| 85% |
With potassium carbonate In ethylene glycol at 120℃; for 4.5h; Inert atmosphere; |
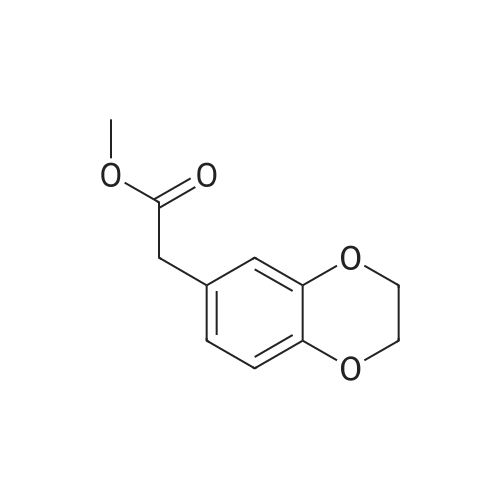
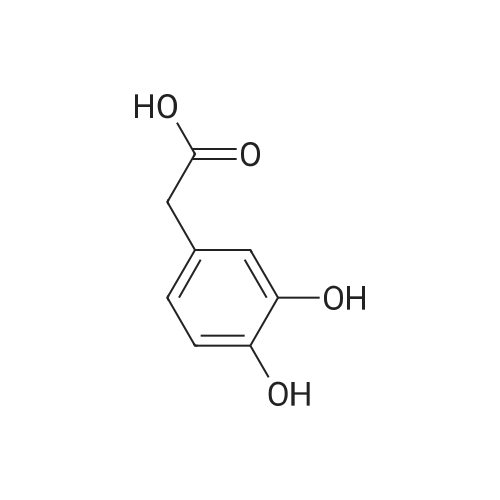
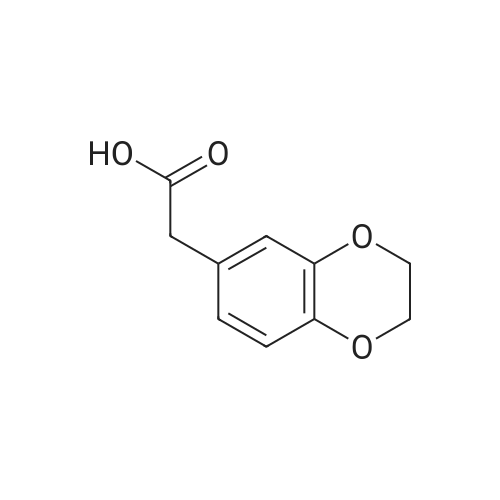
120.1 Step 1. 2-(2,3-dihydrobenzo[b][1,4]dioxacyclohexene-6-yl)acetic acid (GEN1-159-1)
1,2-dibromoethane (1.13 g, 6.0 mmol) was added to a solution of 2-(3,4-dihydroxyphenyl)acetic acid (500 mg, 3.0 mmol) and K2CO3 (1.24 g, 9.0 mmol) in ethylene glycol (5.0 mL). The resulting mixture was stirred at 120°C for 4.5 hours under a N2 atmosphere. The reaction mixture was cooled to room temperature and diluted with water (40 mL). EtOAc (40 mL) was added and the mixture was stirred for 5 minutes. The organic layer was discarded, and the aqueous layer was acidified with 2M HCl to pH <1. The aqueous layer was extracted with EA (60 mL*3). The combined organic layer was washed with water (150 mL), brine (150 mL), dried over Na2SO4 and it was concentrated to obtain a black oil. The crude product was purified by silica gel column chromatography and eluted with (EA/(PE + EA)=60%) to obtain the desired compound (495 mg, yield 85%) as a black oil. MS (ESI): Rt = 1.01 min, m/z 217 [M+Na]+, Purity: 100% 254 nm, 100% 214 nm. |
| 52% |
With potassium carbonate In ethylene glycol at 120℃; for 4.5h; |
7 3,4-Ethylenedioxyphenylacetic acid (11) and 3,4-propylenedioxyphenylacetic acid (12)
To a solution of 3,4-dihydroxyphenylacetic acid (500mg, 3.0mmol) and either 1,2-dibromoethane (1.13g, 6.0mmol) or 1,3-dibromopropane (1210mg, 6.0mmol) in ethylene glycol (5mL) was added anhydrous K2CO3 (1240mg, 9.0mmol). After heating at 120°C for 4.5h, the mixtures were cooled, diluted with H2O (50mL), acidified to pH N HCl), and extracted with EtOAc (3×20mL). The organic layers were concentrated under reduced pressure, and residues were purified by column chromatography (EtOAc/ Hexane=50:50 for 11; 40:60 for 12) to give 1144 (301mg, 52%) or 1245 (260mg, 42%). 11 1H NMR: (300MHz, CDCl3, δ) 3.56 (s, 2H, CH2), 4.23 (s, 4H, CH2), 6.75-6.87 (m, 3H, CH), 11.64 (br s, 1H, -COOH). 12 1H NMR: (250MHz, CDCl3, δ) 2.19 (app p, 2H, J=7.5Hz, CH2), 3.55 (s, 2H, CH2), 4.21 (m, 4H, CH2), 6.81-6.95 (m, 3H, CH), 10.50 (br s, 1H, -COOH). |
|
With potassium carbonate |
120.1 Step 1.
Step 1. 2-(2,3-dihydrobenzo[b][1,4]dioxacyclohexene-6-yl)acetic acid (GEN1-159-1) 1,2-dibromoethane (1.13 g, 6.0 mmol) was added to a solution of 2-(3,4-dihydroxyphenyl)acetic acid (500 mg, 3.0 mmol) and K2CO3 (1.24 g, 9.0 mmol) in ethylene glycol (5.0 mL). The resulting mixture was stirred at 120°C for 4.5 hours under a N2 atmosphere. The reaction mixture was cooled to room temperature and diluted with water (40 mL). EtOAc (40 mL) was added and the mixture was stirred for 5 minutes. The organic layer was discarded, and the aqueous layer was acidified with 2M HCl to pH <1. The aqueous layer was extracted with EA (60 mL*3). The combined organic layer was washed with water (150 mL), brine (150 mL), dried over Na2SO4 and it was concentrated to obtain a black oil. The crude product was purified by silica gel column chromatography and eluted with (EA/(PE + EA)=60%) to obtain the desired compound (495 mg, yield 85%) as a black oil. MS (ESI): Rt = 1.01 min, m/z 217 [M+Na]+, Purity: 100% 254 nm, 100% 214 nm. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping