| 95% |
|
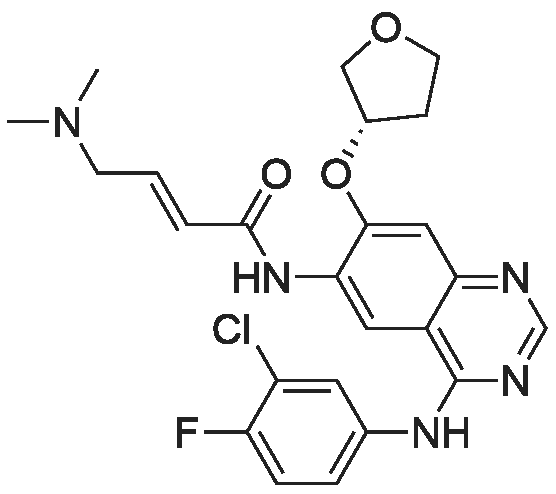
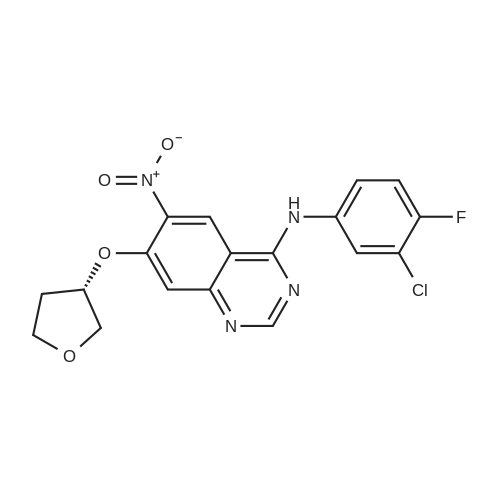
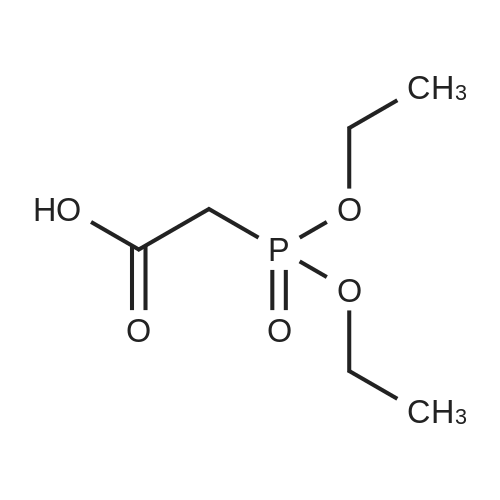
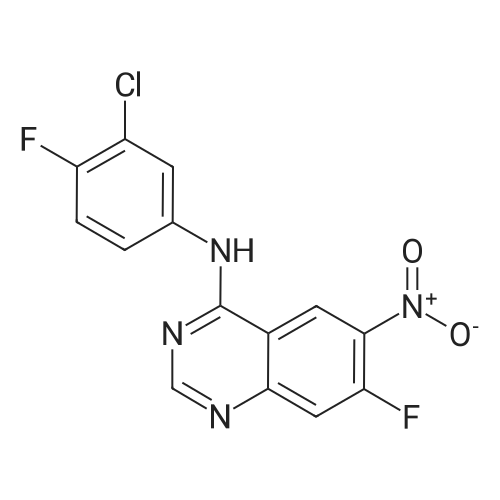
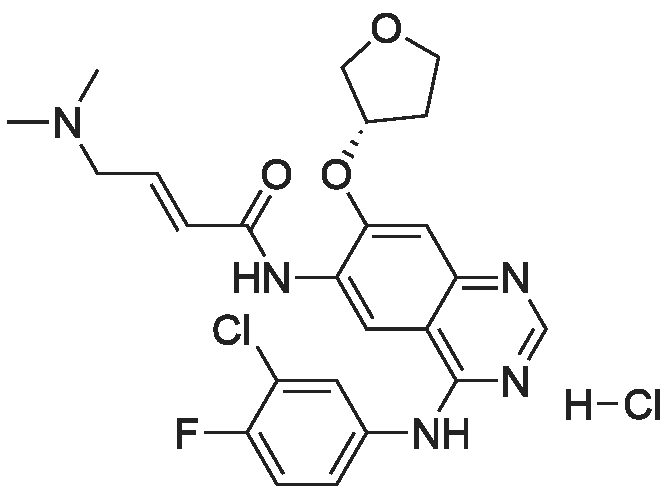
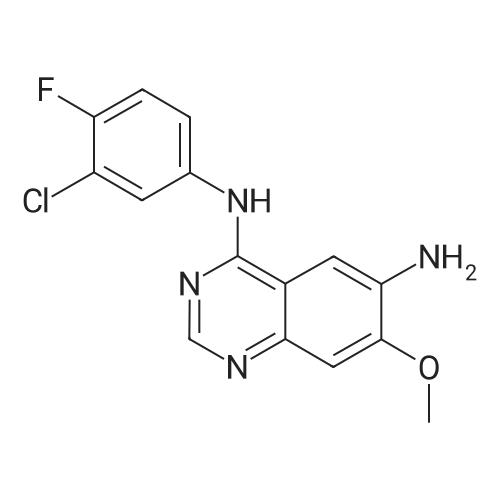
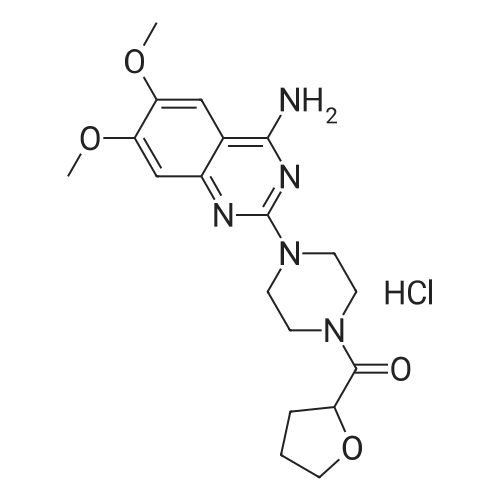
(E)-4-Dimethylaminocroton hydrochloride 9.65 g was sequentially added to a 500 ml four-necked flask.93 g of N-methylpyrrolidone was stirred and cooled to -5 C, and 6.6 g of thionyl chloride was slowly added dropwise to the stirring solution to ensure that the temperature of the reaction solution was between 0 and -5 C, and within the above temperature range after the completion of the dropwise addition. Take a fall of 20min.N-4-[(3-chloro-4-fluorophenyl)]-7-[(3S)-tetrahydrofuran-3-yl]oxy}-4,6-quinazolinediamine 12.45g Dissolved in 62g of N-methylpyrrolidone as a dropping solution,Slowly add this solution to the above stirring solution and ensure that the temperature is between 0 and -5 C.After the completion of the dropwise addition, the mixture was stirred for 15 minutes in the above temperature range to obtain an acylation reaction liquid.Then, 100 g of water is slowly added dropwise to the above reaction solution and the temperature is controlled to not exceed 15 C, and then a 20% NaHH solution is slowly added dropwise thereto to about 66.3 g to rho Eta = 9 to 10, and the controlled temperature does not exceed 25 during the dropwise addition. C. After the dropwise addition, the reaction liquid was extracted three times (353 g X 3 ) with butyl acetate at 45 C, and the organic phase was collected, washed with water at 45 C for 5 times (250 g X 5 ), and then directly stirred for crystallization (stirring and crystallization) The temperature is selected from the table shown in Table 2) 3h. Filtration, crystals, butyl acetate 53g rinse the filter cake once, and finally dried under vacuum at 40 C for 2h to obtain a white solid. The yield, yield, purity and content of impurity I are shown in Table 2. |
| 93% |
With Bromoform; triethylamine; triphenylphosphine; In dichloromethane; at 25℃; for 1h; |
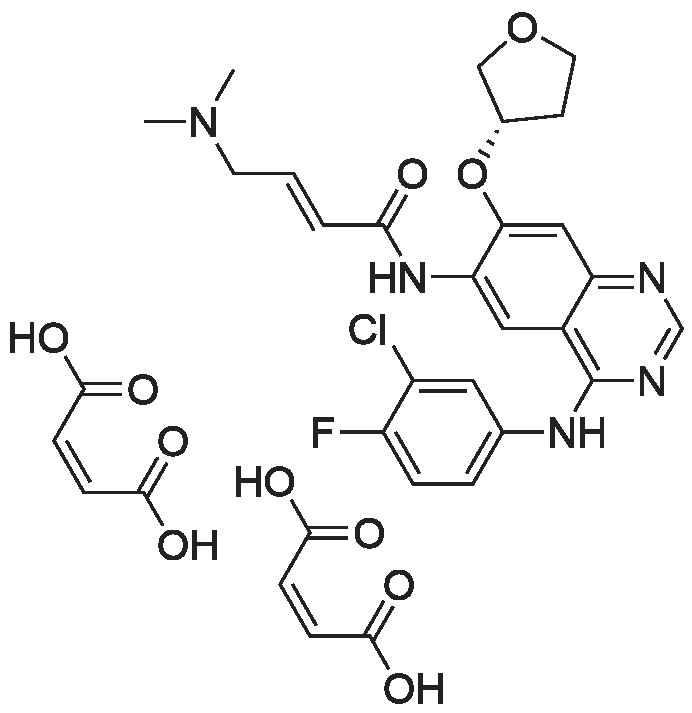
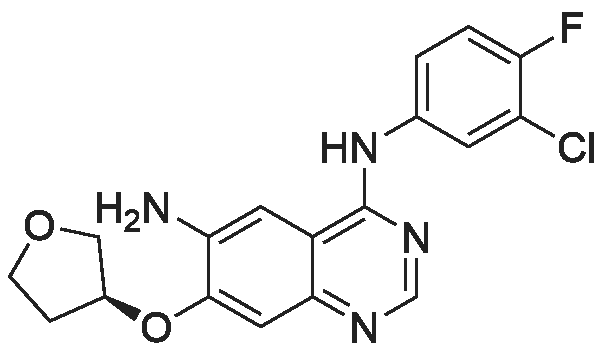
50.1 g of dimethylamino croton hydrochloride,108.0 g of N4-(3-chloro-4-fluoro-phenyl)-7-((S)-tetrahydrofuran-3-yloxy)quinazoline-4,6-diamine,84.2 mL of triethylamine and 100.3 g of carbon tetrabromide were dissolved in dry 1944 mL of dichloromethane, and 79.4 g of triphenylphosphine was added in portions at 25 C with stirring.After 60 min, the reaction was filtered, and the filtrate was washed with water (1900 mL×2×).The residue was recrystallized from 390 mL of isopropanol to give 130.2 g of afatinib.The molar yield was 93% and the product purity was 98%. |
| 87.4% |
|
(1) To the 1 L bottle is added in three trans -4 - dimethylamino crotonic acid hydrochloride 50 g, N - methyl pyrrolidone 100 g, ethyl acetate 400 g, cooled to 5 C, to the system adds by drops the chlorination sulfoxide 36.1 g, after dropping, 5 C conditions under stirring, HPLC monitoring to the reaction is complete (SM1 residue 1.8%). (2) Will be N4 - (3 - chloro -4 - fluoro - phenyl) -7 - (S)- tetrahydrofuran -3 - oxyacetyl) quinazoline - 4, 6, - diamine (SM2) 45 g N - methyl pyrrolidone for 225 g dissolved, the solution is dropped is added to step (1) in the reaction system, after dropping, 0 C reaction, TLC monitoring reaction to SM2 reaction is complete. (3) Temperature control 20 C following, to the step (2) in the system drop of pure water 1.35 kg, for 20 weight % sodium hydroxide solution to adjust pH=9, ethyl acetate 675 g * 2 extraction, control gathering temperature is 45 C, combined with the organic phase, purified water 0.9 kg * 2 washing, organic phase in the 45 C concentrated under reduced pressure to V (the remaining system)/m (SM2) is about 10, lowering the temperature to -5 C, crystallization 2 hr, filtered, the filter cake in the 50 C vacuum drying, to obtain yellowish solid 51 g, yield 87.4%. The detection HPLC purity of 99.6%. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping