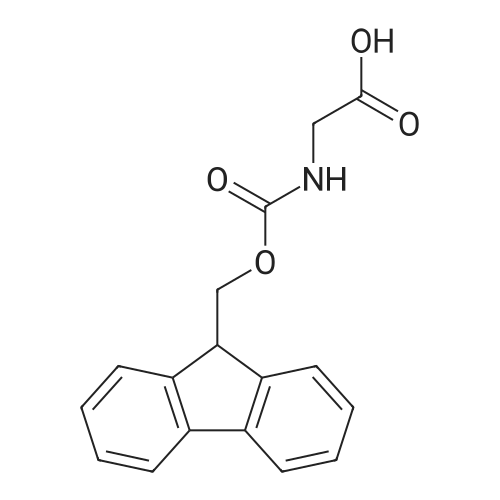
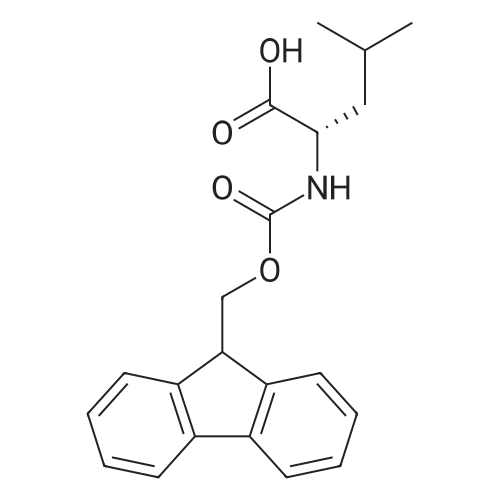
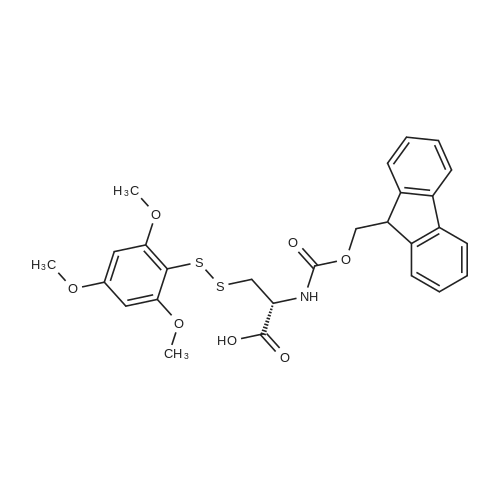
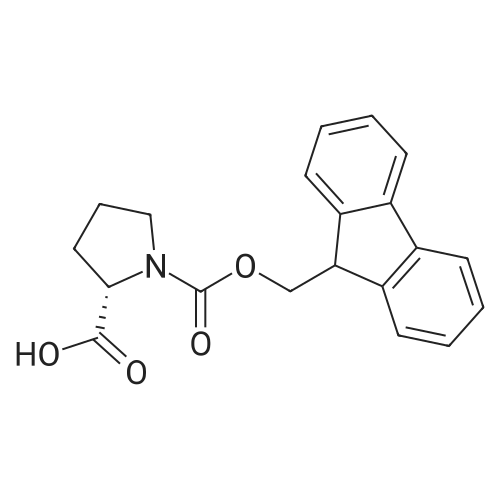
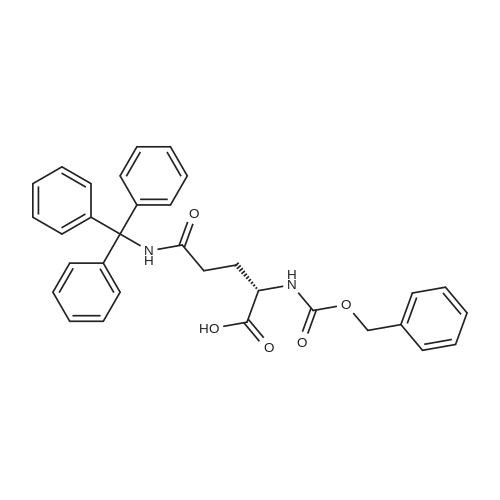
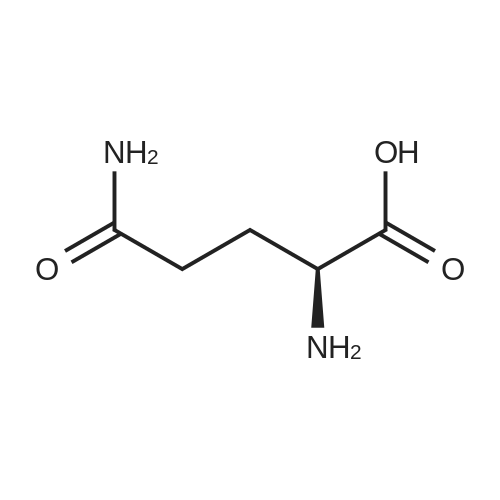
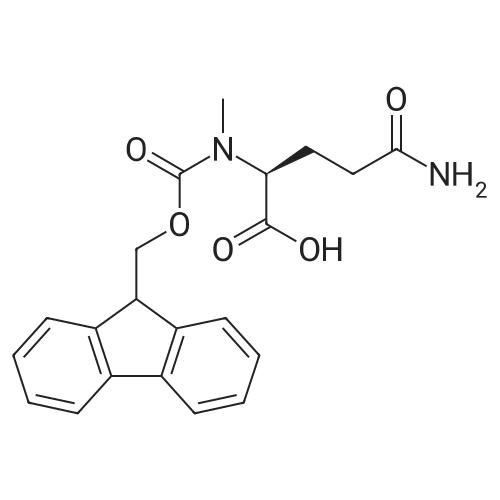
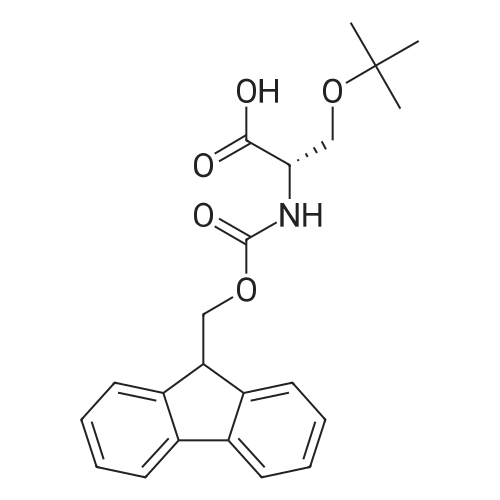
|
|
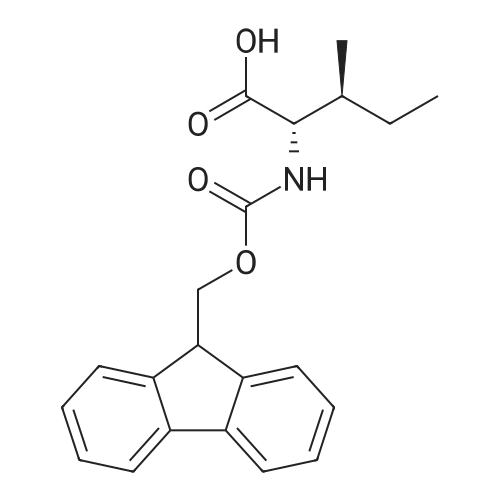
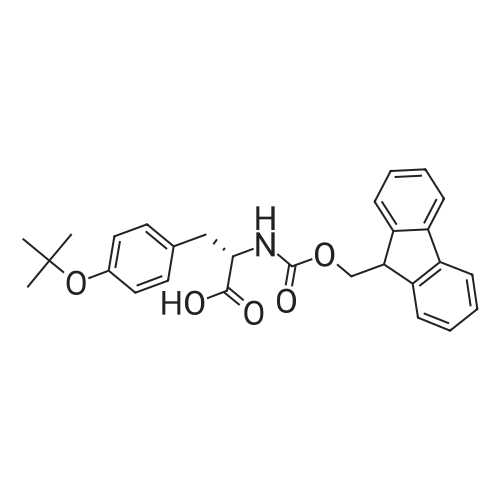
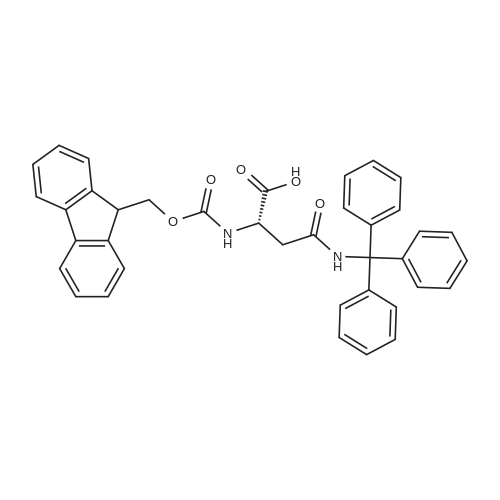
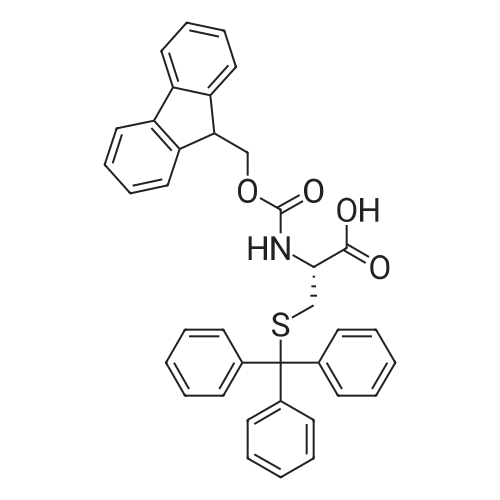
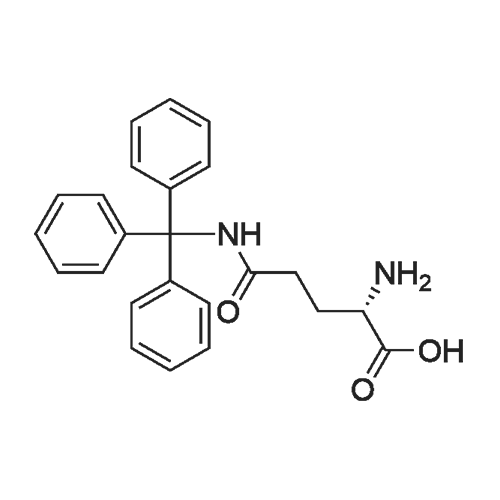
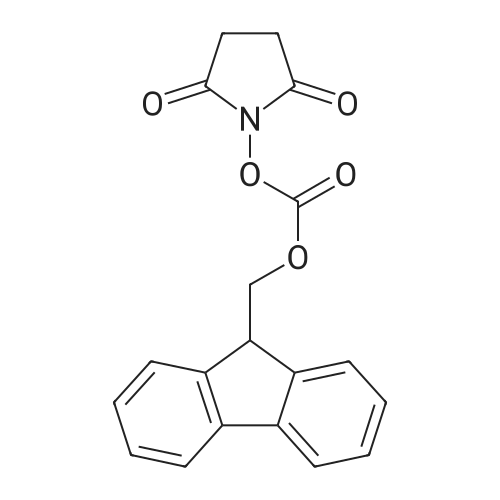
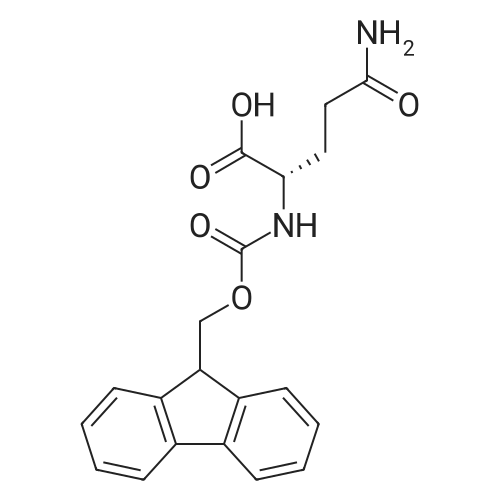
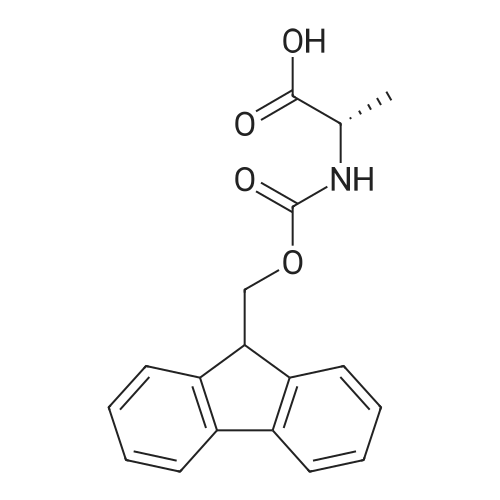
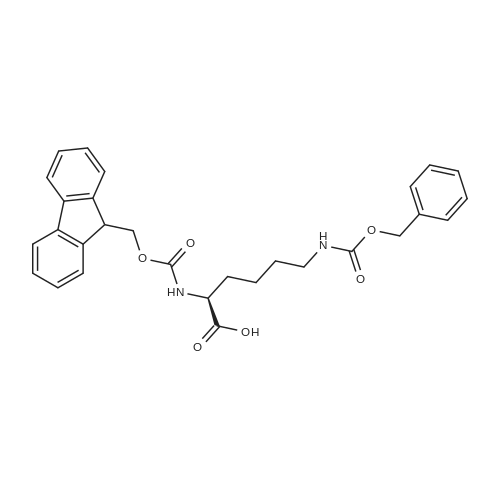
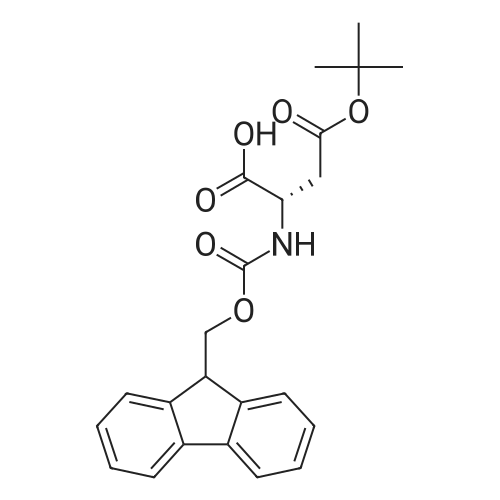
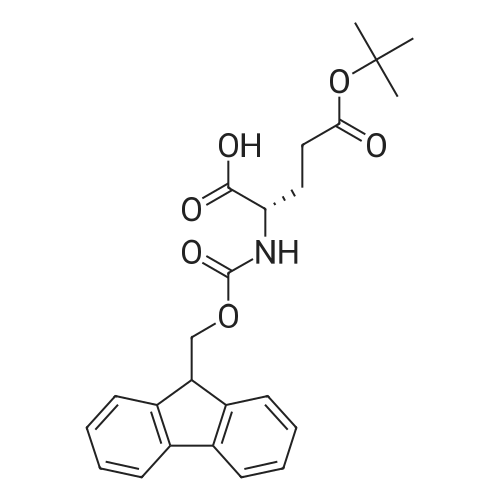
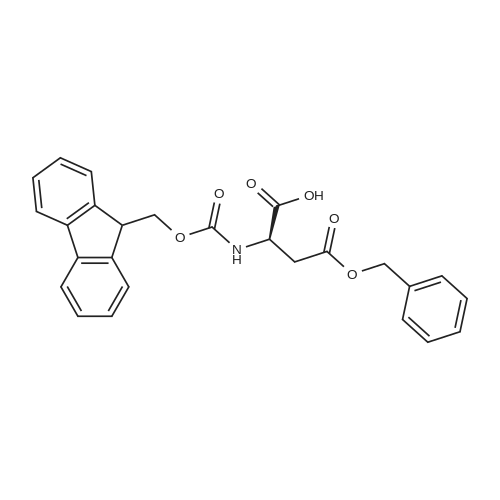
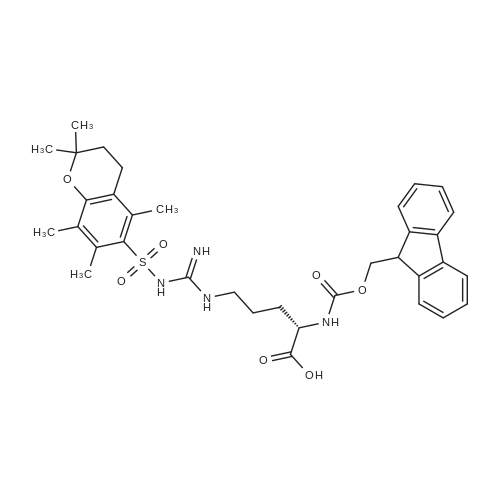
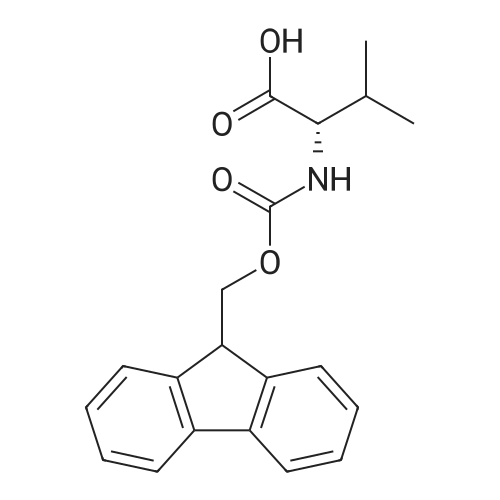
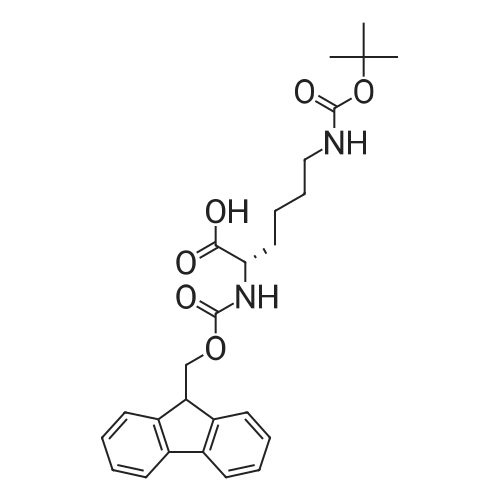
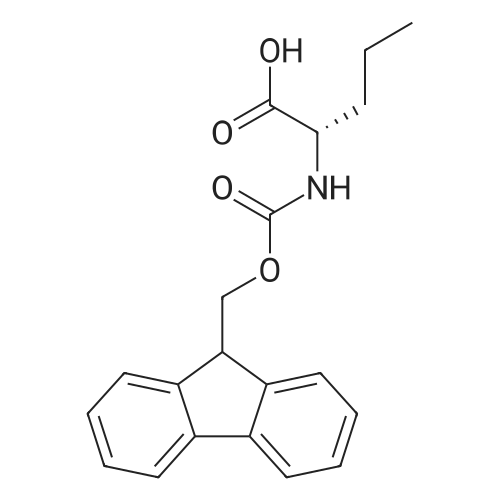
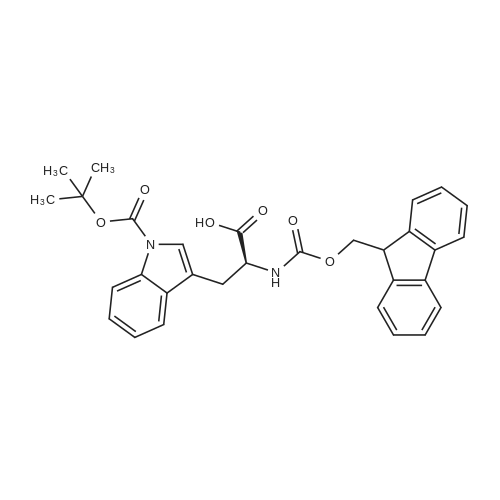
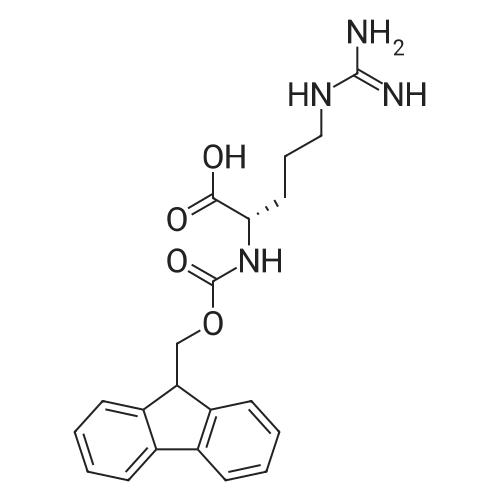
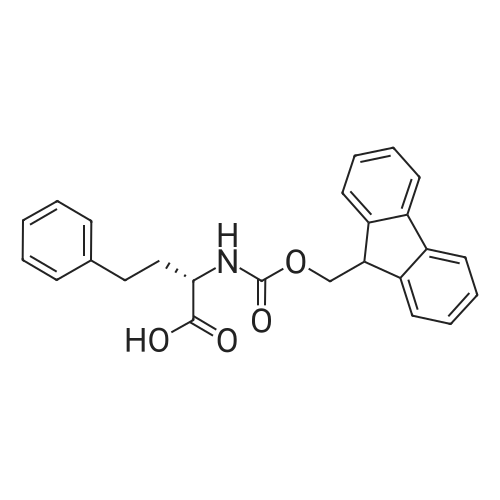
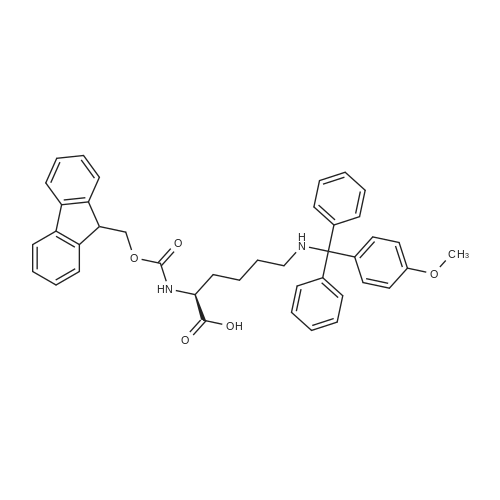
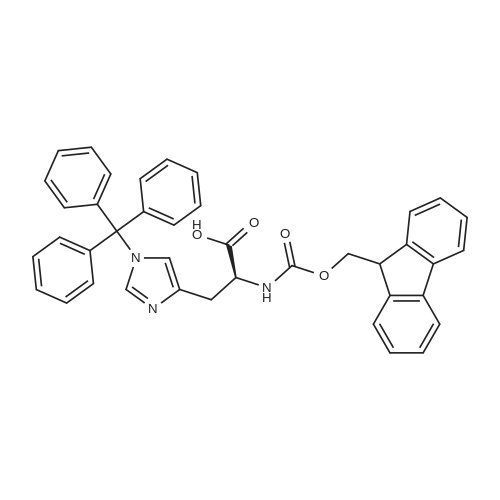
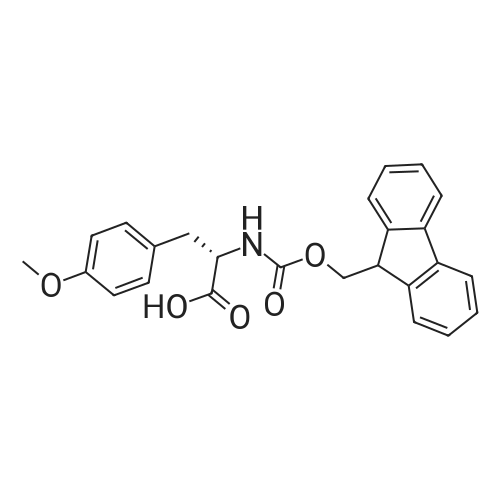
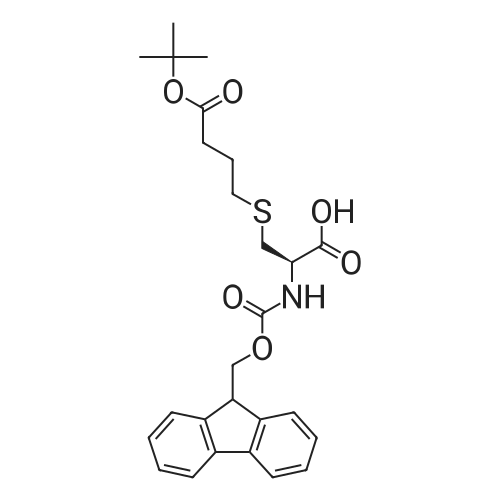
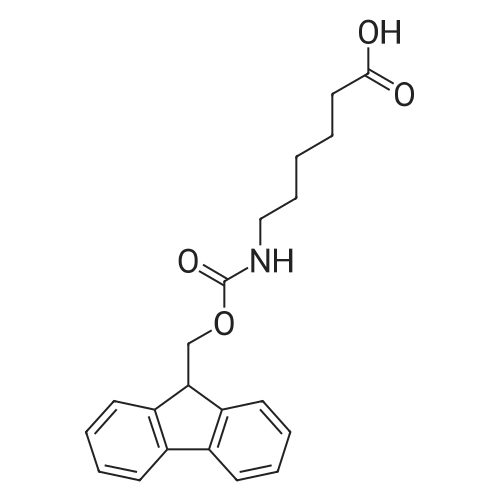
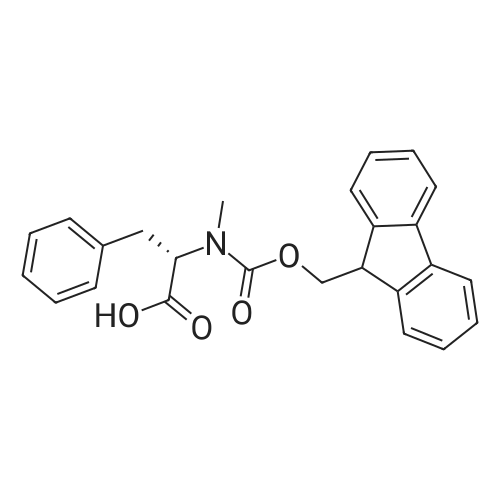
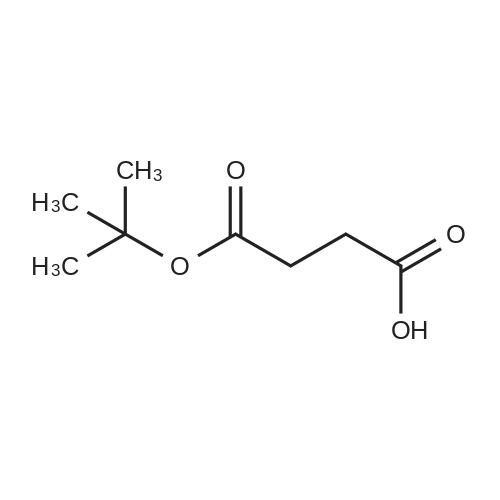
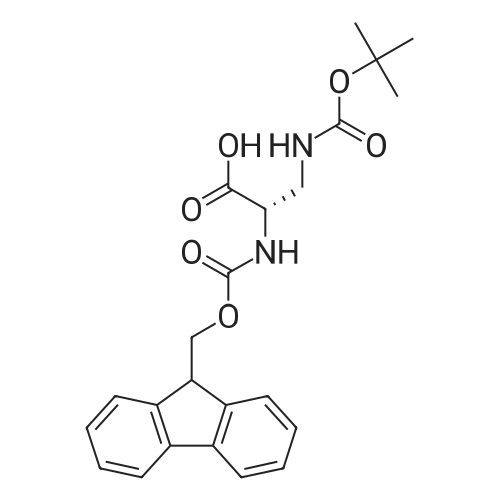
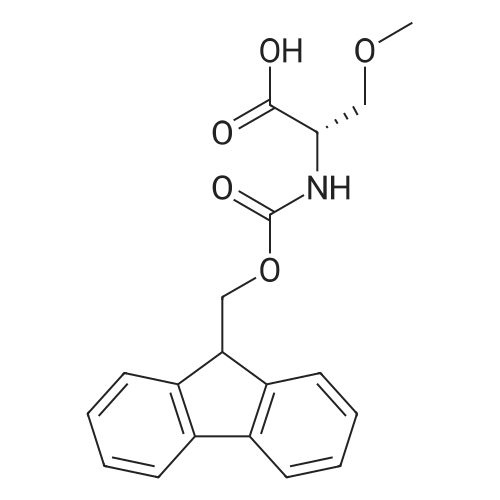
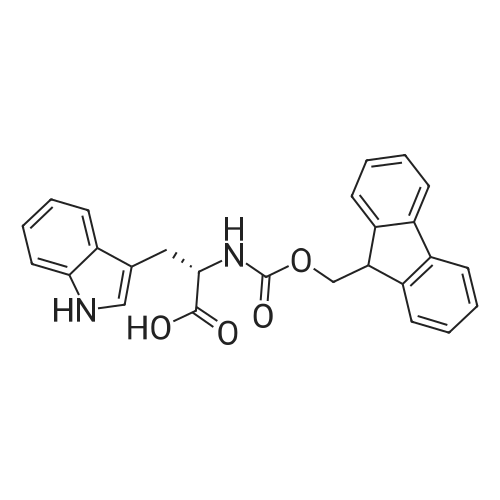
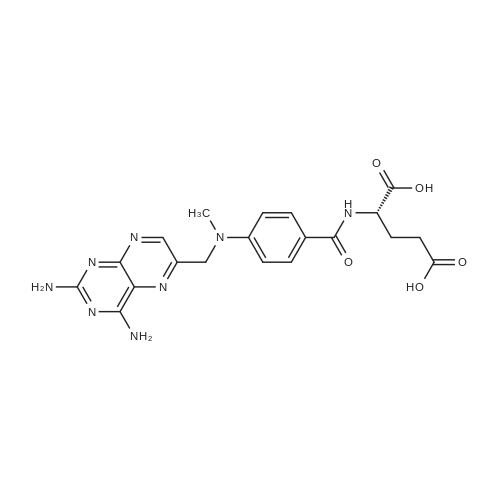
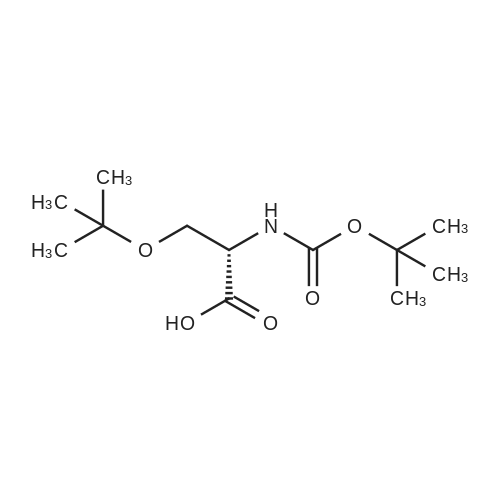
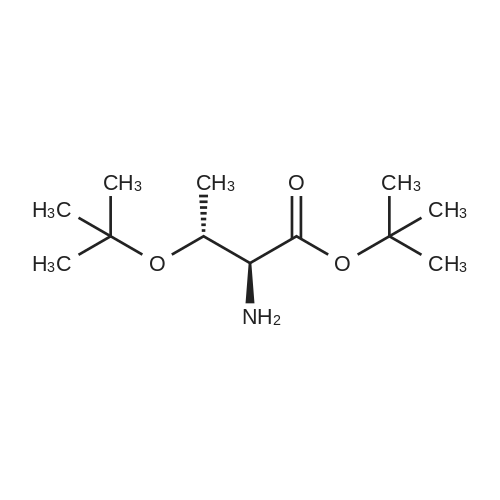
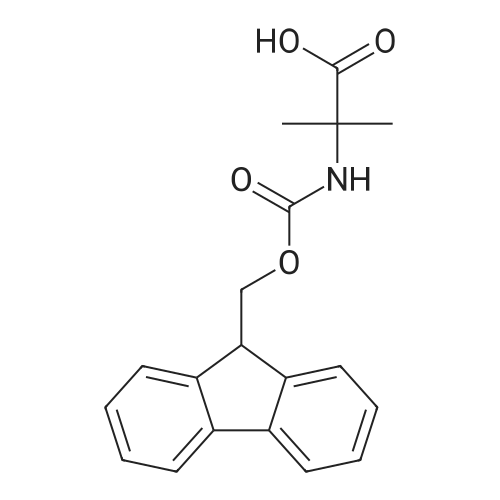
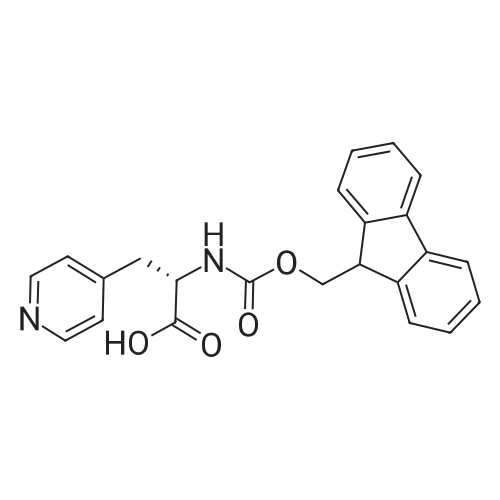
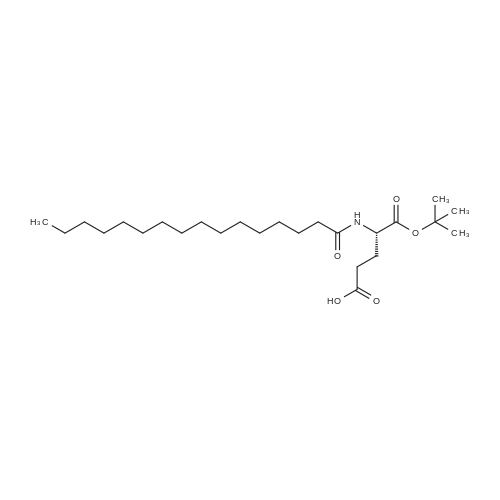
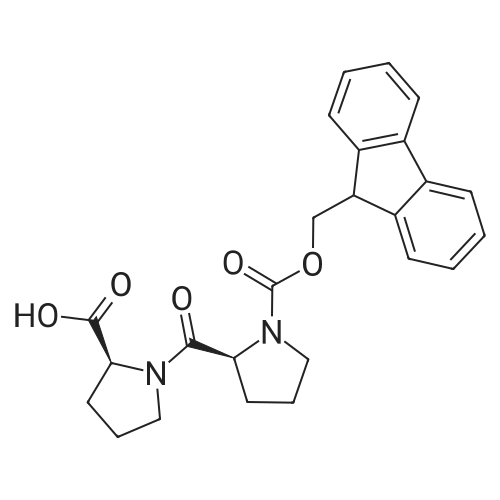
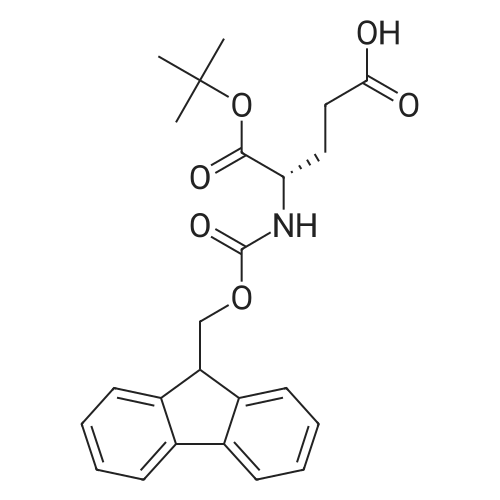
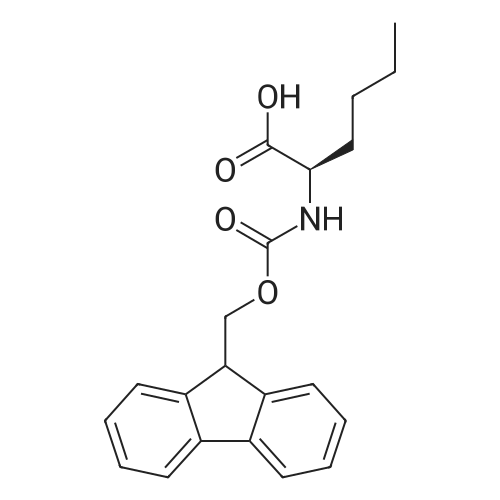
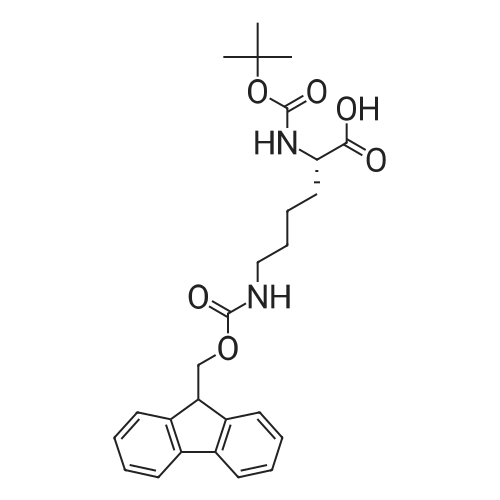
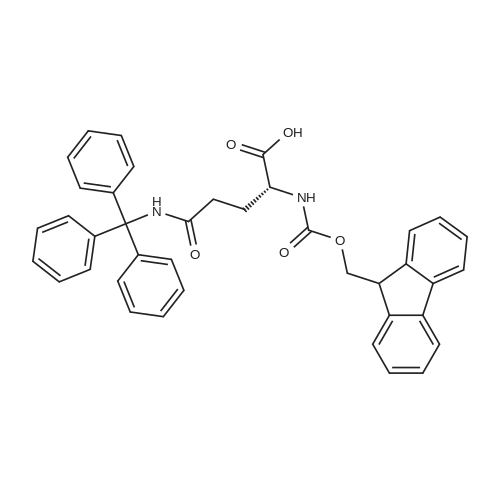
The peptide component of Compound 1 is synthesized by automated solid-phase synthesis using Fluorenylmethyloxycarbonyl (Fmoc)/tert-Butyl (t-Bu) chemistry on a Symphony 12-channel multiplex peptide synthesizer (Protein Technologies, Inc. Tucson, Ariz.). The synthesis resin consists of 1% DVB cross-linked polystyrene (Fmoc-Rink-MBHA Low Loading resin, 100-200 mesh, EMD Millipore, Temecula, Calif.) at a substitution 0.3-0.4 meq/g. Standard side-chain protecting groups are as follows: tert-butyloxycarbonyl (Boc) for Trp and Lys; tert-butyl ester (OtBu) for Asp and Glu; tBu for Ser, Thr and Tyr; and triphenylmethyl (Trt) for Gln; N-alpha-Fmoc-N--4-methyltrityl-L-lysine (Fmoc-Lys(Mtt)-OH) was used for the lysine at position 20 of SEQ ID NO: 3 and Nalpha,N(im)-di-Boc-L-histidine (Boc-His(Boc)-OH) was used for the histidine at position 1. Fmoc groups were removed prior to each coupling step (2×7 minutes) using 20% piperidine in dimethylformamide (DMF). All standard amino acid couplings are performed for 1 hour, using an equal molar ratio of Fmoc amino acid (EMD Millipore, Temecula, Calif.), diisopropylcarbodiimide (DIC)(Sigma-Aldrich, St. Louis, Mo.) and Oxyma (Oxyma Pure, Iris Biotech, Marktredwitz, Germany), at a 9-fold molar excess over the theoretical peptide loading and at a final concentration of 0.18 M in DMF. Two exceptions are the glutamine residue at position 3 of SEQ ID NO: 5, which is double-coupled (2×1 hour), and the histidine residue at position 1 of SEQ ID NO: 5, which was coupled at a 6-fold molar excess using 1-Hydroxy-7-azabenzotriazole (HOAt) instead of Oxyma for 18 hours. After completion of the synthesis of the linear peptide, the resin was transferred to a disposable fritted 25 mL polypropylene syringe (Torviq, Niles, Mich.) equipped with a polytetrafluoroethylene (PTFE) stopcock (Biotage, Charlotte, N.C.) and the 4-Methyltrityl (Mtt) protecting group on the lysine at position 20 of SEQ ID NO: 5 was selectively removed from the peptide resin using three treatments with 20% hexafluoroisopropanol (Oakwood Chemicals, West Columbia, S.C.) in DCM (2×10 minutes and 1×45 minutes) to expose the free epsilon amine of the lysine at position 20 and make it available for further reaction. Subsequent attachment of the fatty acid-linker moiety is accomplished by performing two succeeding couplings of [2-(2-(Fmoc-amino)ethoxy)ethoxy]acetic acid (Fmoc-AEEA-OH) (ChemPep, Inc. Wellington, Fla.; 3-fold excess of amino acid (AA):1-[Bis(dimethylamino)methylene]-1H-1,2,3-triazolo[4,5-b]pyridinium-3-oxid hexafluorophosphate (HATU): N,N-diisopropylethylamine (DIPEA) [1:1:5 mol/mol] for a 3 hour coupling time), followed by coupling of Fmoc-glutamic acid alpha-t-butyl ester (Fmoc-Glu-OtBu)(Ark Pharm, Inc. Libertyville Ill., 3-fold excess of AA:HATU:DIPEA [1:1:5 mol/mol] for a 3 hour coupling time). In each case, the Fmoc moiety is removed as described above. Finally, mono-OtBu-octadecanedioic acid (WuXi AppTec, Shanghai, China) is coupled to the resin over 18 hours using a 3-fold excess of acid:HATU:DIPEA (1:1:5 mol/mol). After the synthesis is complete, the peptide resin is washed with dichloromethane (DCM), diethyl ether and thoroughly air dried by applying vacuum suction to the syringe for 5 minutes. The dry resin is treated with a cleavage cocktail (trifluoroacetic acid (TFA): anisole: water: triisopropylsilane, 88:5:5:2 v/v) for 2 hours at room temperature to release the peptide from the solid support and remove all side-chain protecting groups. The resin is filtered off, washed twice with neat TFA, and the combined filtrates are treated with cold diethyl ether to precipitate the crude peptide. The peptide/ether suspension is then centrifuged at 4000 rpm to form a solid pellet, the supernatant is decanted, and the solid pellet is triturated with ether two additional times and dried in vacuo. The crude peptide is solubilized in 20% acetonitrile/water and purified by RP-HPLC on a C8 preparative column (Luna 21×250 mm, Phenomenex, Torrance, Calif.) with linear gradients of acetonitrile and water using three different buffer systems: 1) 0.1 M ammonium acetate in water, pH 5.0; 2) 0.1% TFA in water; and 3) 5% acetic acid in water. Subsequent lyophilization of the final main product pool yields the lyophilized peptide acetate salt. In a synthesis performed essentially as described above, the purity of Compound 1 is assessed using analytical RP-HPLC and found to be >97%. The molecular weight is determined by analytical electrospray MS. The molecular weight of Compound 1 is calculated to be 4535.0 Daltons while the observed deconvoluted averaged molecular weight was determined to be 4535.0 Daltons and the following ions were observed: 1512.3 (M+3H), 1134.3 (M+4H), 908 (M+5H). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping