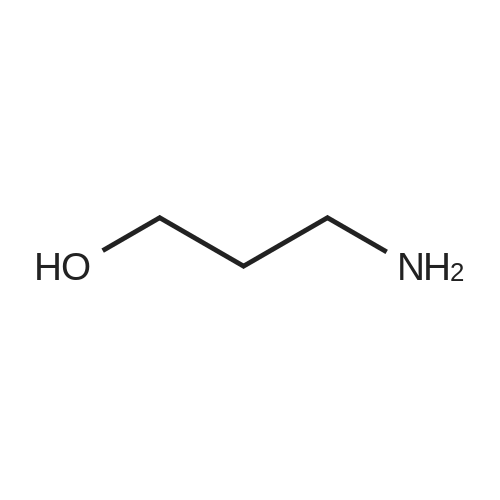
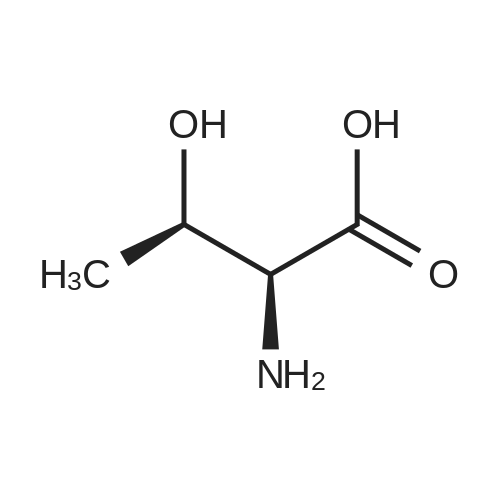
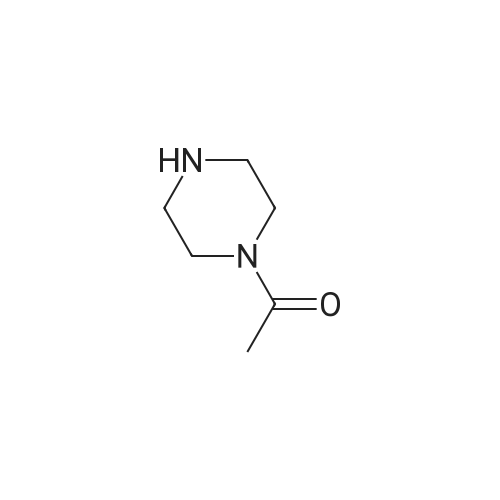
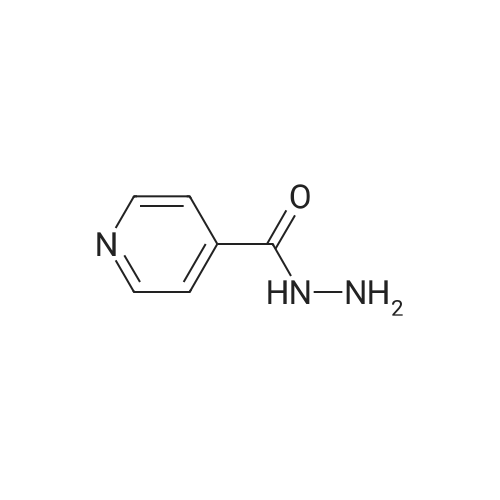
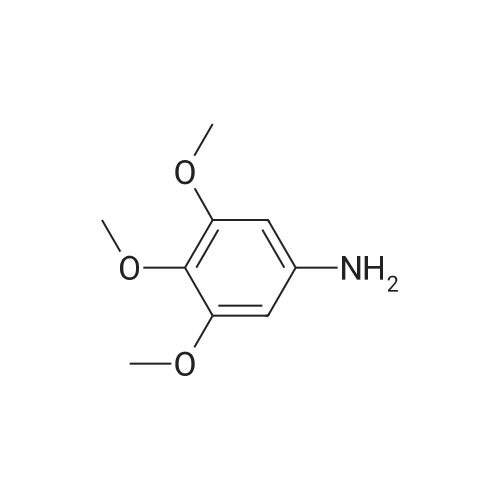
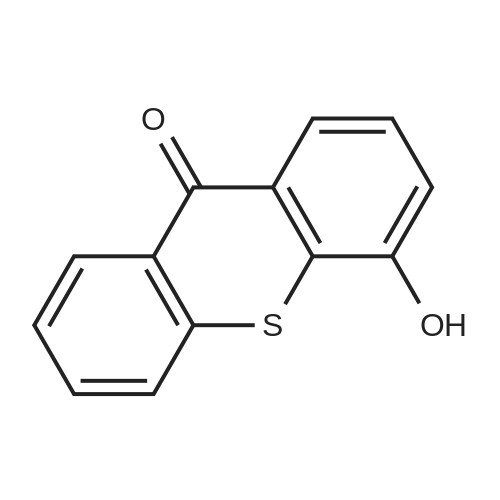
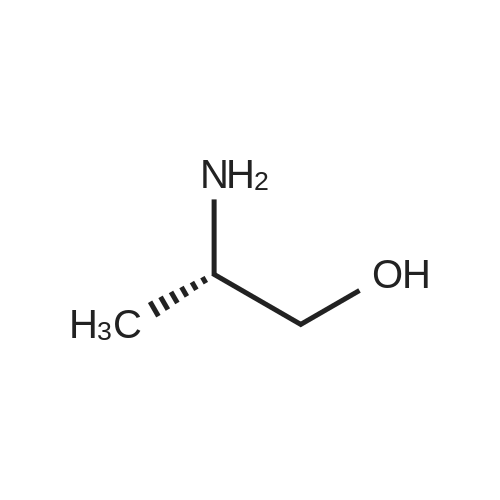
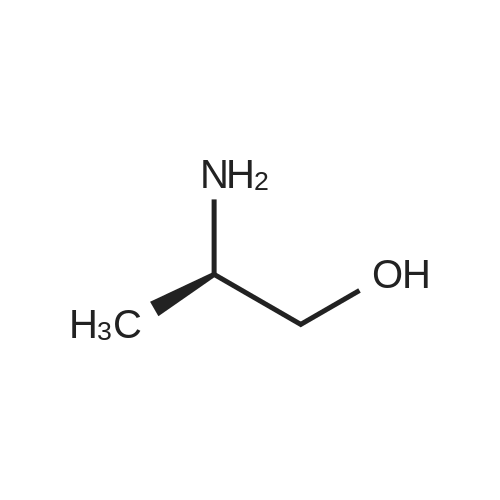
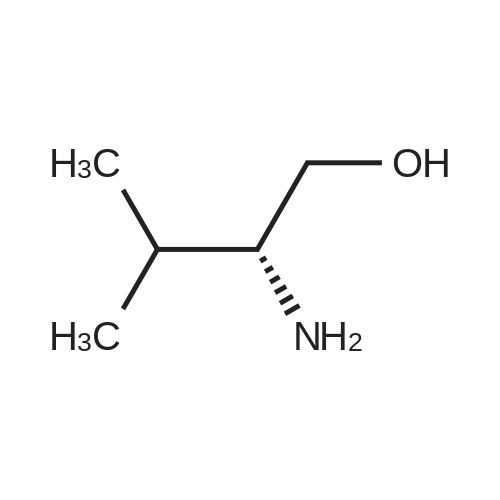
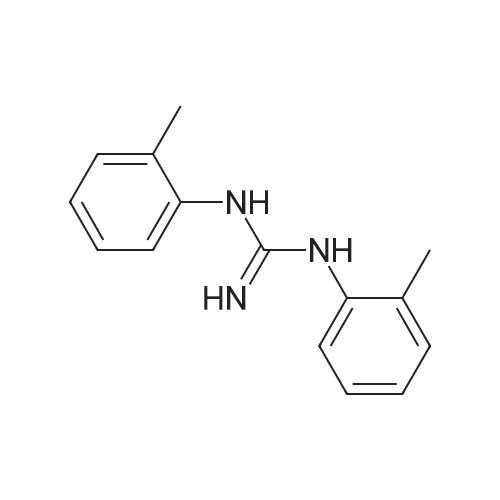
| 1.97% |
With copper(l) iodide; potassium carbonate; In methanol; at 100℃; for 48h; |
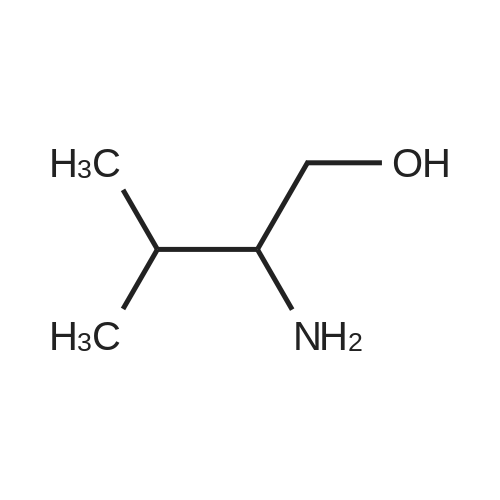
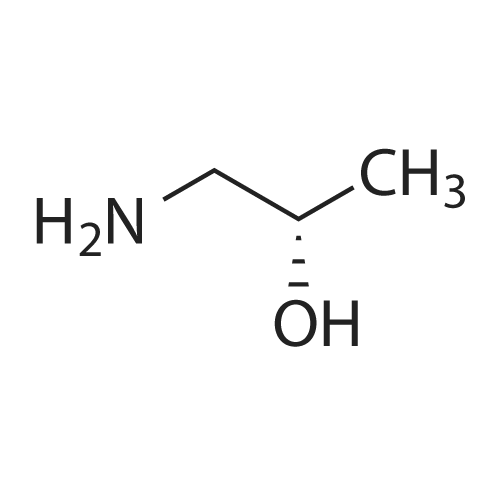
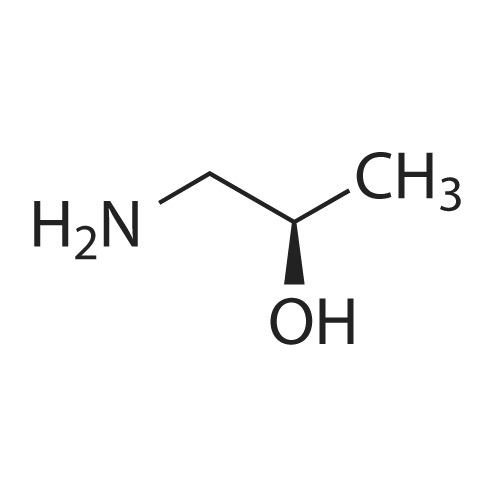
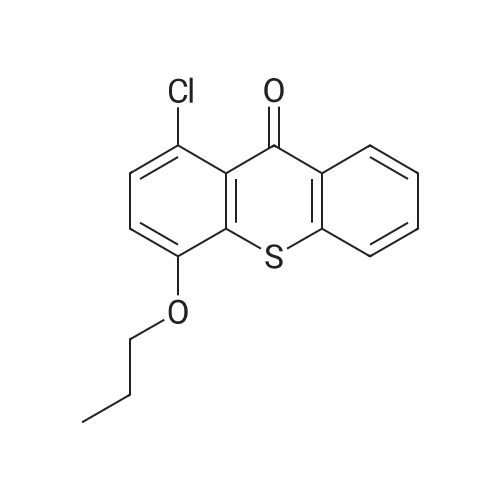
General procedure: 1-Chloro-4-propoxy-9H-thioxanthen-9-one (Tx, 450 mg, 1.48 mmol) and a suitable chiral aminoalcohol (9-16, 1.72 mmol) were dissolved in methanol (30 mL) and CuI (0.15 mmol) and K2CO3(1.92 mmol) were added. The reaction mixture was heated at 100 C in a muffle furnace for 48 h.After the completion of the reaction, the crude material was filtrated, washed with dichloromethane,and the organic solvents were evaporated under reduced pressure. Then, the obtained solid wasdissolved in 50 mL of dichloromethane and extracted with HCl 1M(3 50 mL). The aqueous layer wasbasified with NaOH 20% and extracted with dichloromethane (3 100 mL). The organic layers weregathered, washed with water (3 50 mL), dried over anhydrous sodium sulphate and the solvent wasevaporated under reduced pressure. Then, a solid phase extraction using a cation exchange cartridgeDiscovery DSC-SCX was applied to further purify the extracted material. First, an activation ofthe cartridge with dichloromethane (50 mL) was performed followed by loading the cartridge withthe sample (previously incorporated in silica). Then, elution was carried out with the followingsolvents: dichloromethane, a mixture of dichloromethane/methanol 5:5 (v/v), methanol 100% andNH3 2% in methanol. The fractions containing the chiral ATxs were gathered and the solvent wasevaporated under reduced pressure. A flash column chromatography with n-hexane/ethyl acetate ingradient and crystallization from chloroform and petroleum ether (4:1) were also performed to obtainpure compounds.(S)-1-((1-Hydroxypropan-2-yl)amino)-4-propoxy-9H-thioxanthen-9-one (ATx 1 (+)). Yield: 1.97%. m.p.:116-118 C (dec.); [alpha]25D +112 (c = 3.4 103 g/mL in dichloromethane). IR (KBr) numax: 3419, 3270,2962, 2925, 2873, 2359, 2341, 1618, 1568, 1507, 1435, 1293, 1269, 1252, 1225, 746 cm1. 1H-NMR(300.13 MHz, CDCl3): delta: 8.50 (1H, d, J = 8.0 Hz, H-8), : 7.56 (2H, m, H-5 and H-6), : 7.43 (1H, m, H-7),: 7.13 (1H, d, J = 9.0 Hz, H-3), : 6.85 (1H, d, J = 9.0 Hz, H-2), : 4.03 (2H, t, J = 6.5 Hz, H-a), : 3.81 (3H,m, H-10 and CH2OH), : 1.90 (2H, m, H-b), : 1.32 (3H, d, J = 6.2 Hz, H-20), : 1.12 (3H, t, J = 7.4 Hz,H-c). 13C-NMR (75.47 MHz, CDCl3): delta: 183.55 (C-9), 146.50 (C-4), 144.02 (C-1), 136.85 (C-6), 131.92(C-8a), 129.85 (C-4a), 129.24 (C-9a), 126.08 (C-8), 125.98 (C-7), 125.20 (C-5), 119.12 (C-3), 114.10 (C-10a),109.80 (C-2), 72.48 (C-a), 65.96 (C-CH2OH), 51.91 (C-10), 22.66 (C-b), 16.86 (C-20), 10.71 (C-c). e.e. > 99%(HPLC; column: Lux Amylose-1 (250 4.6 mm i.d., 5 m), Mobile phase: n-hexane:ethanol (70:30v/v), 0.5 mL/min, max 254 nm). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping