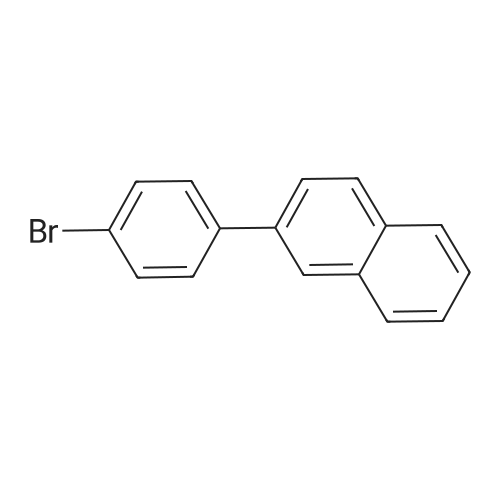
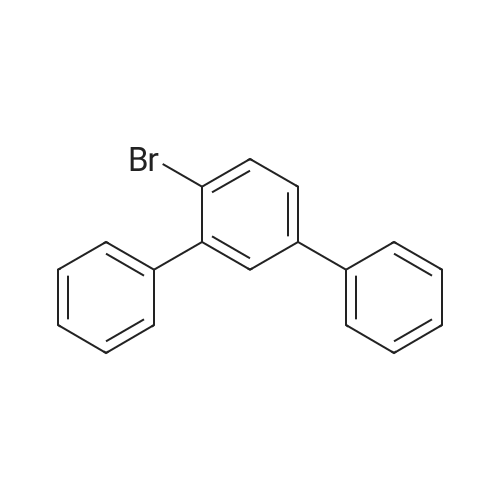
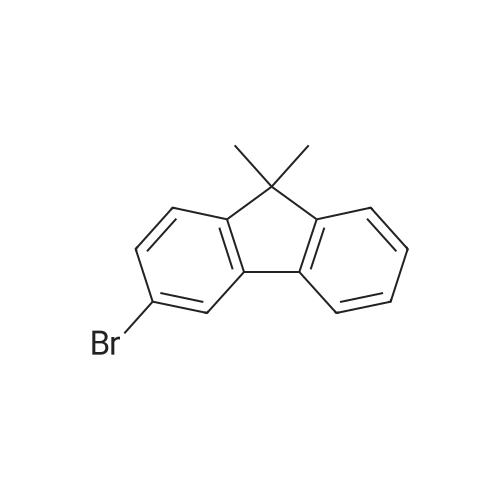
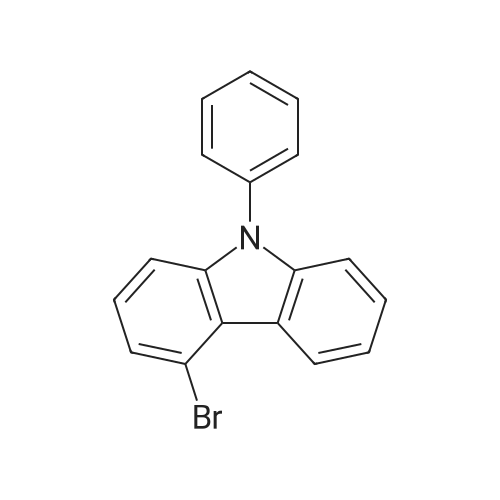
| 94% |
With 1,1'-bis(diisopropylphosphino)ferrocene; [Pd(dippf)(maleimide)]; sodium t-butanolate; In toluene; at 70℃; for 20h;Inert atmosphere; Sealed tube; Schlenk technique; |
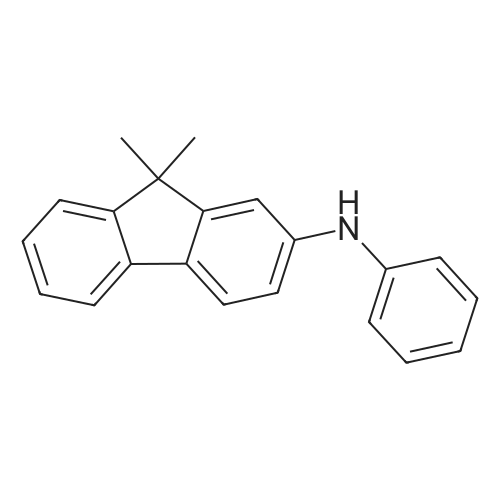
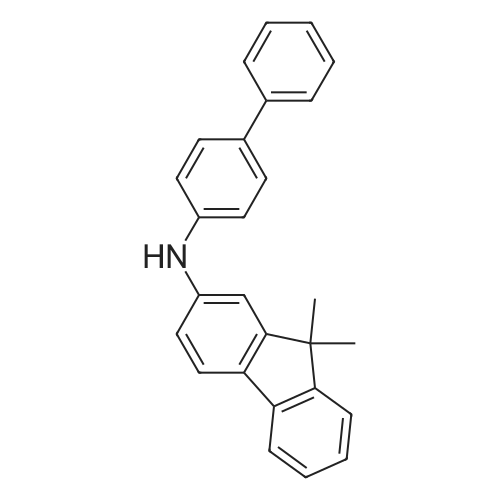
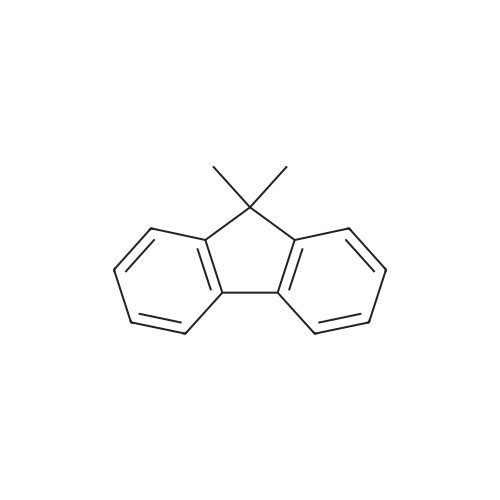
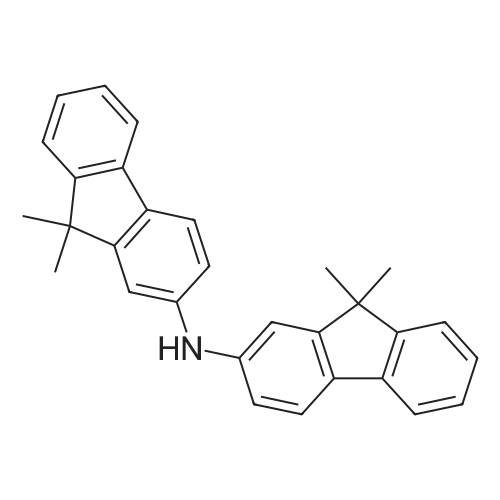
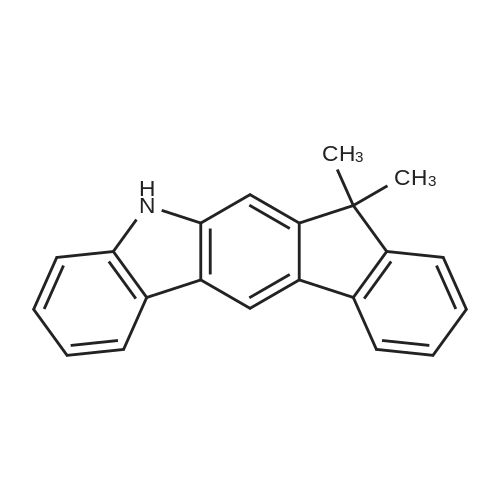
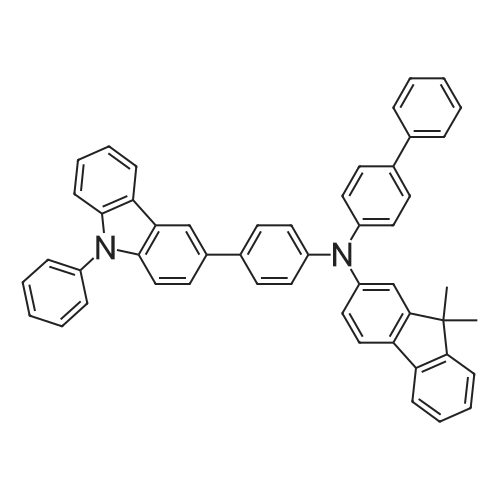
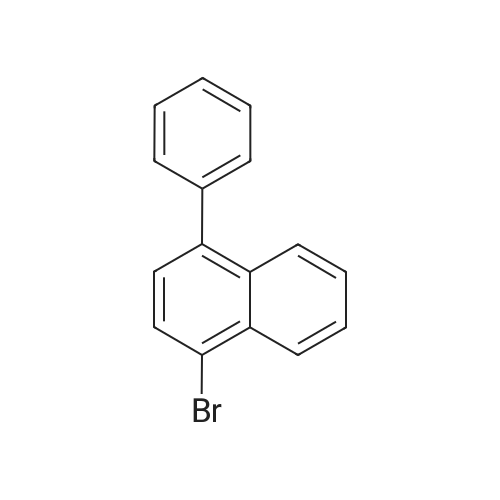
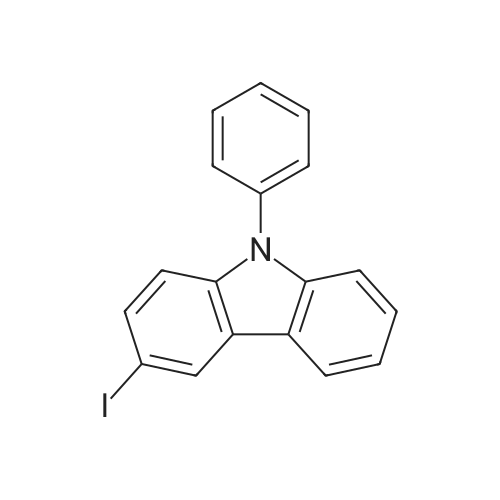
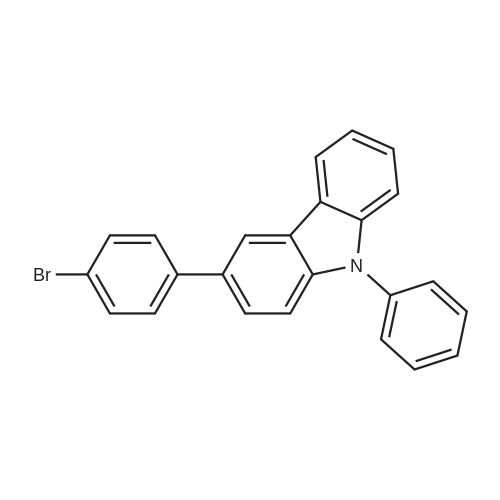
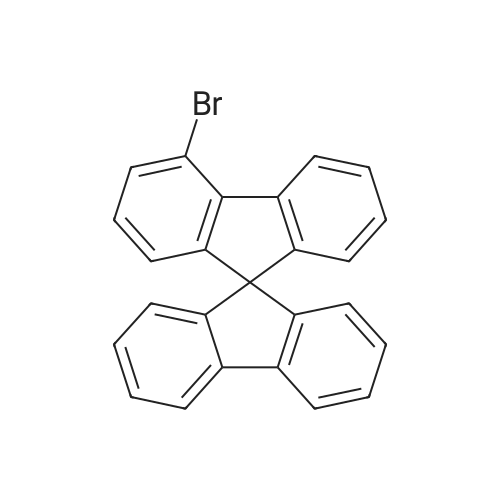
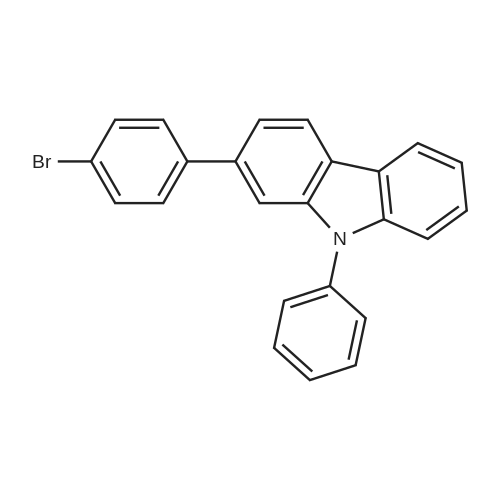
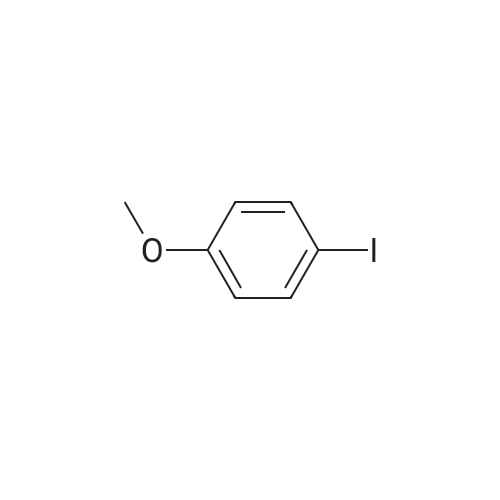
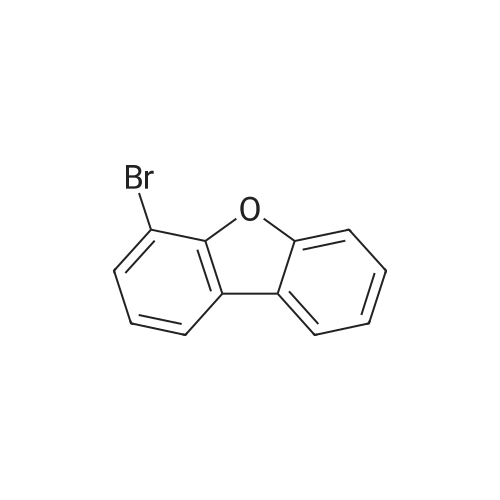
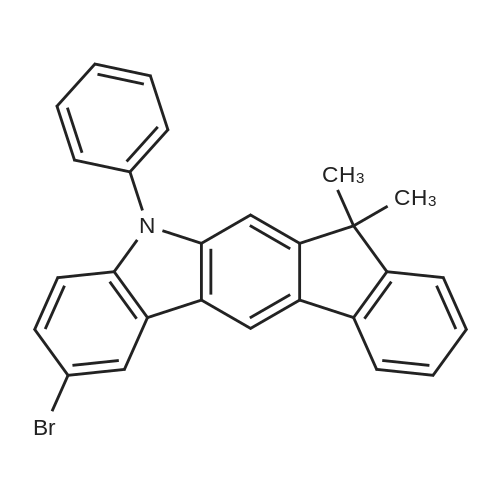
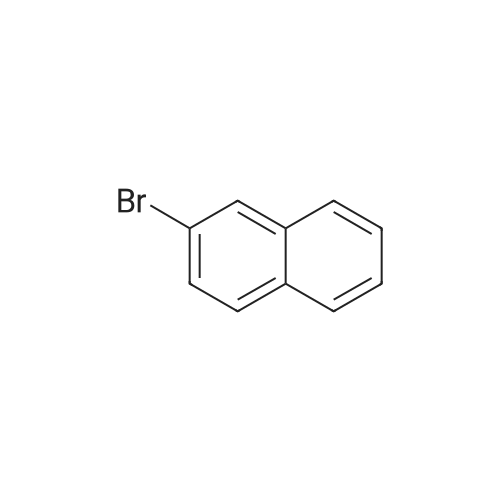
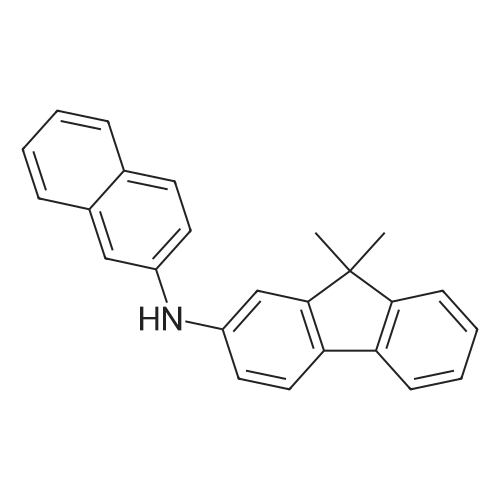
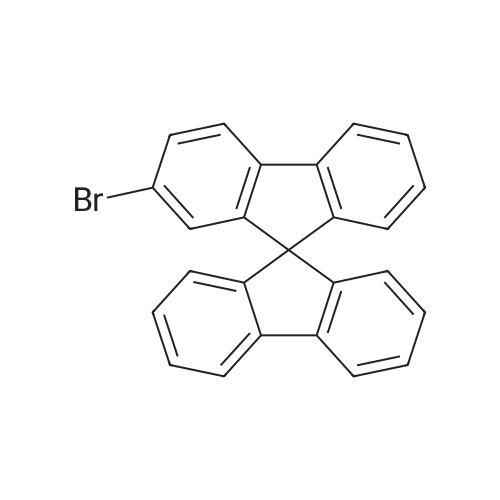
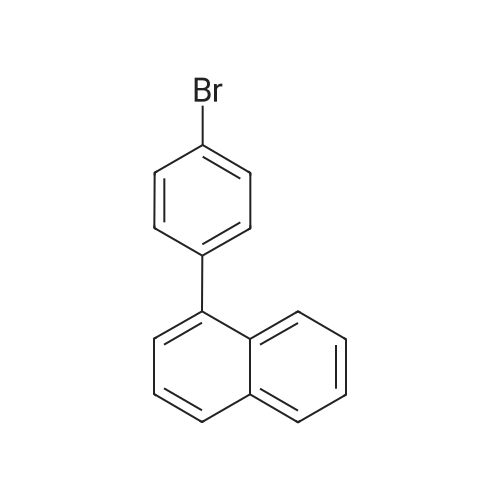
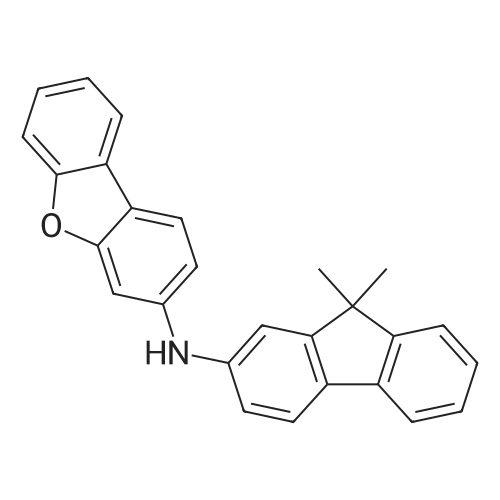
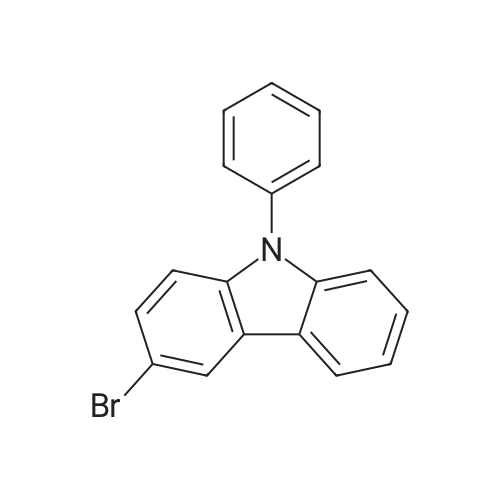
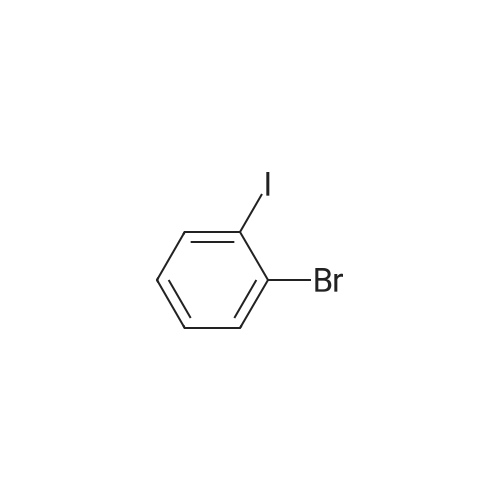
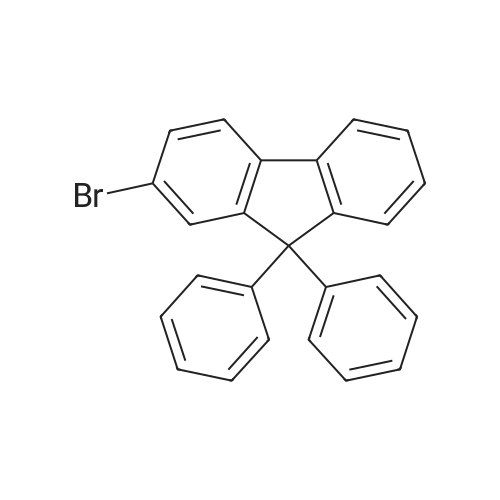
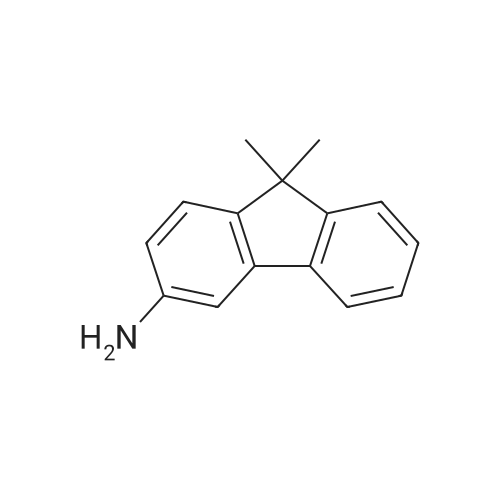
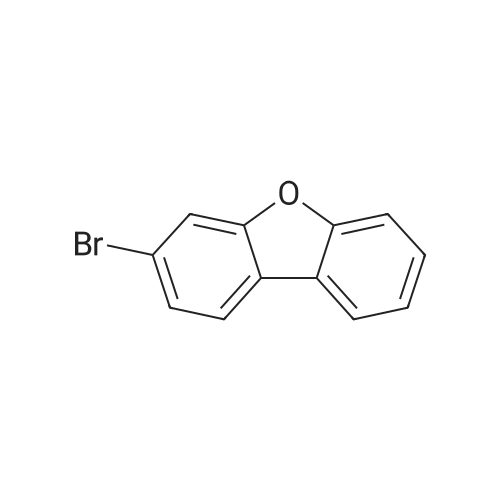
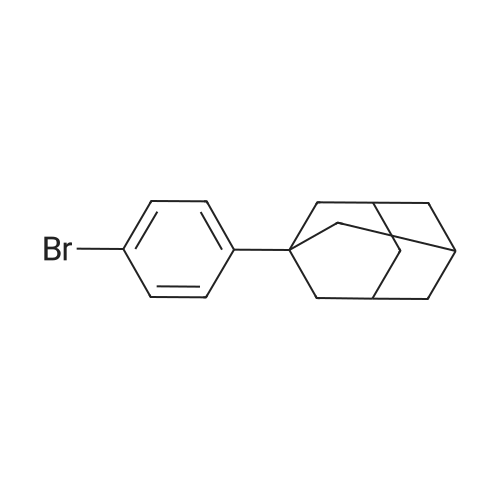
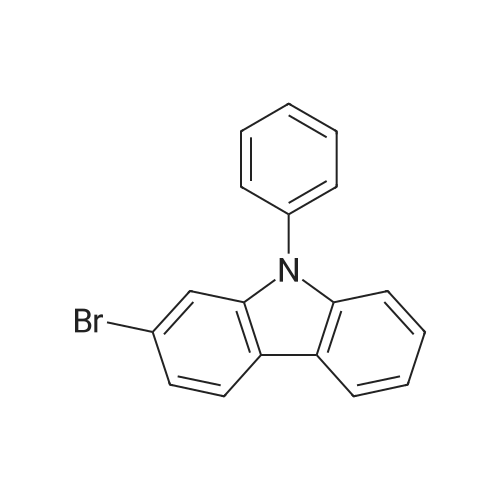
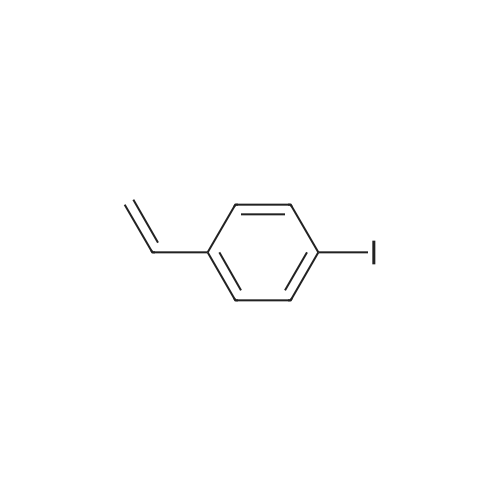
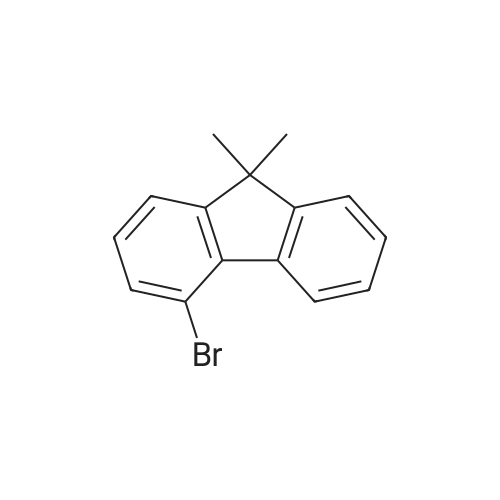
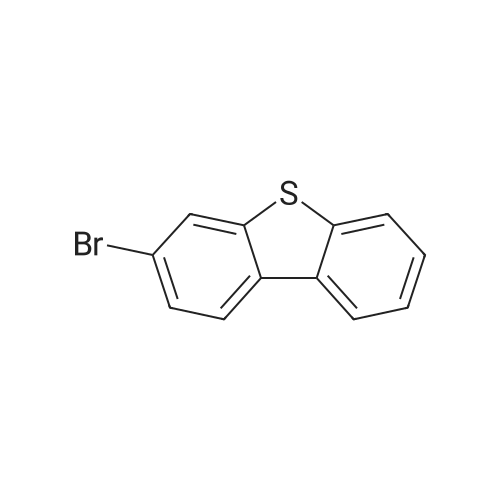
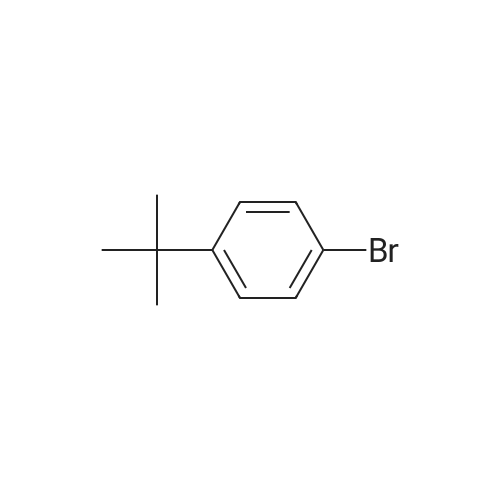
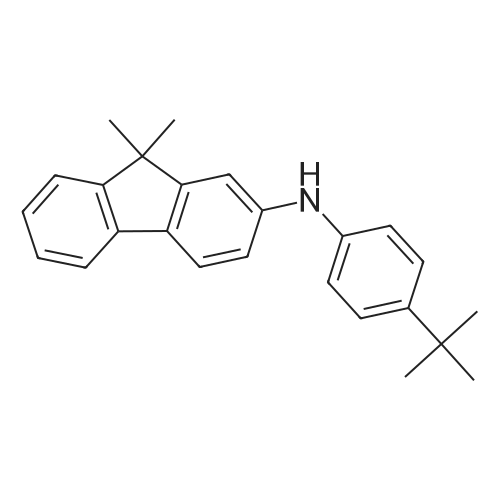
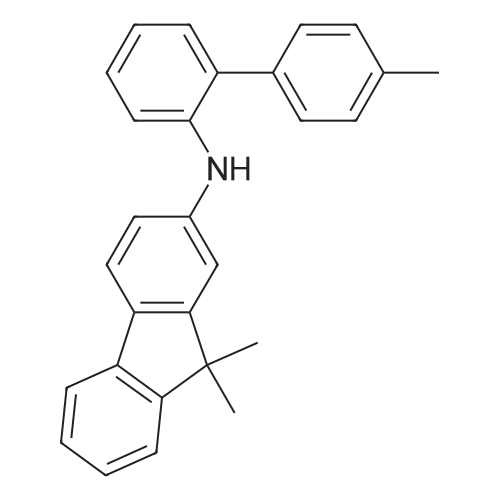
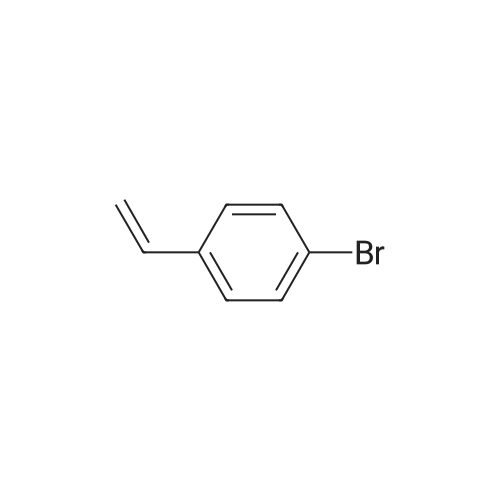
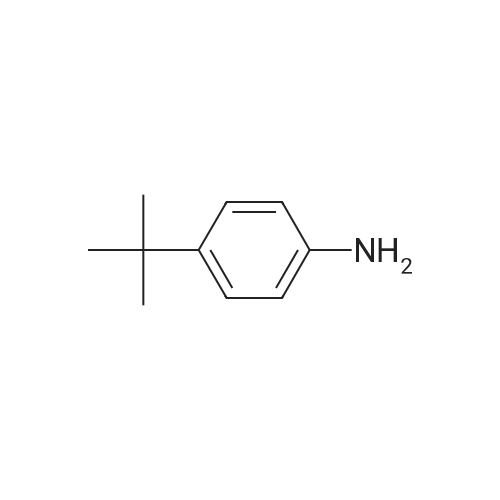
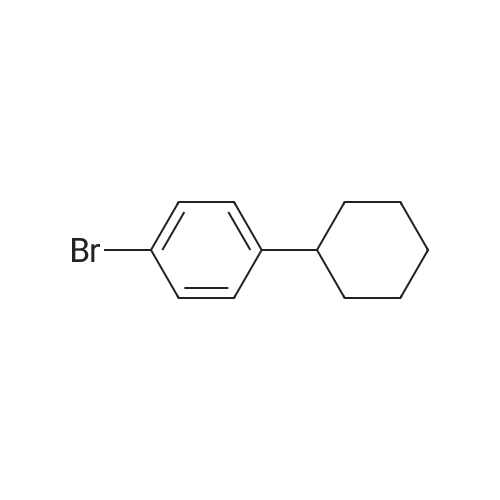
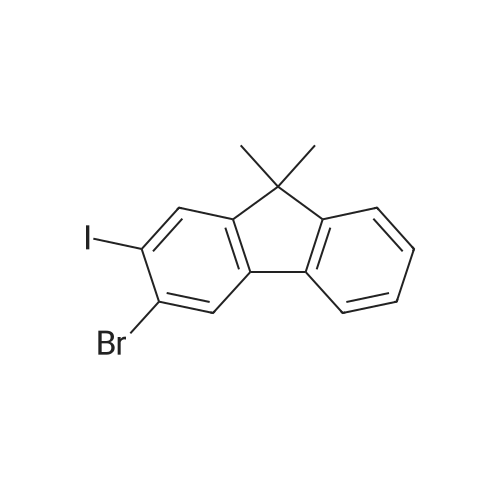
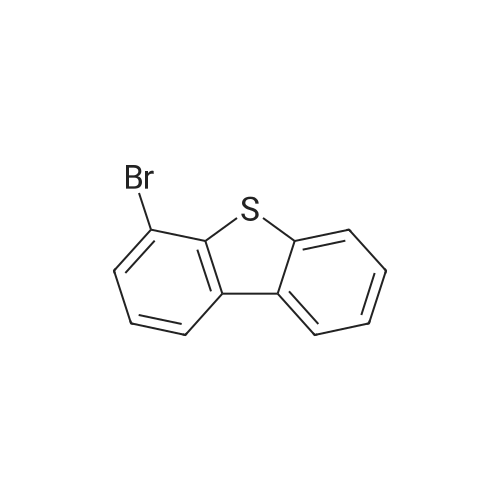
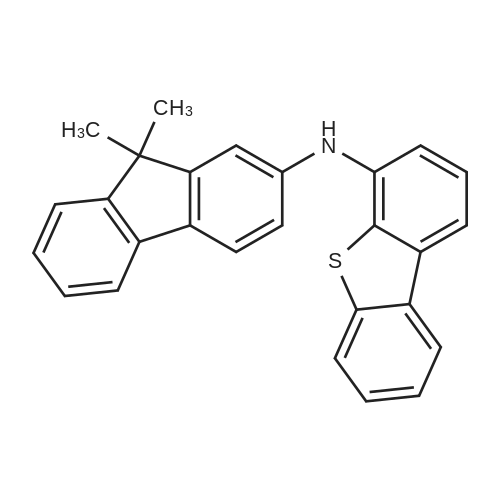
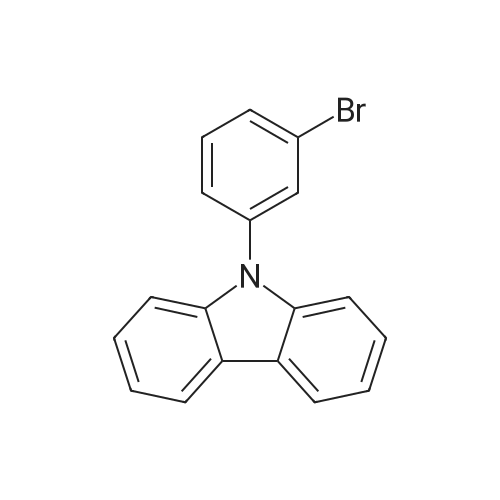
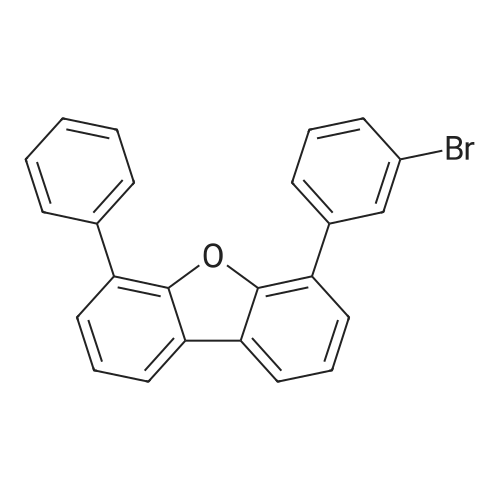
Examples 4a to 4i General Protocol (0096) All experiments were carried out in 20 mL headspace vials for gas chromatography that were sealed with crimped aluminium caps featuring Teflon-coated butyl rubber septa (both available, for example, from VWR). For the heating of the vessels, cylindrical aluminium blocks 8 cm high were used, corresponding precisely in their diameter to the hotplates of laboratory magnetic stirrers (e.g. Heidolph Mr 2002). These aluminium blocks were provided with ten holes 7 cm deep and the same diameter as the reaction vessels, and a hole for accommodating a temperature sensor. (0097) Vacuum distributors for connection to the Schlenk line were made for the simultaneous evacuation and refilling of ten vessels at a time. For this purpose, ten vacuum-compatible 3 mm Teflon tubes were each connected at one end with adapters to accommodate Luer-Lock syringe needles and were connected at the other end to a steel tube, which can be connected via a vacuum hose to the Schlenk line. (0098) For the implementation of the experiments, the aryl bromide (1.00 mmol), the corresponding primary amine (1.00 mmol) and sodium tert-butanolate (118 mg, 1.20 mmol) were weighed out in air into the reaction vessels, 20 mm magnetic stirring cores were added, and the vessels were given an airtight seal with septum caps, using crimping tongs. Ten reaction vessels at a time were introduced into the holes in an aluminium block, and connected to the vacuum distributor via hollow needles which were inserted through the septum caps. (0099) The reaction vessels were subsequently evacuated and charged with nitrogen gas three times in succession, in unison. When the reaction vessels had been given an inert gas atmosphere in this way, pressure compensation with the external atmosphere was produced at the vacuum line via an excess-pressure valve. Using a syringe, a stock solution of [Pd(dippf)(maleimide)](1.24 mg, 0.002 mmol) and dippf (0.85 mg, 0.002 mmol) in toluene (2 mL) was injected through the septum caps. The aluminium block was then brought to 70 C. and the needles of the vacuum distributor were removed. After a reaction time of 20 hours, the vessels were cooled and carefully opened and the reaction medium was diluted with dichloromethane (30 mL) and water (30 mL). The aqueous phase was adjusted to a pH of 7 using 1N hydrochloric acid, separated from the organic phase and extracted with dichloromethane (2×20 mL). The combined organic phases were dried over magnesium sulphate, filtered and finally analyzed by thin-layer chromatography. (0100) The solvent was removed under reduced pressure (40 C., 500 mbar) and the crude product which remained was purified by column chromatography (basic Al2O3, diethyl ether:hexane or ethyl acetate:hexane). Example 4a Synthesis of Compound 3a (0101) (0102) According to the general protocol, compound 3a was prepared starting from 1a (398 mg, 1.00 mmol) and 2a (209 mg, 1.00 mmol) and was isolated by column chromatography (Al2O3, diethyl ether:hexane=1:1) in 94% yield (496 mg, 0.94 mmol). 1H NMR (400 MHz, chloroform-d): delta=8.41 (d, J=1.3 Hz, 1H), 8.26 (d, J=7.8 Hz, 1H), 7.75-7.60 (m, 9H), 7.55-7.44 (m, 5H), 7.41-7.21 (m, 6H), 7.13 (d, J=7.0 Hz, 1H), 5.91 (s, 1H), 1.55 ppm (s, 6H), 13C NMR (101 MHz, chloroform-d): delta=155.3, 153.1, 142.6, 142.0, 141.3, 140.0, 139.3, 137.7, 134.6, 133.2, 132.5, 129.9 (2C), 128.1, 127.4 (2C), 127.0 (2C), 126.9, 126.0 (2C), 125.0, 123.9, 123.5 (2C), 122.4, 120.8, 120.3, 120.0, 119.9, 119.1, 118.1, 116.8, 112.2, 110.0, 109.9, 46.8, 27.2 ppm (2C). CHN: Calculated for C3H30N2: C, 88.94; H, 5.74; N, 5.32. found: C, 88.79; H, 5.86; N, 5.19. |
| 94% |
With 1,1'-bis(diisopropylphosphino)ferrocene; 1,1?-bis(diisopropylphosphino)ferrocene palladium(0)-maleimide; sodium t-butanolate; In toluene; at 70℃; for 20h;Schlenk technique; Sealed tube; Inert atmosphere; |
General procedure: All tests were carried out in 20 mL headspace vials for the gas chromatography, which vials were sealed with aluminum flange caps with teflon-coated butyl rubber septa (both available, for example, from VWR). To control the temperatures of the vessels, 8 cm-high cylindrical aluminum blocks were used, the diameter of which exactly corresponds to that of the hot plates of laboratory magnetic stirrers (e.g. Heidolph Mr 2002). These aluminum blocks were provided with ten 7 cm deep holes with the diameter of the reaction vessels and a hole for accommodating a temperature sensor. To enable the simultaneous evacuation and refilling of ten vessels, vacuum distributors were manufactured for connection to the Schlenk line. To this end, ten 3 mm vacuum-tight teflon hoses were respectively connected at one end to adapters to accommodate Luer Lock syringe needles and at the other end to a steel tube that can be connected to the Schlenk line via a vacuum hose. To carry out the tests, the aryl bromide (1.00 mmol), the corresponding primary amine (1.00 mmol) and sodium-tert-butanolate (118 mg, 1.20 mmol) were weighed out in air into the reaction vessels, 20 mm magnetic stirring bars were added and the vessels were sealed airtight with septa caps using flanging pliers. 10 reaction vessels were respectively inserted into the holes of an aluminum block and connected to the vacuum distributor via cannulas, which were bored through the septa caps. The reaction vessels were then collectively evacuated and flushed with nitrogen three times in succession. After the reaction vessels were provided with an inert gas atmosphere in this way, a pressure equalization with the outside atmosphere was created in the vacuum line via the relief valve. A stock solution of [Pd(dippf)(maleimide)] (1.24 mg, 0.002 mmol) and dippf (0.85 mg, 0.002 mmol) in toluene (2 mL) was injected through the septa caps with the aid of a syringe. The aluminum block was then brought to 70 C. and the needles were removed from the vacuum distributor. After a reaction time of 20 hours, the vessels were carefully opened after cooling and the reaction medium was diluted with dichloromethane (30 mL) and water (30 mL). The aqueous phase was set to pH=7 with 1N hydrochloric acid, separated from the organic phase and extracted with dichloromethane (2×20 mL). The combined organic phases were dried over magnesium sulfate, filtered and then investigated using thin layer chromatography. The solvent was removed in vacuum (40 C., 500 mbar) and the remaining raw product was cleaned using column chromatography (basic Al2O3, diethyl ether:hexane or ethylacetate:hexane). |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping