| 12.6% |
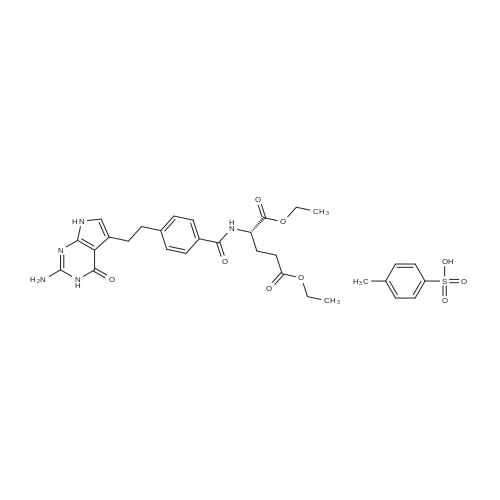
Stage #1: (2S)-2-[4-[2-(2-amino-4-oxo-1,7-dihydropyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]amino}pentanedioic acid With 1-hydroxy-pyrrolidine-2,5-dione; N-[3-(N,N-dimethylamino)-propyl]-N'-ethyl-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In (methylsulfinyl)methane; N,N-dimethyl-formamide at 20℃; for 0.5h;
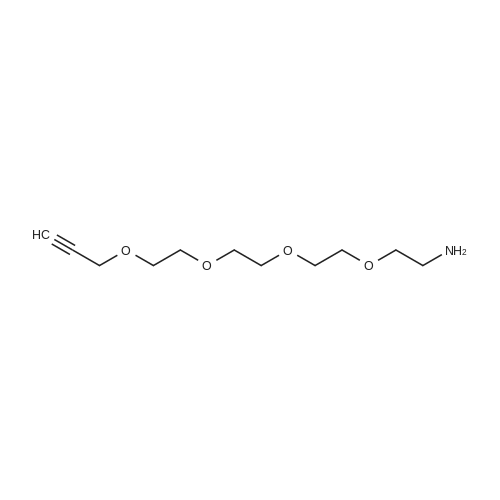
Stage #2: 3,6,9,12-tetraoxapentadec-14-yn-1-amine at 20℃; for 16h; |
32 (S)-18-(4-(2-(2-amino-4-oxo-4,7-dihydro-1H-pyrrolo[2,3-d]pyrimidin-5- yl)ethyl)benzamido)-1-(1-(5-((5-(7-chloro-8-(((R)-1-(5-cyano-2-fluorophenyl)ethyl)amino)-3- fluoro-6-methyl-1,5-naphthyridin-2-yl)pyrimidin-2-yl)oxy)pentyl)-1H-1,2,3-triazol-4-yl)-15-oxo- 2,5,8,11-tetraoxa-14-azanonadecan-19-oic acid
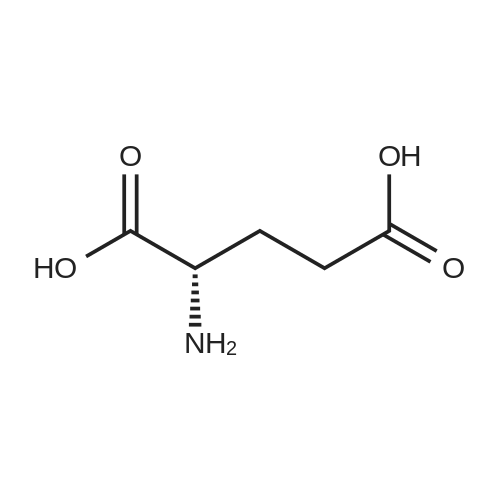
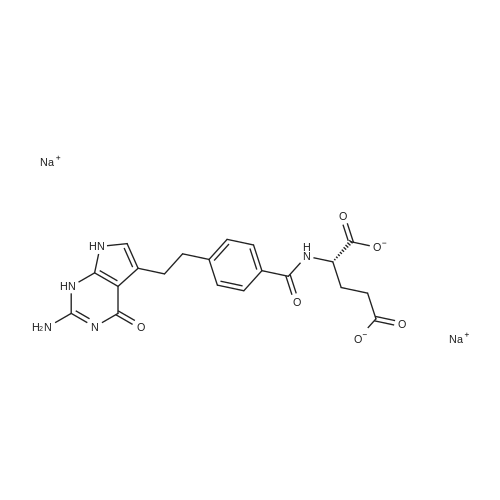
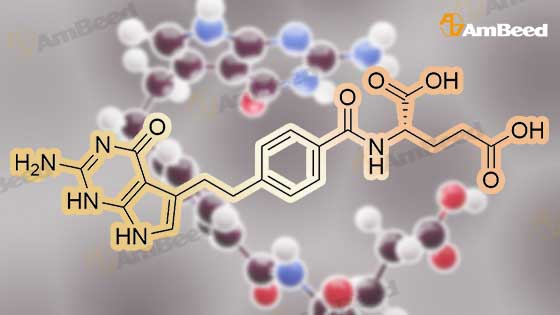
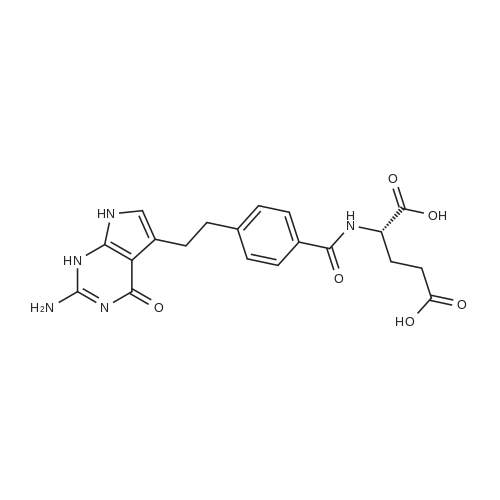
To a solution of (4-(2-(2-amino-4-oxo-4,7-dihydro-1H-pyrrolo[2,3-d]pyrimidin-5- yl)ethyl)benzoyl)-L-glutamic acid (Pemetrexed) (1, 1.0 eq, 0.500 g, 1.17 mmol) in N,N- dimethylformamide (10 mL) and dimethyl sulfoxide (10 mL), N-hydroxysuccinimide (1.1 eq, 0.148 g, 1.29 mmol), 1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC.HCl) (1.1 eq, 0.247 g, 1.29 mmol) and N,N-diisopropylethylamine (3.0 eq, 0.64 mL, 3.51 mmol) were added and the reaction mixture was stirred at room temperature for 30 minutes. Then, 3,6,9,12-tetraoxapentadec- 14-yn-1-amine (1a, 1.1 eq, 0.298 g, 1.29 mmol) was added and the reaction mixture was stirred at room temperature for 16 h. After completion, the reaction mixture was directly purified by prep HPLC (20-55% acetonitrile in water with 0.1% acetic acid). Fractions containing the desired product were combined and lyophilized to dryness to afford (S)-20-(4-(2-(2-amino-4-oxo-4,7-dihydro-1H- pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzamido)-17-oxo-4,7,10,13-tetraoxa-16-azahenicos-1-yn-21-oic acid (2) as a yellow sticky solid. Yield: 0.100 g, 12.6%; LCMS m/z 641.2 [M+1]+ |
| 12.6% |
Stage #1: (2S)-2-[4-[2-(2-amino-4-oxo-1,7-dihydropyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]amino}pentanedioic acid With 1-hydroxy-pyrrolidine-2,5-dione; N-[3-(N,N-dimethylamino)-propyl]-N'-ethyl-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In (methylsulfinyl)methane; N,N-dimethyl-formamide at 20℃; for 0.5h;
Stage #2: 3,6,9,12-tetraoxapentadec-14-yn-1-amine at 20℃; for 16h; |
32 (S)-18-(4-(2-(2-amino-4-oxo-4,7-dihydro-1H-pyrrolo[2,3-d]pyrimidin-5- yl)ethyl)benzamido)-1-(1-(5-((5-(7-chloro-8-(((R)-1-(5-cyano-2-fluorophenyl)ethyl)amino)-3- fluoro-6-methyl-1,5-naphthyridin-2-yl)pyrimidin-2-yl)oxy)pentyl)-1H-1,2,3-triazol-4-yl)-15-oxo- 2,5,8,11-tetraoxa-14-azanonadecan-19-oic acid
To a solution of (4-(2-(2-amino-4-oxo-4,7-dihydro-1H-pyrrolo[2,3-d]pyrimidin-5- yl)ethyl)benzoyl)-L-glutamic acid (Pemetrexed) (1, 1.0 eq, 0.500 g, 1.17 mmol) in N,N- dimethylformamide (10 mL) and dimethyl sulfoxide (10 mL), N-hydroxysuccinimide (1.1 eq, 0.148 g, 1.29 mmol), 1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC.HCl) (1.1 eq, 0.247 g, 1.29 mmol) and N,N-diisopropylethylamine (3.0 eq, 0.64 mL, 3.51 mmol) were added and the reaction mixture was stirred at room temperature for 30 minutes. Then, 3,6,9,12-tetraoxapentadec- 14-yn-1-amine (1a, 1.1 eq, 0.298 g, 1.29 mmol) was added and the reaction mixture was stirred at room temperature for 16 h. After completion, the reaction mixture was directly purified by prep HPLC (20-55% acetonitrile in water with 0.1% acetic acid). Fractions containing the desired product were combined and lyophilized to dryness to afford (S)-20-(4-(2-(2-amino-4-oxo-4,7-dihydro-1H- pyrrolo[2,3-d]pyrimidin-5-yl)ethyl)benzamido)-17-oxo-4,7,10,13-tetraoxa-16-azahenicos-1-yn-21-oic acid (2) as a yellow sticky solid. Yield: 0.100 g, 12.6%; LCMS m/z 641.2 [M+1]+ |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping