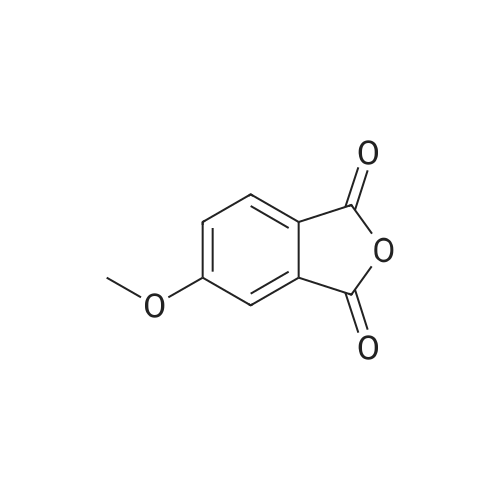
Alternatived Products of [ 21345-01-7 ]
Product Details of [ 21345-01-7 ]
| CAS No. : | 21345-01-7 |
MDL No. : | MFCD00127685 |
| Formula : |
C14H8O4
|
Boiling Point : |
- |
| Linear Structure Formula : | - |
InChI Key : | HDQBHDKNRKRCNH-UHFFFAOYSA-N |
| M.W : |
240.21
|
Pubchem ID : | 12380384 |
| Synonyms : |
|
Application In Synthesis of [ 21345-01-7 ]
* All experimental methods are cited from the reference, please refer to the original source for details. We do not guarantee the accuracy of the content in the reference.
- Downstream synthetic route of [ 21345-01-7 ]
- 1
-
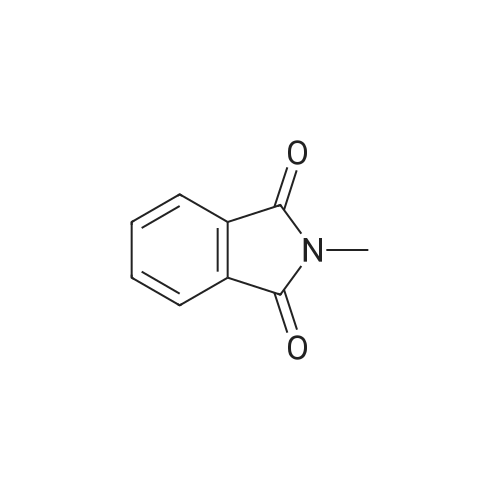
 [ 37951-15-8 ]
[ 37951-15-8 ]

-
 [ 21345-01-7 ]
[ 21345-01-7 ]
| Yield | Reaction Conditions | Operation in experiment |
|
With acetic anhydride; acetic acid; for 3.0h;Resolution of racemate; |
Compound 2 (6.0 g) was dissolved in glacial acetic acid (60 mL) and acetic anhydride (60 mL) and heated to reflux for 3 hrs. The solvent was removed on a rotary evaporator to obtain Compound 3. |
|
With acetic anhydride; In acetic acid; for 3.0h;Reflux; |
Compound 2 (6.0 g) was dissolved in glacial acetic acid (60 mL) and acetic anhydride (60 mL) and heated to reflux for 3 hrs. The solvent was removed on a rotary evaporator to obtain Compound 3. |
|
With acetic anhydride; acetic acid; for 3.0h;Reflux; |
6.0 g of compound 2 was dissolved in 60 ml of glacial acetic acid and 60 ml of acetic anhydride, and the mixture was heated under reflux for 3 hours to obtain compound 3 after removal of the solvent. |
Reference:
[1]Journal of general chemistry of the USSR,1980,vol. 50,p. 1874 - 1878
Zhurnal Obshchei Khimii,1980,vol. 50,p. 2313 - 2318
[2]Patent: WO2013/13609,2013,A1 .Location in patent: Page/Page column 16
[3]Patent: US2015/31721,2015,A1 .Location in patent: Paragraph 0146
[4]Chemistry - A European Journal,2017,vol. 23,p. 10616 - 10625
[5]Patent: TWI582078,2017,B .Location in patent: Paragraph 0145; 0151; 0152
- 3
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
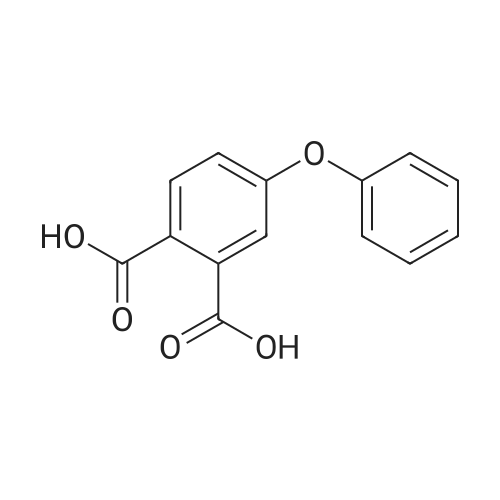
 [ 550-44-7 ]
[ 550-44-7 ]
| Yield | Reaction Conditions | Operation in experiment |
| 64% |
|
The above results show that an exchange occurred between 4-phenoxy-N-methylphthalmide and phthalic anhydride, resulting in a 46% yield of N-methylphthalimide and a 64% yield of <strong>[21345-01-7]4-phenoxyphthalic anhydride</strong> or acid. |
- 4
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
 [ 142-71-2 ]
[ 142-71-2 ]

-
 [ 77447-50-8 ]
[ 77447-50-8 ]
- 5
-
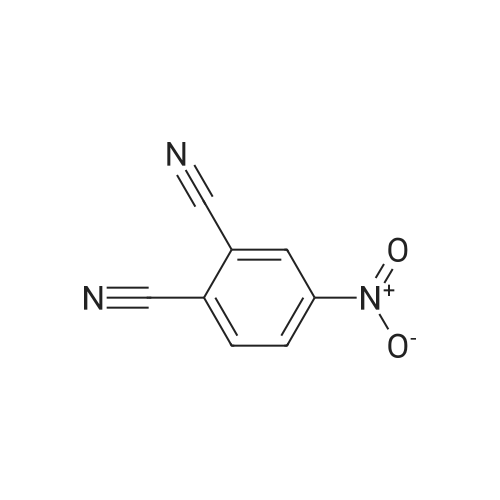 [ 31643-49-9 ]
[ 31643-49-9 ]

-
 [ 21345-01-7 ]
[ 21345-01-7 ]
- 6
-
 [ 108-95-2 ]
[ 108-95-2 ]

-
 [ 21345-01-7 ]
[ 21345-01-7 ]
- 7
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
 [ 39687-95-1 ]
[ 39687-95-1 ]

-
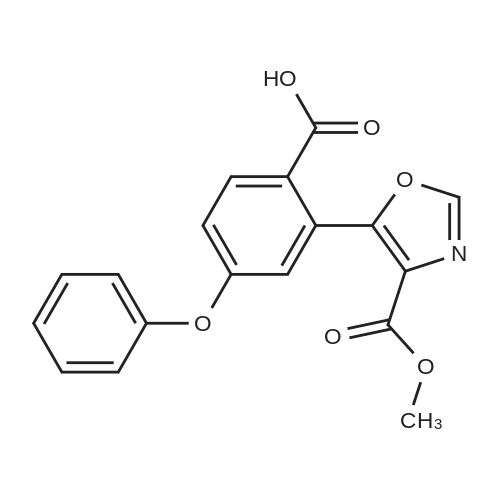 [ 1421312-31-3 ]
[ 1421312-31-3 ]
| Yield | Reaction Conditions | Operation in experiment |
| 9 g |
With 1,8-diazabicyclo[5.4.0]undec-7-ene; In tetrahydrofuran; at 20℃; for 1.0h; |
Compounds 3 (6.0 g) and methyl isocyanoacetate (2.65 g) were dissolved in THF (60 mL). 3.54 g of DBU (CAS No. 6674-22-2) was added in drop-wise at room temperature and stirred for 1 hr. at room temperature. After extracted with ethyl acetate under alkaline conditions to remove the impurities, the pH value of the aqueous phase was adjusted to 3 with diluted HCL Extracted with ethyl acetate, washed with water and dried with anhydrous Na2S04 and fi ltered, the resulting organic phase was distilled on a rotary evaporator to obtain 9.0g of Compound 4. |
| 9 g |
With 1,8-diazabicyclo[5.4.0]undec-7-ene; In tetrahydrofuran; at 20℃; for 1.0h; |
Compounds 3 (6.0 g) and methyl isocyanoacetate (2.65 g) were dissolved in THF (60 mL). 3.54 g of DBU (CAS No. 6674-22-2) was added in drop-wise at room temperature and stirred for 1 hr. at room temperature. After extracted with ethyl acetate under alkaline conditions to remove the impurities, the pH value of the aqueous phase was adjusted to 3 with diluted HCl. Extracted with ethyl acetate, washed with water and dried with anhydrous Na2SO4 and filtered, the resulting organic phase was distilled on a rotary evaporator to obtain 9.0 g of Compound 4. |
| 9 g |
With 1,8-diazabicyclo[5.4.0]undec-7-ene; In tetrahydrofuran; at 20℃; for 1.0h; |
Compound 3 (6.0 g) and 2.65 g of methyl isocyanoacetate were dissolved in 60 ml of tetrahydrofuran and 3.54 g of DBU (CAS Registry No. 6674-22-2) was added dropwise at room temperature and stirred at room temperature 1 hourThe mixture was extracted with ethyl acetate to remove impurities. The aqueous phase was adjusted to pH = 3 with dilute hydrochloric acid, extracted with ethyl acetate, washed with water and dried over anhydrous Na2SO4. After removal of the solvent, 9.0 g of compound 4 was obtained. |
- 8
-
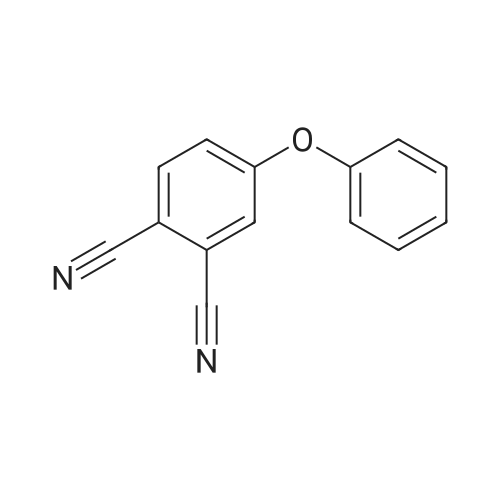 [ 38791-62-7 ]
[ 38791-62-7 ]

-
 [ 21345-01-7 ]
[ 21345-01-7 ]
- 9
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
[(4-hydroxy-1-methyl-7-phenoxy-isoquinoline-3-carbonyl)-amino]-acetic acid
[ No CAS ]
- 10
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
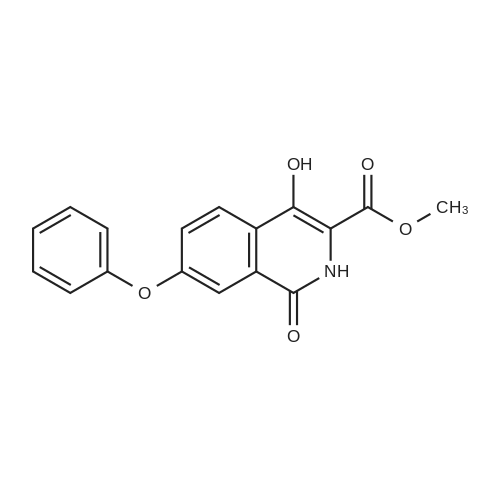 [ 1421312-32-4 ]
[ 1421312-32-4 ]
- 11
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
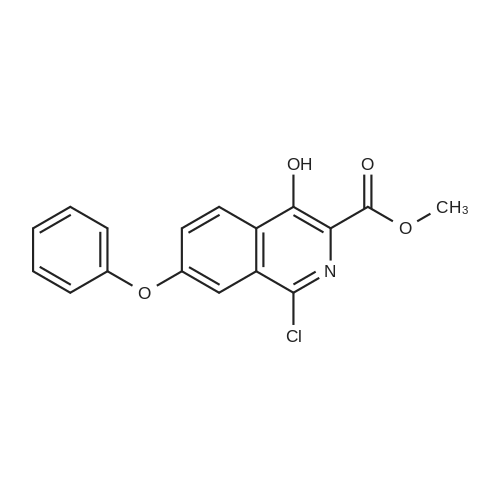 [ 1421312-33-5 ]
[ 1421312-33-5 ]
- 12
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
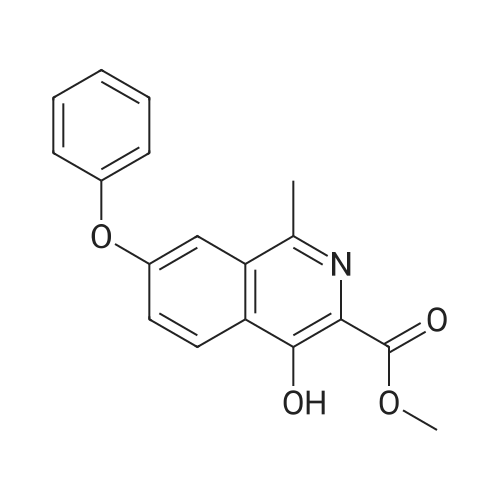 [ 1421312-34-6 ]
[ 1421312-34-6 ]
- 13
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
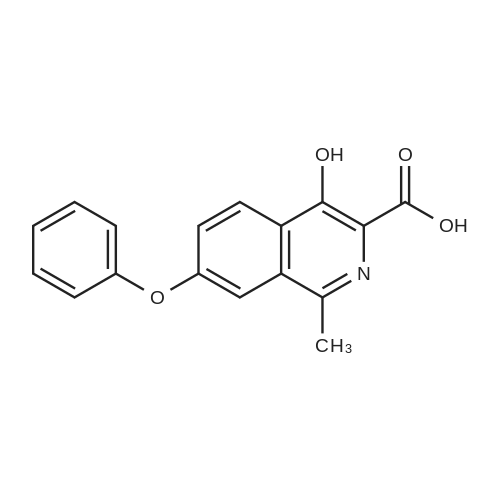 [ 1421312-35-7 ]
[ 1421312-35-7 ]
- 14
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
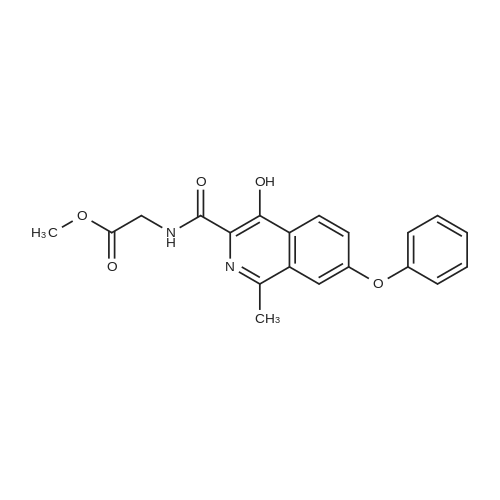 [ 1421312-36-8 ]
[ 1421312-36-8 ]
- 15
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
4-(phenoxy)-N-hydroxyphthalimide
[ No CAS ]
- 16
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
 [ 676-58-4 ]
[ 676-58-4 ]

-
C15H12O4
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 85% |
In tetrahydrofuran; at -10 - 0℃;Inert atmosphere; |
A three-necked flask was charged with compound formula 1 (24.02 g, 100 mmol) and tetrahydrofuran (120 mL),Dissolve with stirring, cool to -10 0 and switch nitrogen 3 times under vacuum.Under the protection of nitrogen, a methylmagnesium chloride tetrahydrofuran solution (2.0M, 55mL) was slowly dropped into the reaction bottle, and the reaction was kept warm for 2 to 4 hours after the dropping was completed.At the end of the reaction, 1 mol / L diluted hydrochloric acid (240 mL) was added to quench the reaction.The aqueous phase was extracted twice with ethyl acetate (120 mL).The combined organic phases were washed once with saturated brine (120 mL), and dried over anhydrous sodium sulfate.After concentration, it was recrystallized from a mixed solvent of petroleum ether and ethyl acetate to obtain Compound 2 (21.78 g, 85%). |
- 17
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
ethyl 4-hydroxy-1-methyl-7-phenoxy-3-isoquinolinecarboxylate
[ No CAS ]
- 18
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C24H27NO7
[ No CAS ]
- 19
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C21H21NO6
[ No CAS ]
- 20
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C25H23NO7S
[ No CAS ]
- 21
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C18H15NO4
[ No CAS ]
- 22
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C19H17NO4
[ No CAS ]
- 23
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C15H11NO3
[ No CAS ]
- 24
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C15H11ClO3
[ No CAS ]
- 25
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C25H23NO6
[ No CAS ]
- 26
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
C24H19NO5
[ No CAS ]
- 27
-
 [ 21345-01-7 ]
[ 21345-01-7 ]

-
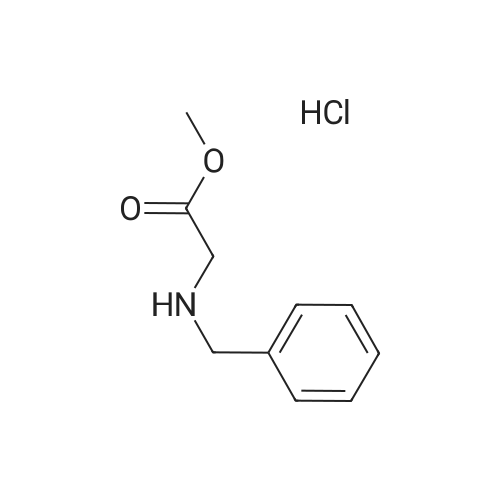 [ 17136-35-5 ]
[ 17136-35-5 ]

-
C24H21NO6
[ No CAS ]
| Yield | Reaction Conditions | Operation in experiment |
| 97% |
With potassium carbonate; In acetone; at 55 - 60℃; for 4.0h; |
Add 50g 4-phenoxy phthalic anhydride and 500g acetone into a 1L three-necked flask,After stirring and dissolving, 58 g of anhydrous potassium carbonate and 54 g of N-benzylglycine methyl ester hydrochloride were added.Slowly raise the temperature to reflux, control the temperature at 5560, react for 4h, after the completion of the reaction, cool to room temperature and filter.The filter cake was washed with acetone (100 g×2) and drained until no droplets dripped to obtain 119 g of wet compound 1 potassium salt.Add 1.2 kg of purified water and 119 g of wet compound 1 potassium salt to a 2L three-necked flask, and stir to dissolve.Control the temperature at 010, adjust the pH value to 23 with 2M hydrochloric acid.Heat preservation and stirring for crystallization for 2h, and wash the filter cake with purified water (100g×2),After being sucked dry, it was dried by blowing at 50-60C for 8 hours to obtain 85 g of white solid compound 1, with a yield of 97%. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science















 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping