| 92.14% |
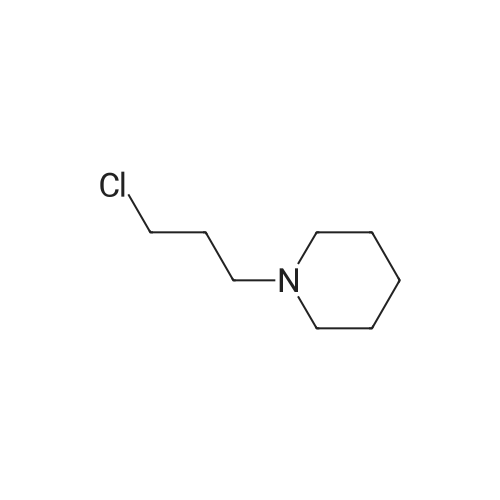
Stage #1: N-(3-chloropropyl)-piperidine With ethyl bromide; iodine; magnesium In tetrahydrofuran at 70℃; for 0.5h; Inert atmosphere;
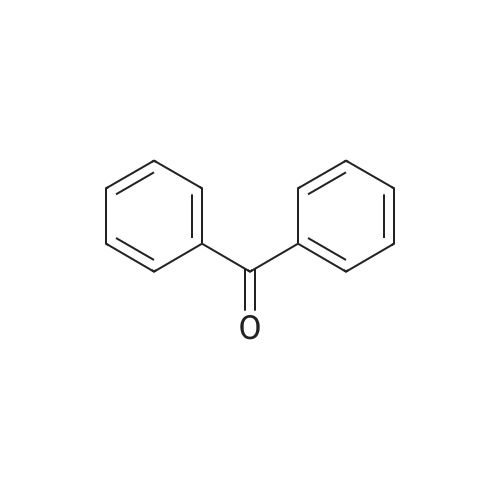
Stage #2: benzophenone In tetrahydrofuran at 70℃; for 2h; Inert atmosphere;
Stage #3: With hydrogenchloride In water; acetone at 5 - 50℃; |
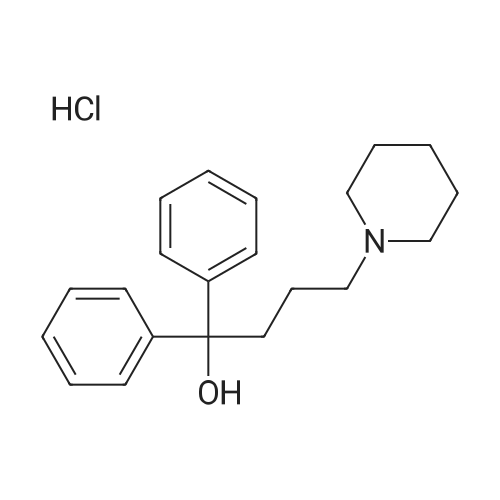
1.S2-6.S2; 1.S3-6.S3; 1-5 S2. Preparation of α, α-diphenyl-1-piperidinebutanol
In a nitrogen atmosphere, 12 g of magnesium strips were added to the bottle, and the reaction was initiated with 40 g of tetrahydrofuran, 0.5 g of iodine, and 0.5 g of bromoethane. After the initiation was completed,A solution of 120 g of intermediate N- (3-chloropropyl) piperidine in tetrahydrofuran was slowly added dropwise.(80g of N- (3-chloropropyl) piperidine is mixed with 40g of tetrahydrofuran). After the addition is completed, the temperature is maintained at 70 ° C and refluxed for 30 minutes. After the temperature is maintained, 161g of benzophenone (0.9eq) is added dropwise while maintaining the reflux temperature. Of tetrahydrofuran solution (81 g of benzophenone dissolved in 80 g of tetrahydrofuran), the reaction was incubated for 2 h. After incubation, cool down to room temperature. The material was poured into 600 mL of ammonium glacial chloride solution (10% by mass), hydrolyzed for 1-2 hours, and filtered to obtain a off-white solid, which was dried in a blast drying oven overnight, weighing 123g, which is α, α -Diphenyl-1-piperidinebutanol.S3. Preparation of α, α-diphenyl-1-piperidinebutanol hydrochloride100g of α, α-diphenyl-1-piperidinebutanol was added with 8 times the mass volume of acetone, and the temperature was raised to 50 ° C. After stirring and dissolving, activated carbon was added to continue stirring for 0.5h, and the filtrate was filtered while hot. Incubate at -45 ° C, and then add 6mol / l hydrochloric acid dropwise at a rate of 2mL / min to the solution pH 6. Keep the temperature and stir for 1-2h at 40-45 ° C. Use a gradient to reduce the temperature to 20-25 ° C and stir for 1-2h. Finally, the temperature was lowered to 5-10 ° C, and the mixture was stirred for 2-4 hours, and then filtered and dried to obtain 103 g of off-white difenidol hydrochloride, and the salt yield was 92.14%. The final product was tested by HPLC, and its purity was 99.95%. The by-product diphenylmethanol was 0.03%. At the same time, the olefinic impurities were 0% and the total impurity content was 0.05%. |
|
Stage #1: N-(3-chloropropyl)-piperidine With ethyl bromide; iodine; magnesium In tetrahydrofuran at 50 - 65℃; for 1h;
Stage #2: benzophenone In tetrahydrofuran for 3h; Further stages; |
S2. Preparation of α, α-diphenyl-1-piperidinebutanol Magnesium shavings 8.80 g (0.367 mol) were mixed with tetrahydrofuran (100 mL), iodine and ethyl bromide were added, and 68 g (0.42 mol) of 1- (3-chloropropyl) piperidine was added at 50 to 65 ° C for 1 hour , Adding 150 mL of tetrahydrofuran and 49 g (0.269 mol) of benzophenone, continuing the reaction for 3 hours, recovering tetrahydrofuran, adding ammonium chloride aqueous solution to obtain α, α-diphenyl-1-piperidine butanol ) 67.0g. S3. Preparation of α, α-diphenyl-1-piperidinebutanol hydrochloride Obtained in step S2 α, α- diphenyl-piperidin-butanol (crude base) was dissolved in 75% methanol, pH 5.8 to 6.98 disodium hydrogen phosphate - sodium dihydrogen phosphate solution, the system was maintained at a pH of 5-6, and heated to 55 , added mass of α, α- diphenyl-1-piperidine butanol of 0.05% by mass of activated carbon for 1 hour, filtered;Hydrochloric acid was added to the resulting filtrate, the pH was adjusted to 5, the control temperature of 50 ~ 60 stirred for 30min, filtered, dried 60 deg.] CDry residue, to give α, α- diphenyl-1-piperidine butanol hydrochloride. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping