FT-IR (KBr) cm-1: 3460, 3379 (NH2 stretch). 1H NMR (DMSO-d6,500 MHz, δ, ppm): 8.19 (2H, d, J 7.7 Hz), 7.39 (2H, d, J 8.3 Hz),7.24 (4H, t, J 7.5 Hz), 7.18 (2H, d, J 7.2 Hz), 6.80 (2H, d,J 7.0 Hz), 5.43 (2H, s). Found: C, 83.3; H, 5.5; N, 5.7%; molecularformula C18H14N2 requires C, 83.7; H, 5.5; N, 5.8%. |
| 78.3% |
With hydrazine hydrate; palladium on activated charcoal In ethanol for 10h; Reflux; Inert atmosphere; |
3.2; 4.2
2) Add 5.2 g of N-p-nitrophenyl oxazole to a 250 mL three-necked flask.0.2g Pd/C, 50mL ethanol mixed,Heating under reflux in an N2 atmosphere,Slowly add 4 ml of hydrazine hydrate and heat to reflux for 10 h at reflux temperature.Cool to room temperature, filter, and steam;Recrystallization from petroleum ether gives N-p-aminophenyl oxazole,Yield 78.3%; |
| 65% |
With tin(ll) chloride In ethanol at 70℃; for 4.5h; |
|
| 48% |
With hydrogenchloride; tin In methanol for 2h; Heating; |
|
| 43% |
With sodium tetrahydroborate; tin(ll) chloride In tetrahydrofuran; methanol at 0 - 20℃; for 3h; Inert atmosphere; |
8 Preparation of Intermediate I-8
Under a nitrogen atmosphere, I-7 (30 g, 104.1 mmol) was dissolved in 0.3 L of dm tetrahydrofuran (THF), then 0.3 L of methanol was added and the mixture was cooled to 0 ° C.After adding sodium borohydride (39.4 g, 1,041 mmol), tin (II) chloride (98.7 g, 520.5 mmol) was added and the mixture was reacted at room temperature for 3 hours. After completion of the reaction, water was added to the reaction solution, and the mixture was extracted with ethyl acetate (EA). After removal of moisture with anhydrous MgSO 4, filtration and concentration under reduced pressure.The residue thus obtained was separated and purified by flash column chromatography to obtain I-6 (11.6 g, 43%). |
|
With hydrogenchloride; tin |
|
|
With hydrazine hydrate In ethanol for 9h; Heating; |
|
|
With tin(ll) chloride |
|
|
Stage #1: 9-(4-nitrophenyl)-9H-carbazole With hydrogenchloride; tin In methanol; water for 16h; Heating / reflux;
Stage #2: With sodium hydrogencarbonate In methanol; water at 20℃; |
1.b
A suspension of compound 2 (9.20 g, 31.9 mmol), tin granules (11.4 g, 95.7 mmol), hydrochloric acid (15.1 ml, 153.0 mmol, 37%) in methanol (200 ml) was heated under reflux for 16 h. The reaction mixture was cooled to room temperature, filtered, neutralized with excess sodium bicarbonate solution (aqueous) and the organic layer extracted into chloroform (3 x 150 ml) and the combined extracts dried (MgSO4), filtered and the solvent removed in vacuo providing a viscous oil. The crude product was purified by columned chromatography [silica gel, eluted with 2:1 hexanes:ethyl acetate, containing 1 % methanol] providing a colorless oil. 1H NMR (500MHz, CDCI3) δ/ppm: 8.19 (dt, 2H, aromatic), 7.45 (m, 2H, aromatic), 7.38 (dt, 2H, aromatic), 7.30-7.34 (m, 4H, aromatic), 6.87 (dt, 2H, aromatic), 3.84 (s, 2H, NH2). |
|
With tin(ll) chloride In ethanol Reflux; |
|
|
With palladium 10% on activated carbon; hydrazine hydrate In ethanol for 24h; Reflux; |
|
|
With palladium on activated charcoal; hydrazine hydrate In ethanol for 6h; Reflux; |
|
|
With hydrazine hydrate |
|
|
With palladium on activated charcoal; hydrazine hydrate In ethanol |
|
|
With palladium on activated charcoal; hydrazine In ethanol |
|
|
With palladium on activated charcoal; hydrogen In N,N-dimethyl-formamide at 50℃; |
|
|
With palladium on activated charcoal; hydrazine In ethanol Reflux; |
|
|
With sodium tetrahydroborate; 1% Pd on activated carbon In methanol at 20℃; for 2.5h; Cooling with ice; |
2.3. Synthesis of 2-(9H-Carbazol-9-yl) Aniline and 4-(9H-Carbazol-9-yl)Aniline
General procedure: 9-(2-nitrophenyl)-9H-carbazole/9-(4-nitrophenyl)-9H-carbazole(1 eq)was dissolved in 5 mLof dry methanol and catalytic amount of 1%Pd/C was added in ice-cold condition under vigorous stirring. NaBH4(2 eq.) was added in portion-wise over a period of 30 min into reactionsolution at ice-cold condition. The reaction mixture was brought toroom temperature and stirred for another 2 h. Then Pd/C was filteredoff and solvent was evaporated using rota-vapour. The residue was extractedwith ethyl acetate and the extract was washed with brine solutionand dried over Na2SO4. Finally, the evaporation of ethyl acetateusing rota-vapour producedwhite coloured solid as product. Yield: 68%. |
|
With palladium on activated charcoal; hydrazine In ethanol for 18h; Reflux; |
|
|
With tin(ll) chloride In ethanol for 3h; Inert atmosphere; |
|
|
With palladium on activated charcoal; hydrazine hydrate |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping