| 93.1% |
With triethylamine; at 0 - 20℃; for 5h; |
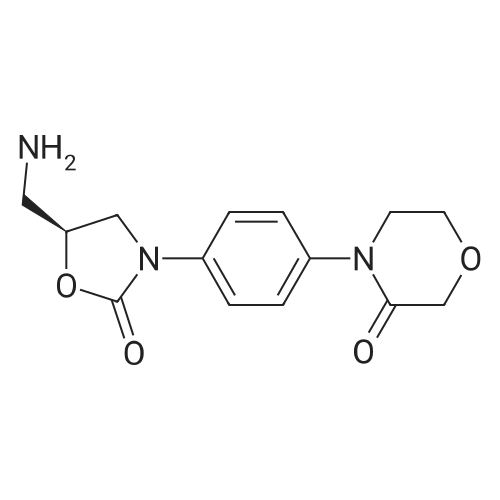
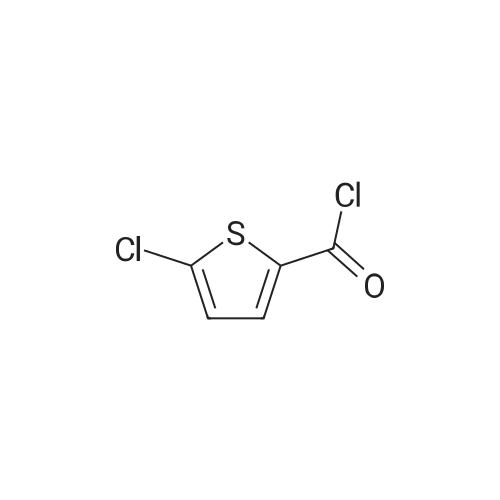
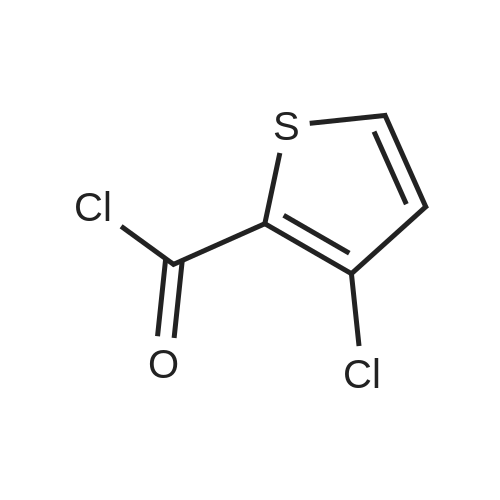
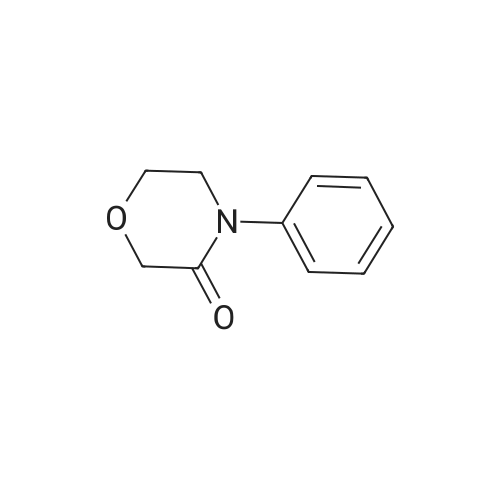
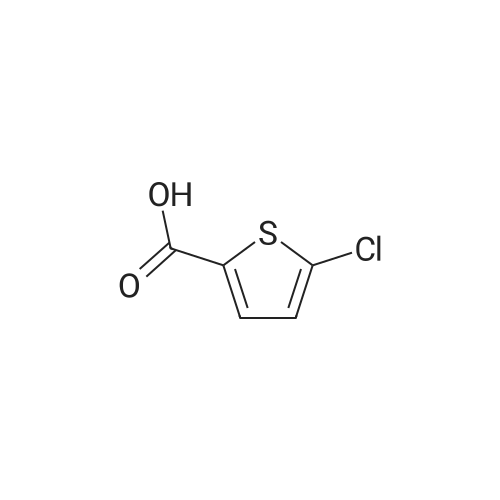
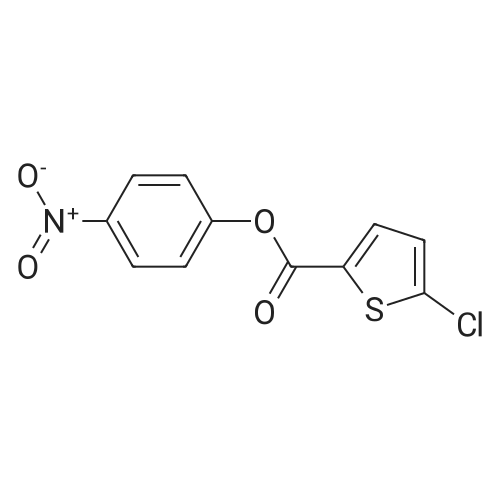
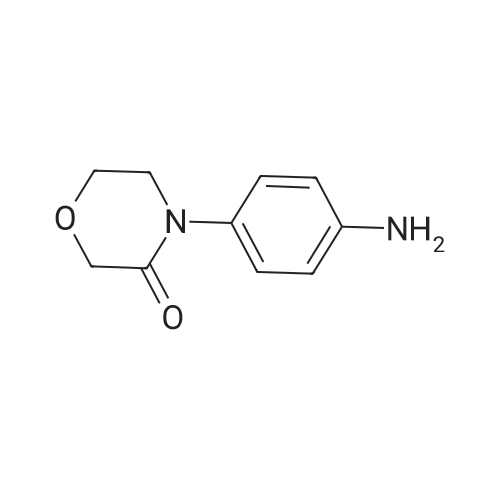
27.5 g (94.4 mmol) of the key intermediate of the synthetic rivaroxaban shown in Formula I prepared in Example 5 and 150 mL of triethylamine were added to the reaction flask, and the solution was added dropwise at 0 C.5-Chloro-2-acylchlorothiophene 22.1 g (122.1 mmol), dripped, warmed to room temperature for 5 hours, added 300 mL of water, stirred for 30 minutes, extracted three times with dichloromethane (450 mL × 3), and the organic layer was saturated with salt It was washed twice with water (400 ml × 2), and the organic layers were combined, dried over anhydrous sodium sulfate, filtered, and the solvent was distilled off under reduced pressure to obtain 38.3 g of a white solid with a yield of 93.1%. |
| 92.8% |
With pyridine; In dichloromethane; at 30 - 40℃; for 5h; |
The reaction flask was charged with 32.9 g of the compound 7 (0.1 mol) prepared in Example 5-2, 300 mL of dichloromethane, 15.8 g (0.2 mol) of pyridine, 25.3 g of compound 8 (0.14 mol), the mixture was stirred at 30 C to 40 C for 5 hours. The reaction was monitored by TLC. The solvent was removed by distillation under reduced pressure. The residue was washed with 200 mL of purified water, and then recrystallized from a mixture of methylene chloride and ethyl acetate in a volume ratio of 5: 1 and dried under reduced pressure to obtain rivaroxaban 40.4 g, a molar yield of 92.8%, an HPLC chemical purity of 99.8% and an optical purity of 100.0%. |
| 92.9% |
With pyridine; In dichloromethane; at 30 - 40℃; for 5h; |
S6, (S)-3-[4-(3-oxo-4-morpholino)phenyl]-5-aminomethyl-1,3-oxazolidin-2-one obtained in the step S5 ( 36.2g, 0.11mol),pyridine (17.4 g, 0.22 mol)and 5-chloro-2-acylchlorothiophene (27.8 g, 0.15 mol) add to dichloromethane (330ml), stir evenly, heat to 30 ~ 40 C, reaction for 5h,The solvent was evaporated under reduced pressure and the obtained residue was washed with purified waterThe crude product was obtained as a white solid. The crude solid was obtained from methylene chloride: ethyl acetate (5:1) (1000 ml), and the mixture was heated to 40 C to dissolve all the solids, and the temperature was lowered to 0 C for 2 h, and the obtained solid was dried at 30~ 40 C for 2 h under reduced pressure obtained as a white solid (44.45 g, 92.9%); mp 229-231 C, [alpha]D20-39.3(c 0.3,DMSO) purity 99.8% (HPLC normalization). Chiral purity was 99.9% (HPLC normalization). |
| 91% |
|
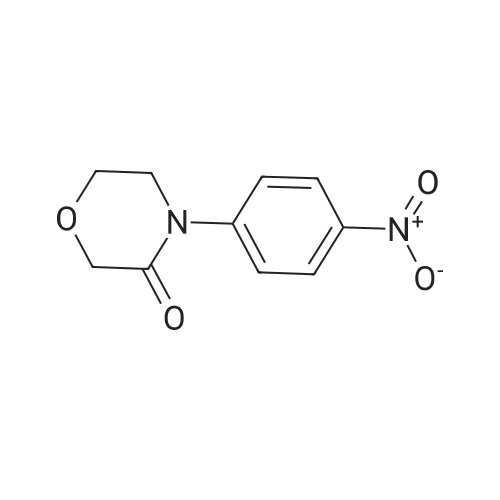
1.5 kg of 4- (4- (5- (aminomethyl) -2-oxyloxazolidin-3-yl) phenyl) morpholino-3-one was put into a 50 L reaction flask,Add 10kg acetone, stirring at 20 ~ 25 10min,Sodium carbonate solution (1 kg of sodium carbonate dissolved in 10 kg of water) was added with stirring,After completion of the addition, the mixture was cooled to 5 to 10 C and a toluene solution of 5-chloro-thiophene-2-carboxylic acid chloride was added dropwise,Containing 5 kg of 5-chloro-thiophene-2-carboxylic acid chloride,Upon completion of the addition,4 kg of acetone was added,After the completion of stirring at room temperature for 60min,The mixture was stirred at 50 to 55 C for 2 hours,Reaction completed,Cooled to 15 C or less,After stirring for 1 hour,filter,The filter cake was washed with 10 kg of water,10 kg of acetone,Drained,Blast-drying for 10 hours gave 1.83 kg of rivaroxaban in a yield of 91%. |
| 90% |
With triethylamine; at 0 - 5℃; for 3h; |
(S)-4-(4-(5-(aminomethyl)-2-oxo-oxazolidine-3-yl) phenyl) morpholinyl-3-ketone solution (0.20mol) add triethylamine (22.2g, 0 . 22mol), cooled to 5 C, dropwise 5-chloro-2-acyl chloride thiophene (36.2g, 0 . 20mol), control the feed liquid temperature is lower than 5 C, 1 hour internal dropping end. Maintain the temperature 0-5 C reaction 2 hours to the reaction is complete. Evaporate solvent 300 ml, solid precipitate in liquid, cooling to 0-5 C stirring 3 hours, filtering, washing, ice-methanol washing, drying the white solid obtained 78.3g, yield 90.0%. |
| 90% |
With sodium carbonate; In water; at 50 - 55℃; for 1h; |
8.25g (0.078mol) sodium carbonate, 90 mL of water, 24g (0.06mol) of compound 8, and 180mL of acetone pointsrespectively into 500mL three-necked flask, the system was cooled to 8-10 , 22.8g (0.12mol) 5 - chloro-thiophene-2-carboxylic acid chloride anda mixed solution of 72mL of toluene was slowly dropwise added to the system dropwise, gradually warmed to 50-55 deg.] C the system was stirred for IH, the system then cooledto room temperature, filtered, the filter cake was dried in vacuo 80 24.6 g of product was obtained and HPLC was 99.64%.The crude product was recrystallized from 200 mL of acetic acidand filtered to give 22.1 g (yield 90%) of white crystalline powder, 99.90% HPLC, mp = 229.2-230.2 C, |
| 86.1% |
With triethylamine; In N,N-dimethyl-formamide; at 30 - 40℃; for 5h; |
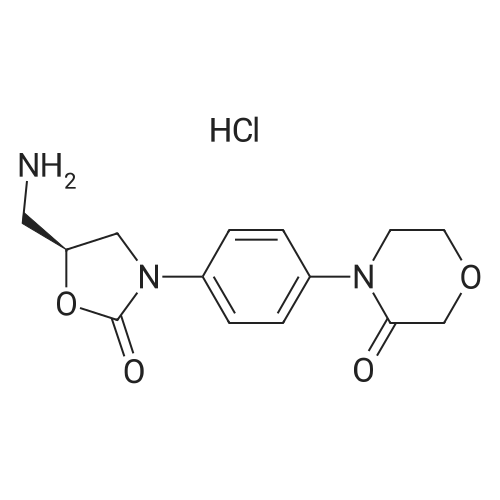
131.1 g (0.4 mol) of 4-{4-[(5S)-5-(aminomethyl)-2-oxo-1,3-oxazolidin-3-yl] prepared in Example 10 was added to the reaction flask. Phenyl}morpholin-3-one hydrochloride (Formula VIII), 500 ml of N,N-dimethylformamide, 50.6 g (0.5 mol) of triethylamine, 101.4 g (0.56 mol) of 5-chlorothiophene-2-carbonyl chloride,Stir well and react at 30C to 40C for 5 hours.TLC control (dichloromethane:methanol = 20:1, volume ratio) was completed. 500 ml of purified water was added and a large amount of solids precipitated.Stir for 2 hours, filter and filter cake washed with 200 ml purified water.Decompression drying, 150.1g of rivaroxaban product Molar yield 86.1%, HPLC chemical purity 99.8%,Optical purity 100.0% |
| 85.7% |
With triethylamine; In N,N-dimethyl-formamide; at 20 - 30℃; for 5h; |
The reaction flask was prepared in Example 5 was added 20.4g (0.07mol) 4- {4 - [(5S) -5- (aminomethyl) -2-oxo-1,3-oxazolidin-3-yl] phenyl} morpholin-3-one (formula VII), 200ml of N, N- dimethylformamide, 10g triethylamine, stir, a solution of 12.6g (0.07mol) at 20 to 30 2- formyl-5-chloro-thiophene chloride, dropwise, reaction was continued incubation at 20 deg.] C to 30 5 h, TLC in the control (dichloromethane: methanol = 20: 1, volume ratio) to complete the reaction, the reaction solution was poured into 500ml purified water, with a large amount of solid precipitates, stirred for 1 hour, filtered, the filter cake was washed with purified water 100ml, drying under reduced pressure, the product enrichment rivaroxaban 26.1g (0.060mol), molar yield 85.7%, HPLC purity 99.6% |
| 85.7% |
With triethylamine; In N,N-dimethyl-formamide; at 20 - 30℃; for 5h; |
The reaction flask was charged with 20.4 g (0.07 mol) of 4-{4-[(5S)-5-(aminomethyl)-2-oxo-1,3-oxazolidin-3-yl] prepared in Example 5. Phenyl}morpholin-3-one (formula VII),200 ml of N,N-dimethylformamide and 10 g of triethylamine were stirred well and 12.6 g (0.07 mol) of 2-chlorocarbonyl-5-chlorothiophene was added dropwise at 20C to 30C.After dripping, continue the incubation at 20C to 30C for 5 hours.TLC control (dichloromethane: methanol = 20:1, volume ratio) was completed. The reaction solution was poured into 500 ml of purified water, a large amount of solids precipitated, stirred for 1 hour, filtered, and the filter cake was washed with 100 ml of purified water. drying,26.4g (0.060mol) of derivaroxaban product with a molar yield of 85.7% and HPLC purity of 99.6% |
| 81% |
With triethylamine; In dichloromethane; toluene; at 0 - 20℃; for 0.5h; |
In a 250ml reaction flask as in formula II compound (10.56g, 36.25mmol), triethylamine (4.04g, 39.88mmol), in methylene chloride (100 mL) was stirred at 0 clear solution. Was added dropwise a toluene solution of Formula IV as 29% of the compound (23.6g, 38mmol), room temperature 0.5h. Saturated aqueous sodium bicarbonate (90mL) and n-heptane (200mL), filtration and drying pale yellow rivaroxaban crude, crystalline water DMF- benefit rivaroxaban product (12.59g, 81.0%). |
| 81% |
With triethylamine; In dichloromethane; toluene; at 0 - 20℃; for 0.5h; |
In 250mL reaction flask as shown in Formula II compound (10.56g, 36.25mmol), Triethylamine (4.04g, 39.88mmol), dichloromethane (100mL) at 0 C stirred solution clear. Dropping As a toluene solution of Formula IV 29% of the compound (23.6g, 38mmol), room temperature 0.5h. Saturated aqueous sodium bicarbonate (90mL) and n-heptane (200mL), filtered and dried to give a crude product of rivaroxaban, which was crystallized from DMF-water to yield a rivaroxaban product (12. 59 g, 81.0%). ESI-MS (m / z): 436 [M + H] +, 438 [M + H] + |
| 81% |
With triethylamine; In dichloromethane; toluene; at 0 - 20℃; for 0.5h; |
To a 250 ml reaction flask a compound of formula II (10. 56 g, 36.25 mmol), triethylamine (4. 04 g, 39.88 mmol) and dichloromethane (100 mL) were added and stirred at 0C. 29% toluene solution of the compound of formula IV was added dropwise (containing 38 mmol of the compound of formula IV,The total mass of toluene solution 23. 6g,The percentage is the amount of the compound represented by formula IV as a percentage of the total mass of the toluene solution) and reacted at room temperature for 0.5 h. A saturated aqueous solution of sodium bicarbonate (90 mL) and n-heptane (200 mL) were added, filtered and dried to obtain crude light yellow rivaroxaban. The crude product was crystallized by DMF-water to obtain rivastane products (12.59 g,Yield 81. 0%, HPLC purity 99. 91%). |
| 67% |
With triethylamine; In toluene; at 30℃; for 1.5h; |
10.5 g of the amine prepared according to Example 26 were suspended in 200 ml of dichloromethane and then 5.4 ml of triethylamine dissolved in 50 ml of dichloromethane were added. This was followed by addition of 14.4 ml of a solution of 5-chlorothiophene-2- carboxylic acid chloride in toluene (2.46 M) and 25 ml of dichloromethane. The reaction mixture was stirred and heated at boiling for 1.5 hours and then slowly cooled below 30C. The separated product was filtered off, washed with dichloromethane (15 ml) and ethanol (2 x 15 ml). The crude product was crystallized from a mixture of acetic acid (20 ml) and ethanol (200 ml). 10.5 g (yield 67%) of rivaroxaban was obtained in the form of an off-white powder with the melt, point of 228-230C, HPLC 99.97%, content of the (R)-isomer below 0.03%. The NMR and MS spectra were in accordance with Example 28. |
| 65.6 g |
With potassium carbonate; at 10 - 15℃; for 4h; |
Aqueous reaction mass of example 4 containing 4-[4-((S)-5-aminomethyl]-2- oxooxazolidin-3-yl)phenyl]morpholine-3-one (VII) is chilled up to 10- 15C . Charged 49.16 gm (0.3562 moles) potassium carbonate at 10- 15C. The solution of 5-chloro thiophene-2 -carbonyl chloride (VIII) 51.6 gm (0.2850 moles) in 200ml dimethyl carbonate was charged to the reaction mixture at 10- 15C. Precipitated reaction mixture was stirred at 10-15C for 4 hours. The product was filtered and washed with water. The product was dried under vacuum at 65C. It is then further purified by methanol purification at reflux temperature followed by DMSO/MeOH purification to get pure |
|
With N-ethyl-N,N-diisopropylamine; In dichloromethane; at 0 - 30℃; for 2h; |
Charge Dichloromethane (250 ml) to reaction mass to obtain solution of compound (VII), which is gradually added at 0-5C to a pre-cooled solution of compound (II) (100 gms), Dichloromethane (700 ml) and Diisopropyl ethyl amine (50 ml). Stirr the reaction mass for 30 mins at same temperature followed by stirring at 25-30C for 30 mins. Cool the reaction mass and stir at 10-15C for 30 mins followed by stirring at 25- 30C for 30 mins. Filtered the solid and dried under vacuum at 45-50C for 5 hrs. |
| 3.7 g |
With pyridine; In dichloromethane; at 0 - 10℃; for 4h; |
(R-1) in dichloromethane was added 1.1 g (13.9 mmol) of pyridine, cooled to 0 C with an ice salt bath, 1.91 g of compound VII (5-chloro-thiophene-2-carboxylic acid chloride) was added dropwise with stirring In 20 ml of methylene chloride solution, the temperature was controlled at 10 C for 4 hours. The organic layer was separated and the organic layer was washed three times with water and dried over anhydrous magnesium sulfate. Distillation of dichloromethane to give a solid residue. To the solid residue was added 150 ml of ethanol, stirred at 50 C for 3 hours, cooled to 10 C, stirred for 1 hour, filtered and the filter cake was washed three times with ethanol. Vacuum drying profit rivaroxaban 3.7g, content of 99.6% (HPLC), The overall yield of compound II to rivaroxaban was 83.4%. |
|
With potassium carbonate; at 10℃; for 3h; |
In the aqueous phase obtained in Example 6,Added 2.54 g (18.4 mmol) of potassium carbonate,Stir and dissolve,Compound VI (1.665 g, 9.2 mmol) was added dropwise.Keep the temperature at 10 C for 3 hours,filter,Filter cake washed,80C blast dryingDerivata Shaban 3.0 grams,The three-step total yield is 75%.Rivaroxaban was confirmed as detected. |
| 26.1 g |
With triethylamine; In N,N-dimethyl-formamide; at 20 - 30℃; for 5h; |
20.4 g (0.07 mol) prepared in Example 5 was added to the reaction flask4-{4-[(5S)-5-(aminomethyl)-2-oxo-1,3-oxazolidin-3-yl]phenyl}morpholin-3-one (Formula VII),200ml of N,N-dimethylformamide, 10g of triethylamine, stir well,12.6 g (0.07 mol) of 2-chlorocarbonyl-5-chlorothiophene are added dropwise at 20C to 30C.After dripping, continue the incubation at 20C to 30C for 5 hours.TLC control (dichloromethane:methanol = 20:1, volume ratio) reacted completely.The reaction solution was poured into 500 ml of purified water, a large amount of solids precipitated, stirred for 1 hour, and filtered.The cake was washed with 100 ml of purified water and dried under reduced pressure to obtain 26.1 g (0.060 mol) of rivaroxaban product.Molar yield 85.7%, HPLC purity 99.6% |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science













 HazMat Fee +
HazMat Fee +

 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping