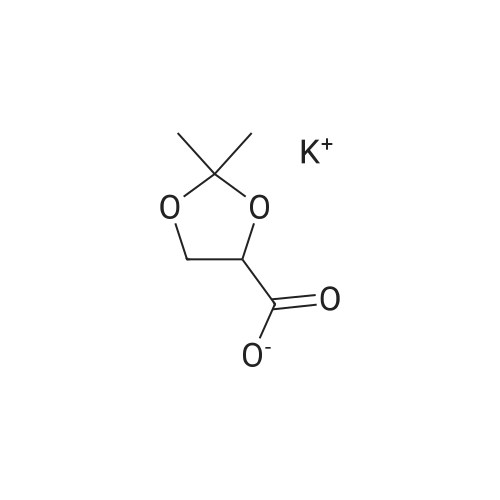
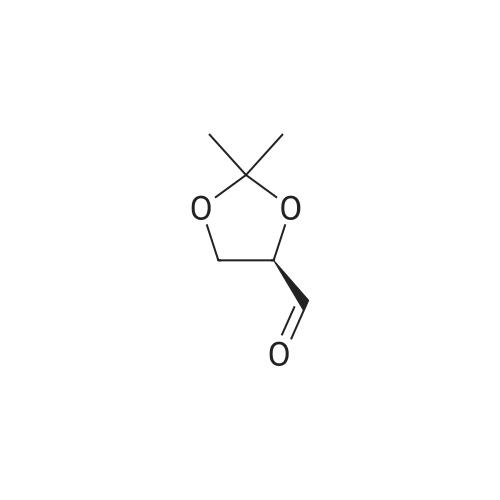
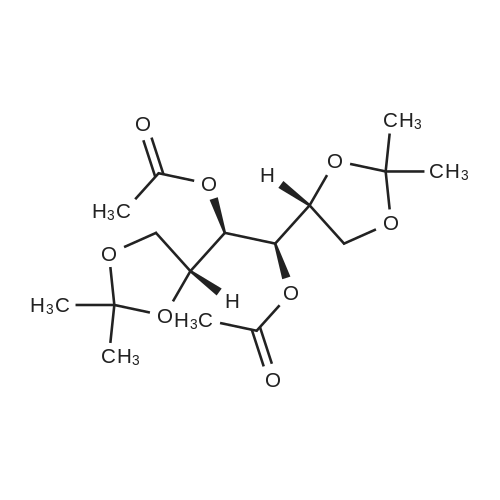
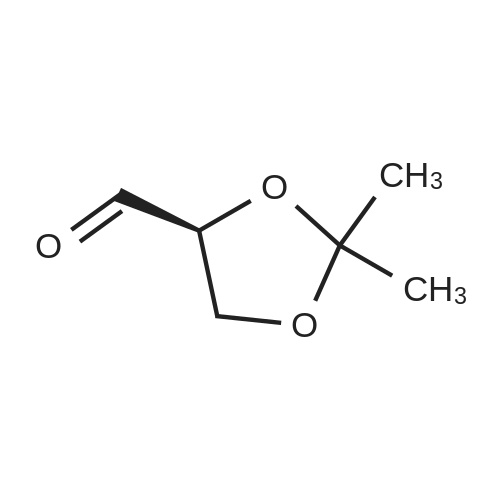
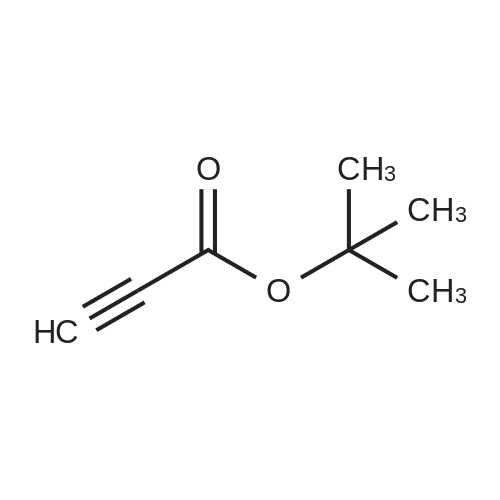
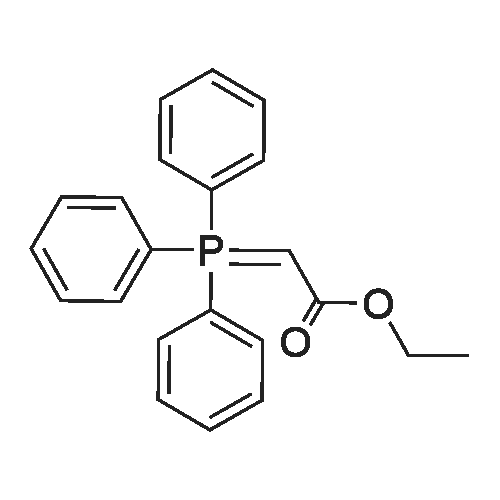
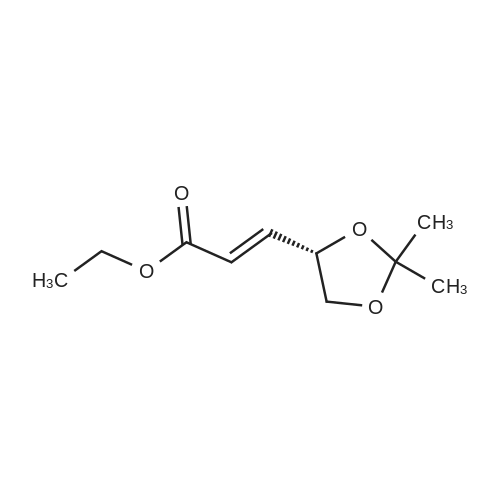
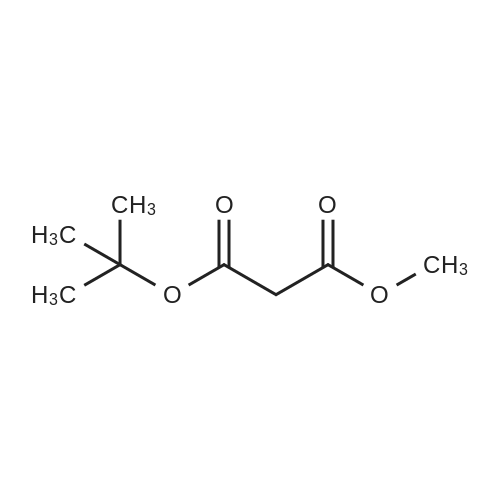
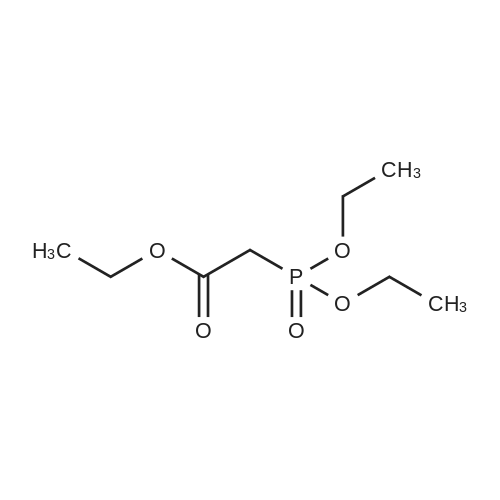
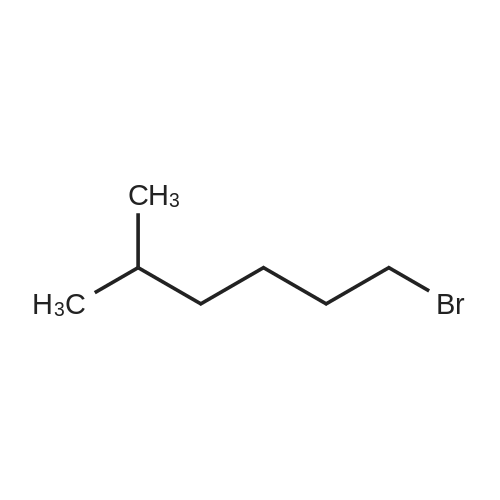
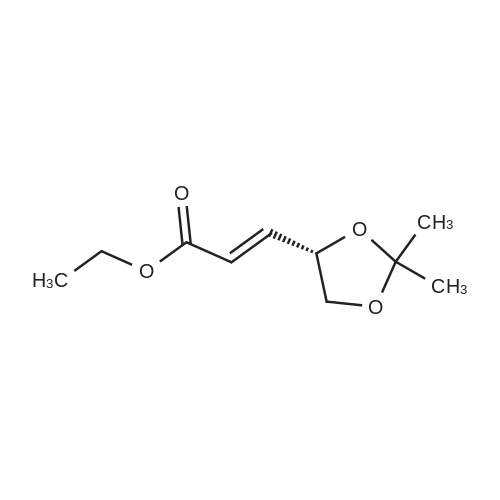
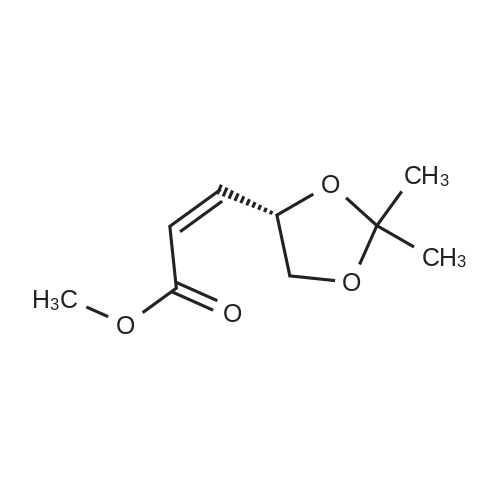
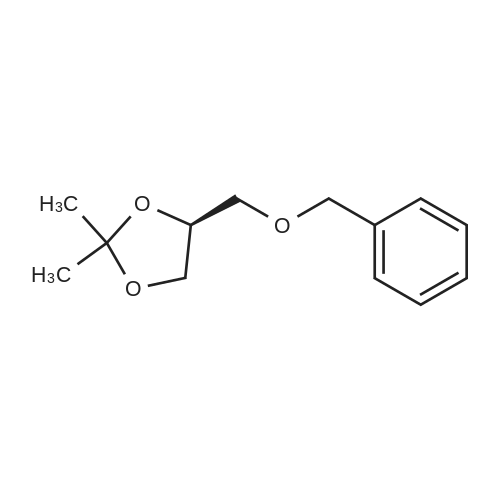
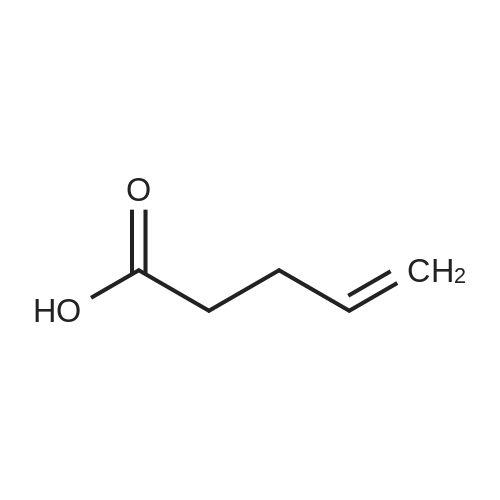
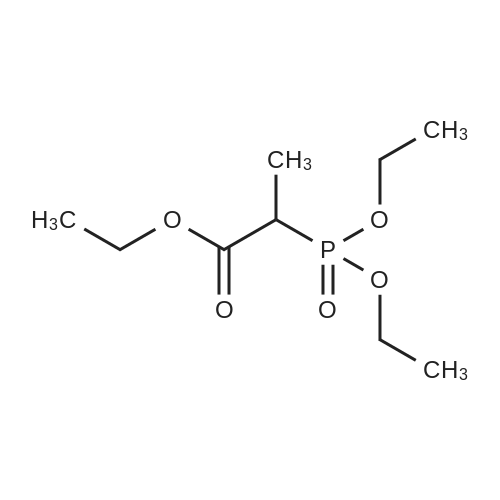
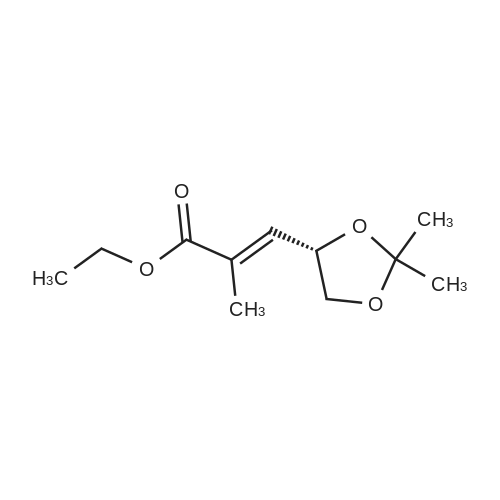
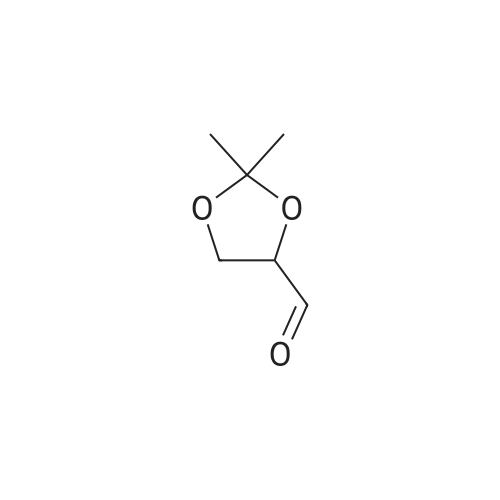
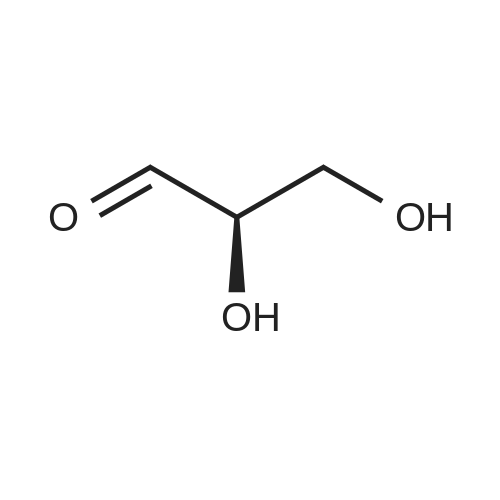
| 99% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer at 20℃; for 1h; |
|
| 99% |
Stage #1: (1S,2S)-1,2-bis((R)-2,2-dimethyl-1,3-dioxolan-4-yl)ethane-1,2-diol With Sodium hydrogenocarbonate In dichloromethane at 23℃; for 0.166667h;
Stage #2: With sodium (meta)periodate In dichloromethane for 3h; |
|
| 98% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 20℃; for 1.5h; |
|
| 98.4% |
With tetrabutylammonium bromide; pyridinium chlorochromate at 20 - 25℃; for 3h; |
1-8 Example 1
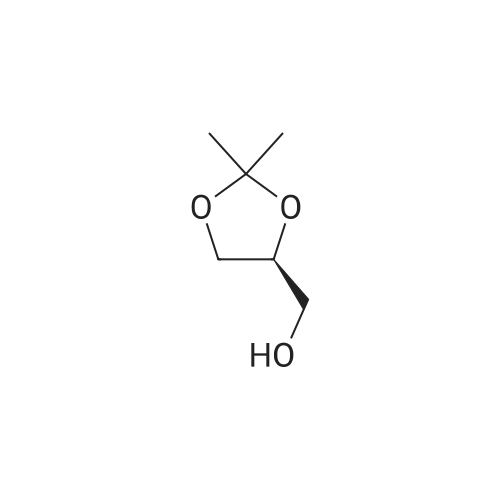
Add 200 g of dioxymethane to the clean reactor, put in 20 g of diacetone-D-mannitol under stirring, and control the temperature at 20 °C to 25 °C.Then, 0.2 g of tetrabutylammonium bromide and 17.9 g of oxidant PCC were added, and the temperature was controlled to keep the reaction at 20°C to 25°C for 3 hours. After the reaction was completed, suction filtration was performed to recover the solids in the reaction solution. , the collected filter was subjected to vacuum distillation to recover the solvent to obtain an oily product R-(+)-2,2-dimethyl-1,3-dioxolane-4-carbaldehyde 19.5g,The product yield was 98.4%, and the purity content was 99.7% |
| 97.9% |
With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; 18-crown-6 ether; Calcium Chloride-Oxygen In water monomer; ethyl acetate at 5 - 10℃; for 1.5h; |
3 Example 3
200 g of ethyl acetate, 23 g of diacetone-D-mannitol, 0.6 g of 15-crown-5-ether, and TEMPO 0.5 g were sequentially added to a clean reactor.After stirring and mixing uniformly, the temperature was slowly lowered to 5 ° C, and 140 g of a calcium hypochlorite aqueous solution having a mass percentage of 10 wt% was added dropwise, and the temperature of the reaction system was controlled to be below 5 ° C during the dropwise addition. After the completion of the dropwise addition, the reaction was continued to make the temperature The oxidation reaction was carried out for 1.5 h at 5 ° C to 10 ° C. After the reaction was completed, it was allowed to stand and layer.The aqueous layer was removed, and the collected organic layer was washed once with 50 g of water, and the aqueous layer was separated by layering.The collected organic layer was added to 20 g of anhydrous sodium sulfate and stirred for 30 min.After suction filtration, the filtrate was collected, and the filtrate was concentrated under reduced pressure to remove solvent.After the obtained wet product is baked,Obtained 22.3 g of dry R-glyceraldehyde acetal,Yield 97.9%,The gas phase content was 99.5%. |
| 96% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 20℃; for 1.5h; |
|
| 95% |
With sodium (meta)periodate; mesoporous silica In dichloromethane at 0℃; for 0.5h; |
|
| 95% |
With sodium (meta)periodate In dichloromethane; water monomer at 20℃; for 3h; |
|
| 95% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer for 2h; |
|
| 94% |
With sodium (meta)periodate; water monomer In dichloromethane at 20℃; for 2h; |
|
| 94% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 5 - 20℃; for 2h; |
1.1 Step 1. Synthesis of (4R)-2,2-dimethyl-1,3-dioxolane-4-carbaldehyde (C15)
To a solution of (lS,2S)-l,2-bis[(4R)-2,2-dimethyl-1,3-dioxolan-4-yl]ethane-l,2-diol C14 (30 g, 114.4 mmol) in DCM (300 mL) was added NaHCCb (12 mL) and the mixture was cooled to 10 °C before NaI04 (49 g, 229.1 mmol) was added portion wise at such a rate that internal temperature stayed below 5 °C. The reaction was stirred at room temperature for 2 hours, then was filtered on Celite. The cake was washed with DCM and the filtrate was concentrated in vacuo , yielding (4R)-2,2-dimethyl-l,3-dioxolane-4-carbaldehyde (28.0 g, 94%) 1H NMR (400 MHz, DMSO-d6) δ 9.60 (s, 1H), 4.52 (ddd, J = 7.3, 4.6, 1.6 Hz, 1H), 4.13 - 4.05 (m, 2H), 1.38 (s, 3H), 1.33 (s, 3H). |
| 94% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 5 - 20℃; for 2h; |
1.1 Step 1. Synthesis of (4R)-2,2-dimethyl-1,3-dioxolane-4-carbaldehyde (C15)
To a solution of (lS,2S)-l,2-bis[(4R)-2,2-dimethyl-1,3-dioxolan-4-yl]ethane-l,2-diol C14 (30 g, 114.4 mmol) in DCM (300 mL) was added NaHCCb (12 mL) and the mixture was cooled to 10 °C before NaI04 (49 g, 229.1 mmol) was added portion wise at such a rate that internal temperature stayed below 5 °C. The reaction was stirred at room temperature for 2 hours, then was filtered on Celite. The cake was washed with DCM and the filtrate was concentrated in vacuo , yielding (4R)-2,2-dimethyl-l,3-dioxolane-4-carbaldehyde (28.0 g, 94%) 1H NMR (400 MHz, DMSO-d6) δ 9.60 (s, 1H), 4.52 (ddd, J = 7.3, 4.6, 1.6 Hz, 1H), 4.13 - 4.05 (m, 2H), 1.38 (s, 3H), 1.33 (s, 3H). |
| 93% |
With sodium (meta)periodate In tetrahydrofuran; water monomer |
|
| 92% |
With sodium hydroxide; sodium (meta)periodate; potassium dihydrogen orthophosphate In dichloromethane; water monomer for 0.333333h; pH 7; |
|
| 92% |
With sodium (meta)periodate In dichloromethane for 0.333333h; Ambient temperature; pH 7; |
|
| 91% |
With sodium (meta)periodate; water monomer In dichloromethane at 32℃; for 1h; |
|
| 90% |
With sodium (meta)periodate In dichloromethane; water monomer |
|
| 87% |
With sodium (meta)periodate; mesoporous silica In dichloromethane at 0℃; |
|
| 86% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In methylene chloride; water monomer at 20 - 25℃; for 2.33333h; |
1.2
Step 2: Preparation of 2,2-dimethyl-[l,3]-dioxorane-4-carboaldehyde; 72.4 g of the compound obtained in Step 1 was dissolved in 724 ml of methylchloride, and 30 ml of saturated sodium bicarbonate was added thereto. The mixture was cooled in a water bath, and 118 g of sodium methaperiodate was added in small portion thereto over a period of 20 min while keeping the temperature at under 25°C. The reaction mixture was stirred at room temperature for 2 hours. After confirmng the completion of the reaction by thin layer chromatography (TLC), 36 g of anhydrous magnesiumsulfate was added to the reaction mixture, and stirred for 20 min. The resulting mixture was filtered and distilled under a reduced pressure at 30°C to remove the solvent, and the residue was further subjected in distillation under an atmospheric pressure at 55°C to completely remove the solvent. The reacting residue was distilled at 10 torr, about 40°C to obtain 61.6 g (yield 86%) of the title compound as a colorless liquid.NMR (300 MHz, CDC13): 1.41(s, 3H), 1.47(s, 3H), 4.07-4.19(m, 2H), 4.35~4.40(m, 1H), 9.71(s, 1H) |
| 86% |
With lead (IV) acetate In ethyl acetate at 20℃; |
|
| 85% |
With sodium (meta)periodate In tetrahydrofuran; water monomer for 4h; |
2; 1.1
NaIO4 (16 g, 74.8 mmol) was added to a stirred solution of (1) (D-mannitol bisacetonide) (20 g, 76.2 mmol) in 9:1 THF- H2O (280 mL), and the resultant mixture was stirred for 4 h. The produced precipitate was filtered off, and most of THF was evaporated under reduced pressure. Then water (20 mL) was added and the aqueous solution was extracted with dichloromethane (6 X 50 mL). The combined organic extracts were dried over MgSO4 and solvent was removed to afford (2) ((R)-2,2-Dimethyl-1,3-dioxolane-4-carbaldehyde)(16.8 g, 85% yield), which was used in the next step without further purification. Characterization of (2): 1H-NMR (250 MHz, CDCl3) 1.4 (s, 3H), 1.5 (s, 3H), 4.1 (m, 2H), 4.4 (m, 1H), 9.7 (d, 3JH-H = 2 Hz, 1H).IR (film): 3417, 2985, 1735, 1372, 1064 cm-1.appearence: colourless oil. |
| 84% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer at 35℃; for 2h; |
|
| 83% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer |
|
| 82% |
With lead tetraacetate |
|
| 82% |
With sodium (meta)periodate In dichloromethane; water monomer Ambient temperature; |
|
| 82% |
With NaIO4*SiO2 In dichloromethane at 20℃; for 1h; Inert atmosphere; |
|
| 81% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer for 3h; |
|
| 80% |
With sodium (meta)periodate; Sodium hydrogenocarbonate; magnesium(II) sulfate In dichloromethane for 0.166667h; |
|
| 78% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer 1.) 10 deg C, 30 min; 2.) 3 h, room temp.; |
|
| 77% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane |
|
| 77% |
With sodium (meta)periodate; mesoporous silica In dichloromethane at 0℃; for 0.5h; |
|
| 77% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 25℃; for 2h; |
|
| 76% |
With lead tetraacetate; potassium carbonate |
|
| 76% |
With lead tetraacetate; potassium carbonate In chloroform at 0 - 20℃; |
|
| 75% |
With sodium (meta)periodate; mesoporous silica In dichloromethane; water monomer at 20℃; for 0.5h; |
|
| 75% |
With sodium (meta)periodate In dichloromethane |
II Synthesis OF DIFLUORO ANALOGS OF LPA
Another approach to the synthesis of difluoromethylene analogs of LPA is depicted in Figure 5. Synthesis of the target LPA analogues 10a and lOb (Figure 5) involved non-reductive deprotection of the penultimate dimethyl phosphates 9 with trimethylsilane bromide to permit incorporation of unsaturated acyl chains. The key step for the synthesis was the introduction of the difluoromethyl group by the 1,1- DIFLUORINATION OF A C-1 aldehyde. Thus, commercially-available D-mannitol 1,2 : 5,6- bis-acetonide was OXIDATIVELY cleaved with NAI04 to afford the acetonide-protected D-glyceraldehyde 2. 10 Addition of (diethylamino) sulfur trifluoride (DAST) to a solution of the aldehyde 2 in CH2C12 afforded the difluorinated compounds in high yield after purification by distillation under reduced pressure. Next, acidic cleavage of the acetonide-protecting group provided the diol intermediate 4. The crude diol obtained after removal of the acetonide was immediately converted to the bis-silyl ether 5, and the more labile TBDMS ether of the primary alcohol was cleaved selectively by treatment with a solution of pyridinium hydrofluoride in a mixture of pyridine and THF at rt. Initial attempts to obtain the primary alcohol 6 from bis-TBDMS ether 5, utilizing 4.0 eq. of pyridinium hydrofluoride resulted in disappointing yields (17%) after 48 h at rt. However, increasing to 6.0 equiv. gave the primary alcohol in good yield (73%) after 20 h at rt. The primary alcohol 6 was then phosphorylated with dimethylphosphoryl chloride in the presence of t-BuOK to give good yield of phosphate 7. The 2-TBDMS ether was further deprotected with tetra (N-BUTYL) ammonium fluoride (TBAF) in THF to give alcohol 8 in 72% yield; neutralization of TBAF with acetic acid permitted desilyation of the secondary alcohol without the migration of phosphate. DCC-promoted ESTERIFICATION of alcohol S with oleic acid or palmitic acid provided good yield of esters 9a and 9b, respectively. Importantly, the introduction of the acyl groups at this stage circumvents problems with acyl group migration during other synthetic operations. Finally, treatment of protected phosphates 9 with bromotrimethylsilane and subsequent addition of 5% aq. methanol provided the desired DIFLUORINATED LPA analogues 10 in essentially quantitative yield.(R)-Glyceraldehyde acetonide (2) was prepared from D-MANNITOL-L, 2: 5,6-bis- acetonide as DESCRIBEDLO to give aldehyde 2 as a clear liquid: [A] 20D : + 64.4 (lit. 19 [AD + 64. 9). |
| 75% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer for 3h; |
|
| 75% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer at 20℃; for 2h; |
|
| 75% |
With sodium (meta)periodate; mesoporous silica In dichloromethane; water monomer at 20℃; for 1h; |
101.6 Step 6: (R)-2,2-Dimethyl-[l,3]dioxolane-4-carbaldehyde
Sodium periodate (57.0 g, 270 mmol) was dissolved in hot water (115 mL) and silica (200 g, 60 A 220-440 mesh, particle size 35-75 μιη) was added. The mixture was stirred vigorously until a free flowing powder was obtained. This was added to a solution of 1,2:5,6-bis-O-(1-methylethylidene)-D-mannitol (50 g, 190 mmol) in dichloromethane (1.0 L) and the reaction was stirred at room temperature for 1 hour. The resultant mixture was filtered through a pad of Na2SO4 and the solids washed thoroughly with dichloromethane. The combined organic extracts were evaporated in vacuo to yield 37.2 g (75%) of the title compound as a colorless oil. 1H NMR (400 MHz, CDC13) δ 9.73 (d, J = 1.9 Hz, 1H), 4.38 (ddd, 7 = 7.4, 4.7, 1.9 Hz, 1H), 4.18 (dd, J = 8.8, 7.4 Hz, 1H), 4.10 (dd, J = 8.8, 4.7 Hz, 1H), 1.49 (s, 3H), 1.43 (s, 3H). |
| 75% |
With sodium (meta)periodate In dichloromethane; water monomer at 20℃; for 1h; Molecular sieve; |
106.1 Step 1: (R)-2,2-Dimethyl-[1,3]dioxolane-4-carbaldehyde
Sodium periodate (57.0 g, 270 mmol) was dissolved in hot water (115 mL) and silica (200 g, 60 220-440 mesh, particle size 35-75 μm) was added. The mixture was stirred vigorously until a free flowing powder was obtained. This was added to a solution of 1,2:5,6-bis-O-(1-methylethylidene)-D-mannitol (50 g, 190 mmol) in dichloromethane (1.0 L) and the reaction was stirred at room temperature for 1 hour. The resultant mixture was filtered through a pad of Na2SO4 and the solids washed thoroughly with dichloromethane. The combined organic extracts were evaporated in vacuo to yield 37.2 g (75%) of the title compound as a colorless oil. 1H NMR (400 MHz, CDCl3) δ 9.73 (d, J=1.9 Hz, 1H), 4.38 (ddd, J=7.4, 4.7, 1.9 Hz, 1H), 4.18 (dd, J=8.8, 7.4 Hz, 1H), 4.10 (dd, J=8.8, 4.7 Hz, 1H), 1.49 (s, 3H), 1.43 (s, 3H). |
| 73% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 0℃; for 2h; Inert atmosphere; |
|
| 72% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane for 2h; |
|
| 72% |
With N-Bromosuccinimide; water monomer; potassium carbonate In acetonitrile var. of reagent, amount of catalyst, time; |
|
| 70% |
With sodium (meta)periodate; mesoporous silica In dichloromethane; water monomer at 20℃; for 0.166667h; |
|
| 67% |
With sodium (meta)periodate; water monomer; Sodium hydrogenocarbonate In dichloromethane at 30℃; for 4.33333h; |
|
| 67% |
With sodium (meta)periodate; Sodium hydrogenocarbonate for 1.5h; Inert atmosphere; |
|
| 65% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer |
|
| 65% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer at 20℃; for 2h; |
|
| 63% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane |
|
| 62% |
With N-(tert-butyl)-N',N',N",N"-tetramethylguanidinium meta-iodoxybenzoate In dichloromethane at 40℃; for 2.5h; |
|
| 61% |
With sodium hydroxide; sodium (meta)periodate In water monomer for 1.5h; pH = 7.0; |
|
| 59% |
With lead tetraacetate |
|
| 55% |
With potassium metaperiodate; Sodium hydrogenocarbonate In dichloromethane at 25 - 30℃; for 2h; |
|
| 55% |
With sodium (meta)periodate In dichloromethane |
|
| 52% |
With N-Bromosuccinimide; triphenylbismuthine; potassium carbonate In water monomer; acetonitrile for 6h; Ambient temperature; |
|
| 45% |
With lead tetraacetate In ethyl acetate at 25℃; for 2.5h; |
|
| 42% |
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer at 22℃; for 1.5h; |
|
| 40.1% |
With sodium (meta)periodate; Potassium bicarbonate In tetrahydrofuran; water monomer at 18℃; for 2.5h; Cooling with ice; |
2 Example 2
Was added to a 250 mL round bottom flaskNaIO4 (15 g, 70 mmol),Water (80 mL) was stirred, and KHCO3 (14 g, 140 mmol) was added in portions under ice-cooling,A solution of compound 12 (17.75 g, 70 mmol) in THF (55 mL) was added dropwise,30min drops finished, remove the ice bath,18 reaction 2h,Suction filtration turbid solution,Extracted three times with 20 mL of ethyl acetate,Merge several layers,Dried over anhydrous magnesium sulfate,Steamed out of the solvent to light yellowish oily liquid,Vacuum distillation65 ° C fractions,To give 7.135 g of a colorless oily liquid,The yield was 40.1%. |
|
With lead tetraacetate; benzene |
|
|
With lead tetraacetate In benzene for 1.5h; Ambient temperature; |
|
| 60 mg |
With A-26 resin (IO4(1-)) In water monomer at 20℃; for 2h; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer for 1h; Ambient temperature; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer at 0 - 5℃; for 1h; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer for 1h; Ambient temperature; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer for 1h; Ambient temperature; |
|
|
With lead tetraacetate In ethyl acetate Ambient temperature; |
|
|
With lead tetraacetate In tetrahydrofuran at 15℃; for 2h; |
|
|
With lead tetraacetate In toluene |
|
|
With lead tetraacetate In tetrahydrofuran 1.) 0-4 deg C, 30 min, 2.) RT, 30 min; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer at 25℃; for 0.5h; |
|
|
With sodium (meta)periodate In water monomer at 5 - 10℃; for 1h; |
|
|
With sodium (meta)periodate In water monomer for 1.5h; Ambient temperature; pH 6-7; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In methanol; water monomer at 22℃; |
|
|
With sodium (meta)periodate In methanol; water monomer for 0.5h; Ambient temperature; |
|
|
With phosphoric acid disodium salt; sodium periodate In water monomer for 0.25h; Ambient temperature; |
|
|
With lead tetraacetate |
|
|
With sodium (meta)periodate In water monomer for 0.5h; |
|
|
With lead tetraacetate In benzene for 0.333333h; |
|
|
With sodium (meta)periodate In water monomer for 0.166667h; ice-cooling; |
|
|
With lead tetraacetate In benzene Ambient temperature; |
|
|
With lead tetraacetate In tetrahydrofuran at 15℃; for 1.5h; |
|
|
With lead tetraacetate In tetrahydrofuran |
|
|
With sodium (meta)periodate In methanol; water monomer |
|
|
With lead tetraacetate In ethyl acetate for 1h; Ambient temperature; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate at 0℃; for 3h; |
|
|
With sodium (meta)periodate In tetrahydrofuran; water monomer for 1h; |
|
|
With lead tetraacetate In ethyl acetate |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In methanol; water monomer at 20℃; for 4h; ultrasonic irradiation; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane at 20℃; for 5h; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In water monomer at 20℃; for 1h; |
|
|
With sodium (meta)periodate |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer for 2h; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In methanol; water monomer at 0℃; for 1h; |
|
|
With sodium (meta)periodate; Sodium hydrogenocarbonate In dichloromethane; water monomer at 20℃; for 3h; |
|
|
With sodium (meta)periodate |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping