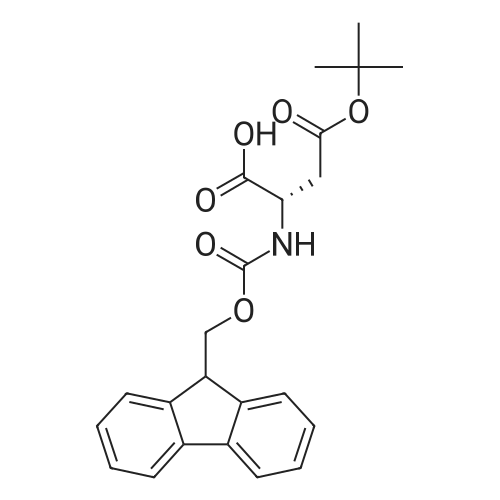
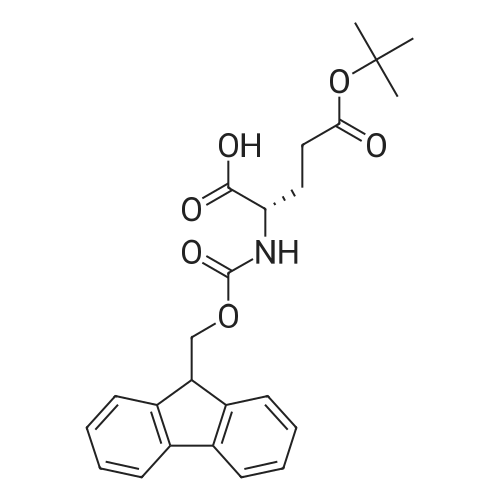
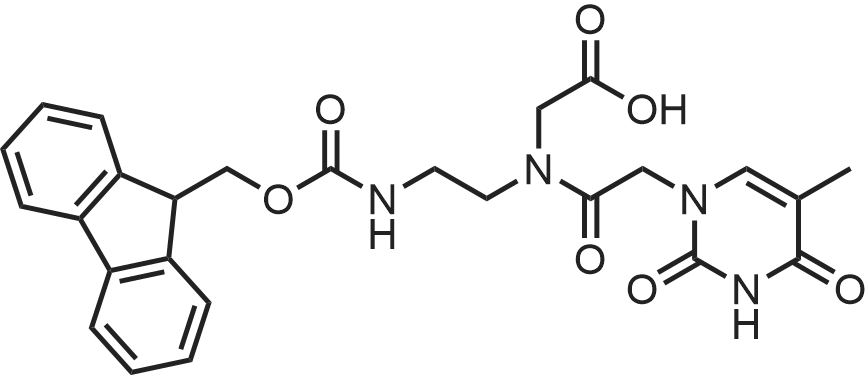
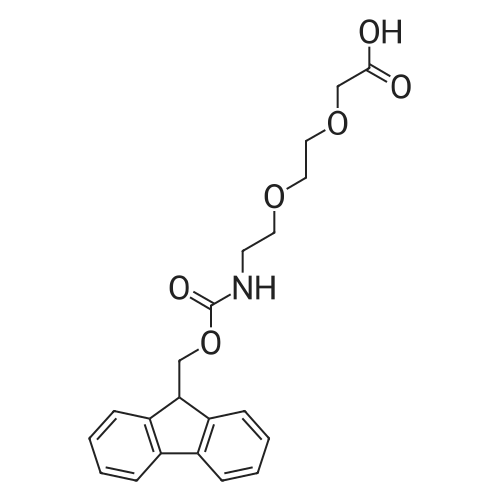
|
|
Example 14 30 mg of sodium palmitoyltaurine (PT) was used in place of 30 mg of SMT in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 22.4percent. |
|
|
Example 18 40 mg of PMT was used in place of 40 mg of SMT in Example 15 and treated similarly as in Example 15, to give a liposome composition of the 6-CF entrapment ratio of 6.8percent. |
|
|
Example 19 40 mg of ODS was used in place of 40 mg of SMT in Example 15 and treated similarly as in Example 15, to give a liposome composition of the 6-CF entrapment ratio of 6.5percent. |
|
|
Example 2 15 mg of SMT was used in place of 30 mg of SMT in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 34.9percent. |
|
|
Example 3 45 mg of SMT was used in place of 30 mg of SMT in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 39.4percent. |
|
|
Example 4 60 mg of SMT was used in place of 30 mg of SMT in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 46.3percent. |
|
|
Example 5 30 mg of sodium palmitoylmethyltaurine (PMT) was used in place of 30 mg of SMT in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 32.3percent |
|
|
Example 7 15 mg of ODS was used in place of 30 mg of ODS in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 24.1percent. |
|
|
Example 8 45 mg of ODS was used in place of 30 mg of ODS in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 38.3percent. |
|
|
Example 9 60 mg of ODS was used in place of 30 mg of ODS in Example 1 and treated similarly as in Example 1, to give a liposome composition of the 6-CF entrapment ratio of 40.1percent. |
|
|
EXAMPLE 2 The procedure of Example 1 was followed, but 15 mg of sulfatide was used in place of 30 mg of sulfatide, to yield a liposome composition entrapping 6-CF at a 23.3percent entrapment ratio and having a 42.4° C. phase transition temperature. |
|
|
EXAMPLE 13 The procedure of Example 11 was followed, but 15 mg of sulfatide was used in place of 30 mg of sulfatide, to yield a liposome composition entrapping 6-CF and CDDP at respective entrapment ratios of 16.5percent and 16.2percent and having a 42.4° C. phase transition temperature. |
|
|
EXAMPLE 15 The procedure of Example 11 was followed, but 45 mg of sulfatide was used in place of 30 mg of sulfatide, to yield a liposome composition entrapping 6-CF and CDDP at respective entrapment ratios of 17.6percent and 16.8percent and having a 41.7° C. phase transition temperature. |
|
|
EXAMPLE 17 The procedure of Example 11 was followed, but 210 mg of DPPC and 90 mg of DSPC were used in place of 270 mg of DPPC and 30 mg of DSPC, to yield a liposome composition entrapping 6-CF and CDDP at respective entrapment ratios of 21.7percent and 18.1percent and having a 44.5° C. phase transition temperature. |
|
|
EXAMPLE 3 The procedure of Example 1 was followed, but 45 mg of sulfatide was used in place of 30 mg of sulfatide, to yield a liposome composition entrapping 6-CF at a 18.9percent incorporation ratio and having a 42.4° C. phase transition temperature. |
|
|
EXAMPLE 2 The procedure of Example 1 was followed using a chloroform solution containing 1percent of a 7:3 (w/w) mixture of dipalmitoylphosphatidylcholine and distearoylphosphatidylcholine and a processing temperature of 60° C. to give liposomes with a 6-CF trap ratio of 32.2percent. |
|
|
Using the thus-obtained MLV dispersion and following the procedure of Example 1, there was obtained liposomes with a 6-CF trap ratio of 21.3percent. |
|
|
EXAMPLE 3 The procedure of Example 1 was followed using a chloroform solution containing 1percent of distearoylphosphatidylcholine and a processing temperature of 70° C. to give liposomes with a 6-CF trap ratio of 37.7percent. |
|
|
To a 30-ml portion of MLV as obtained by combining such batches was added 10 ml of ethyl ether and the mixture was processed in the same manner as in Example 4 to give liposomes with an improved 6-CF trap ratio. |
|
|
EXAMPLE 7 An SUV obtained by comminuting the MLV produced in Example 4 by means of a probe-type ultrasonic shaker was used and processed in the same manner as in Example 4 to give liposomes with an improved 6-CF trap ratio. |
|
|
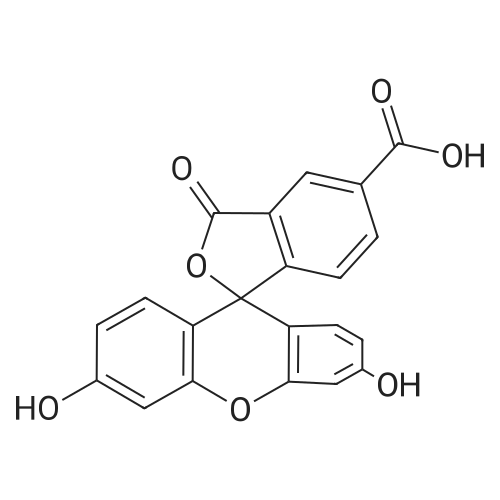
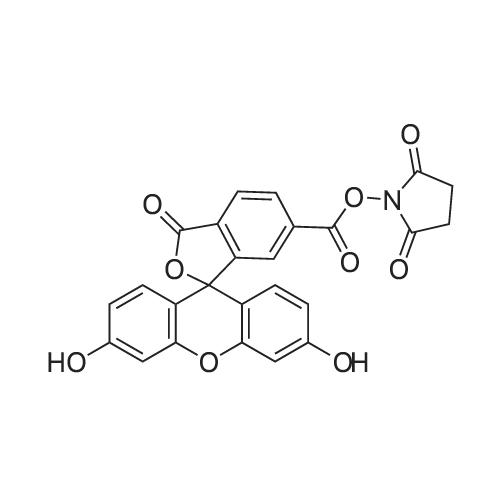
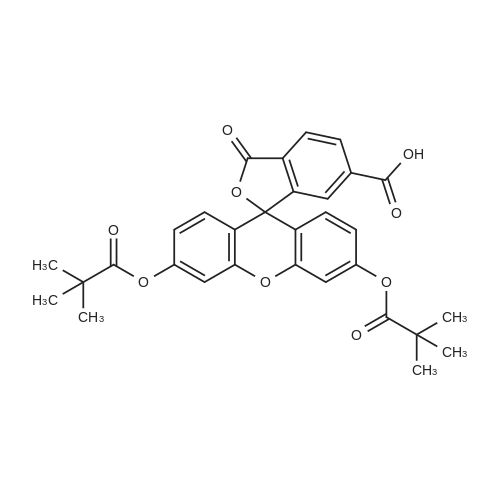
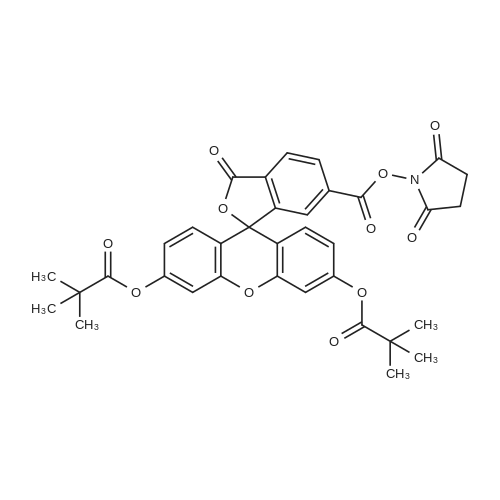
The preferred fluorochromes for use in the fluorochrome-protein conjugates are selected from the group consisting of...fluorescein-6-isothiocyanate,5-carboxyfluorescein, succinimidyl ester,6-carboxyfluorescein, succinimidyl ester,5-(and-6)-carboxyfluorescein, succinimidyl ester,... |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping