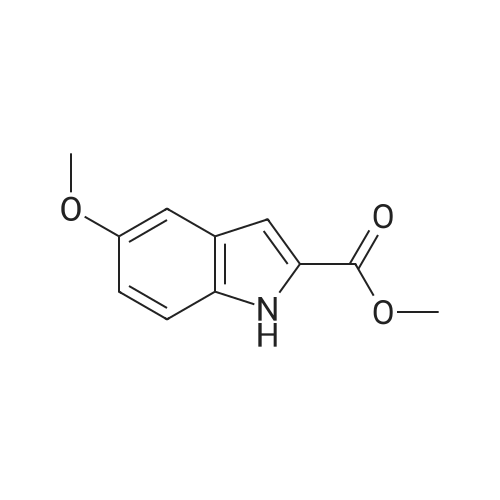
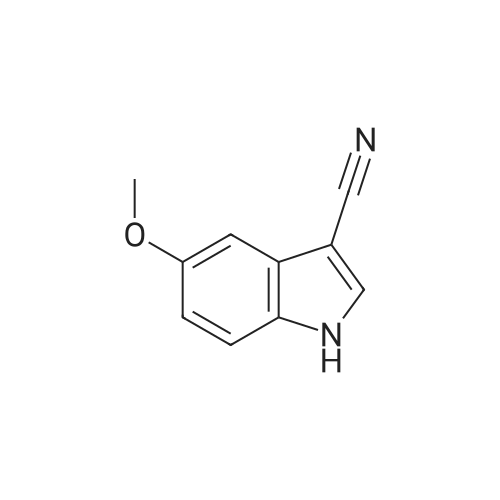
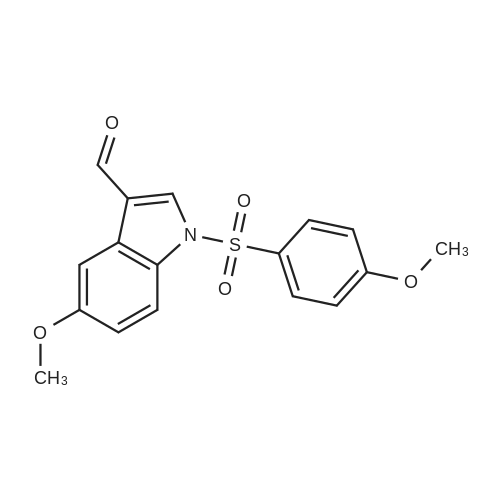
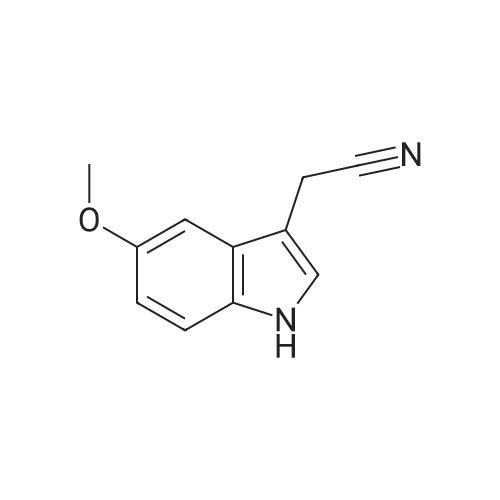
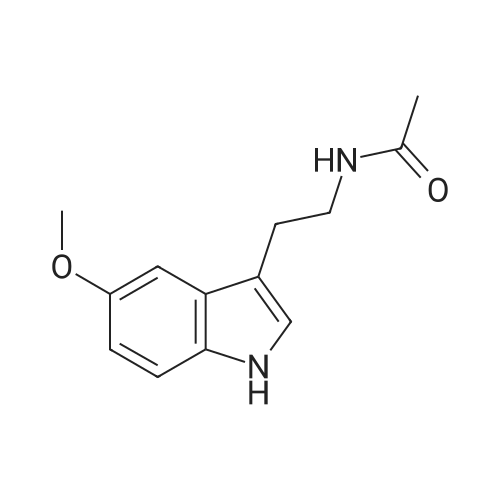
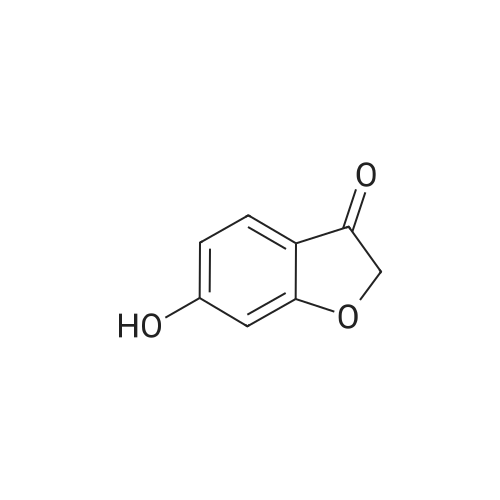
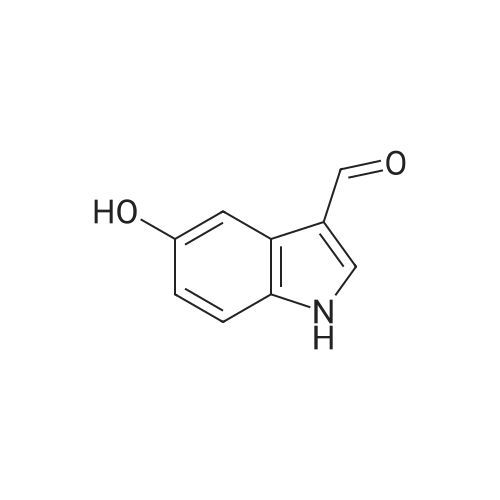
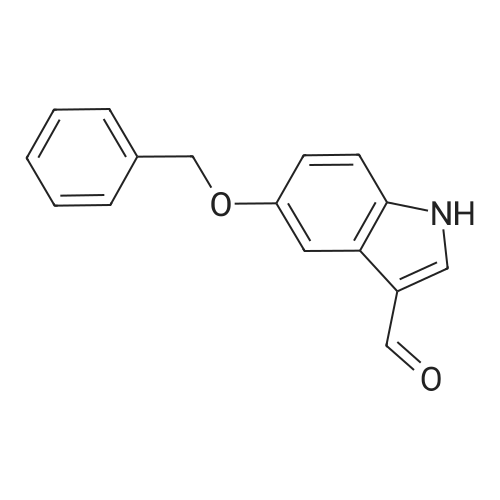
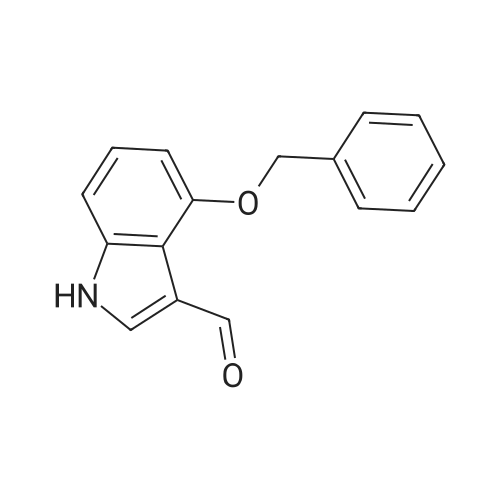
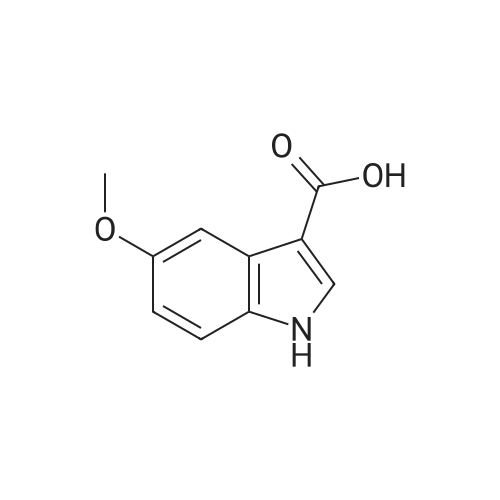
| 95% |
With trichlorophosphate |
|
| 94% |
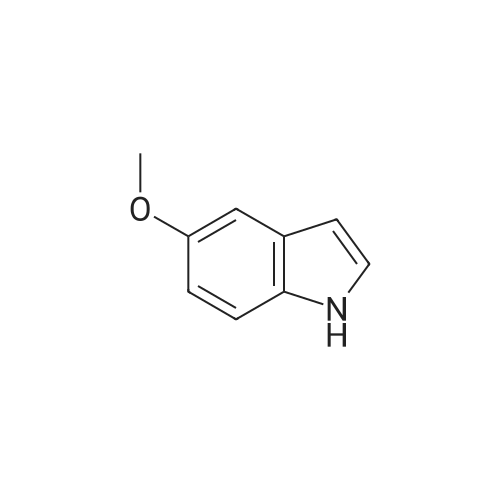
Stage #1: N,N-dimethyl-formamide With oxalyl dichloride at 0℃; for 1h;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 20℃; for 5h;
Stage #3: With sodium hydroxide In N,N-dimethyl-formamide at 100℃; for 0.166667h; |
5.2.1. General procedure for syntheses of compounds (4a-k)
General procedure: Oxalyl chloride (0.3 mL) was added in a drop-wise manner to cooled (ice-bath) DMF (3 mL) under stirring. The mixture was then stirred at 0 °C for 1 h. A solution of the substituted indole (4 mmol) in DMF (1.5 mL) was then added to the reaction mixture in a dropwise manner. The resulting mixture was stirred at room temperature for 5 h. A 2 N solution of sodium hydroxide (2 mL) was then added, and the mixture was heated at 100 °C for 10 min. The mixture was then cooled and extracted with ethyl acetate (3 X 50 mL). The organic layers were combined and washed with sequentially water and brine. The organics were dried (Na2SO4) and distilled to dryness to give the crude residue, which was purified by flash column chromatography using ethyl acetate/petroleum ether (3:1, v/v) as the eluent to give pure indole-3-carbaldehyde (4a-k). |
| 93% |
With trichlorophosphate at 35℃; for 1h; |
|
| 93% |
With trichlorophosphate at 20℃; for 5h; Inert atmosphere; |
|
| 92% |
With trichlorophosphate at 0 - 55℃; for 2h; |
Synthesis of indole-3-carbaldehyde derivatives (18a, 18b)
General procedure: To a solution of compounds 17a, 17b (10 mmol) in 20 mL of DMF, phosphorus oxychloride (1.4 mL, 15 mmol) was added dropwise at 0°C. In 2 h of the reaction at 55°C, the mixture was poured into 150 mL of ice water. 20 % NaOH solution was added to adjust pH at 8.0. The crude product was filtered off and recrystallized from anhydrous ethanol to give the corresponding product. |
| 90% |
With trichlorophosphate at 0 - 40℃; |
|
| 90% |
With 1,2-Diiodoethane; water; triphenylphosphine at 80℃; for 2h; |
3.1 General Procedure for the Formylation of electron-rich and -neutralindoles
General procedure: Into a 25 mL tube, Ph3P (0.75 mmol, 1.5 equiv.), ICH2CH2I (0.75 mmol, 1.5 equiv.),1 (0.5 mmol, 1 equiv.), DMF (2 mL) and H2O (5 mL) were added under an airatmosphere. The mixture was stirred at 80 oC for 2 hours. Then the mixture was cooledto room temperature. A saturated aqueous brine solution was added, and the crudeorganic product was extracted by CH2Cl2. The combined organic phase was dried withanhydrous Na2SO4. After filtration, the solution was concentrated under reducedpressure to remove the solvent. The residue was subjected to flash columnchromatography on silica gel (pentane/ethyl acetate) to give the pure products 4a-4i. |
| 89.4% |
With trichlorophosphate at 35℃; |
4.1.12 1H-Indole-3-carbaldehyde (4a)
General procedure: To a stirring solution of POCl3 (18.36 g, 0.120 mol) in DMF (25 mL), indole (11.7 g, 0.100 mol) in DMF (15 mL) was added dropwise. The mixture was stirred at 35°C for 1 h, and then be poured into ice water and neutralized with 20 % NaOH aq. The solution was filtered and the residue was washed with water and dried under IR to give the compound 4a as yellow needle (13.86 g, 95.48 %). Mp = 192-194°C, lit.31 Mp = 196-197°C. |
| 85% |
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0℃; for 0.5h;
Stage #3: With sodium hydroxide In water Cooling with ice; Reflux; |
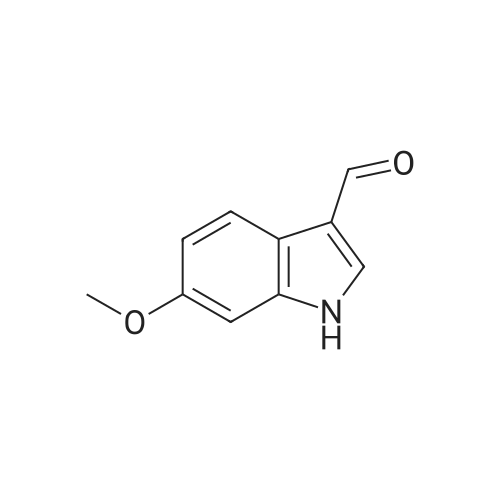
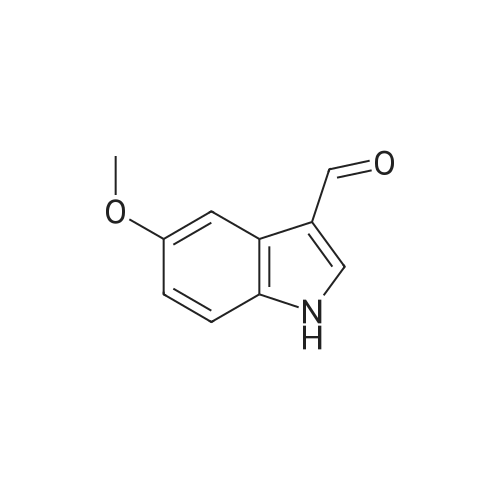
Synthesis of 5-methoxy-indole-3-carbaldehydes; Preparation of S-methoxy-indole-3-carbaldehyde, 5-methoxy-2-methyl-indole-3- carbaldehyde, and 3-formyl-5-methoxy-indole-2-carboxylic acid POCI3 (1.6 ml_, 17 mmol, 1.1 eq.) was added to DMF (6 mL) at 00C and the solution was stirred for 30 minutes. This mixture was added to a stirring solution of the selected 5-methoxy- indole (15.5 mmol, 1 eq.) in DMF (11.5 mL) at 00C. The resulting mixture was stirred at 00C for 30 minutes, then allowed to warm to room temperature. The reaction was poured into ice, basified to pH 10 with 5 N NaOH, warmed to room temperature, refluxed for 5 minutes and allowed to cool to rt. Finally, it was acidified to pH 4 with 2 N HCI and the resulting precipitate was filtered and washed with water until pH 7. The solid product was dried under vacuum.5-Methoxy-indole-3-carbaldehydeYield: 85%. MS (m/z): 176.2 (MH+). |
| 83% |
With trichlorophosphate at 0 - 40℃; for 1.5h; |
3.1.1. General Procedure for Synthesizing the Indoles-3-Carboxaldehydes (4b and 4e-4g)
General procedure: Phosphorus oxychloride (0.42 g, 2.74 mmol) was added dropwise to a solution of the indole 5b, 5e-5g (0.30 g, 2.29 mmol) in DMF (0.84 g, 11.4 mmol) at 0 °C for 30 min. The solution was then heated at 40 °C for 1 h. Ice was added to the reaction vessel, followed by a solution of sodium hydroxide (2 M). The solution was refluxed for 40 min. The mixture was cooled and extracted using ethyl acetate, and the organic phase was washed with brine. The organic extracts were combined, dried over Na2SO4, and concentrated. The crude residue was purified by chromatography on a silica gel column using hexane-ethyl acetate as an eluent to obtain the desired product [19]. |
| 80% |
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0 - 20℃; for 2h;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 20 - 60℃; for 1h; |
|
| 79% |
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 38℃; for 1.25h;
Stage #2: With sodium hydroxide In water at 170℃; for 0.0833333h; |
|
| 79% |
With phosphorus pentachloride at 20℃; for 1h; |
6 example 6
A 5-methoxy-1H-indole (14.7 g, 0.1 mol) was dissolved in N, N -dimethylformamide (73.5 g, 1 mol) in a 500ml of reaction flask at 0 ° C with mechanical agitation, then phosphorus pentachloride / N, N -dimethylformamide mixed formylation reagent (15 g, 0.12 mol) was added in portions. The reaction mixture was stirred at room temperature for 1 hour. After completion of the reaction, 100 mL of water was added and stirred at room temperature for 1 hour. The reaction was neutralized with 2N sodium hydroxide solution and the resulting suspension was filtered. The filter cake was dissolved in methanol / tetrahydrofuran and the pH was adjusted to 1 with concentrated hydrochloric acid. The solution was stirred at room temperature for 45 minutes and neutralized with 4N sodium hydroxide solution to neutral. The solvent was partially evaporated to give the product, filtered and washed with water. Finally, 5-methoxy-1-hydrogen-indole-3- carboxaldehyde was obtained (14 g, 79%). This product can be used in the production of Tegaserod Hydrogen Maleate. |
| 69% |
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 20℃; for 0.75h; Cooling with ice;
Stage #2: 5-methoxylindole at 40℃; for 1.25h; |
General procedure 2 for the synthesis of substituted indole-3-carbaldehydes
The Vilsmeier-Haack reagent was generated by the addition of 2 ml of POCI3 over the course of15 minutes to 8 ml of DMF cooled in an ice-salt bath. After the addition was complete, the icebath was removed and the contents of the flask allowed to warm to room temperature overapprox. 30 minutes. The substituted indole (21.9 mmol) was dissolved in 10 ml of DMF andadded over a period of 15 minutes to the formylating mixture. The stirring was continued for an10 hour during which the flask contents were heated to 40°C in a hot water bath. A total of 50 mlice cold H20 and 20 ml of 5 M NaOH were added, the mixture quickly brought to a boil and leftto cool slowly. The crystals were removed by filtration, washed with cold water and vacuumdried. The products thus obtained were in most cases sufficiently pure for the subsequentreactions, and the impure aldehydes were recrystallized from ethanol-water mixtures. Yields15 varied from 48 to 90%.; The compound was synthesized from 5-methoxy-1H-indole (170 mmol) according to the25 general procedure 2, with the exception that it was scaled up proportionally to the 5-methoxyindole (69% yield).LC-MS: m/z = 176.11; tR = 2.20 min. |
| 63% |
With Vilsmeier reagent at 20℃; for 1h; |
26.a
(a) 5-methoxyindole (200 mg, 1.36 mmol) dissolved in anhydrous DMF,Drip the newly prepared Vilsmeier reagent,Allow to react at room temperature for one hour, then pour into ice water and filter by suction.Dryed to a yellow solid 150 mg, yield 63%. |
| 59% |
With trichlorophosphate |
|
| 54% |
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.166667h;
Stage #2: 5-methoxylindole at 45℃; for 2h; |
|
|
With trichlorophosphate und anschliessenden Hydrolysieren mit wss.Natronlauge; |
|
|
With trichlorophosphate at 35℃; for 1h; |
|
|
With hydrogenchloride; trichlorophosphate 1.) 0 deg C, 1 h, 2.) MeOH, THF, RT, 45 min; Yield given. Multistep reaction; |
|
|
With trichlorophosphate at 0℃; |
|
|
With trichlorophosphate at 0 - 60℃; |
|
|
With sodium hydroxide; trichlorophosphate |
|
|
With trichlorophosphate at 0 - 23℃; |
|
|
With trichlorophosphate |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate for 0.5h; Cooling with ice;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0 - 100℃; for 3.08333h; |
General procedure for the formylation of indoles 4a-4h or azaindole 5
General procedure: Phosphorous oxychloride (2 mmol) was added dropwise to dimethylformamide (3 mL) cooled under ice-bath and allowed to stir for 30 min. A solution of indoles 4a-4h or azaindole 5 (1 mmol) in DMF (5 mL) was added dropwise for 5 min at 0 oC. The mixture was further allowed to stir for 3 h at 90-100 oC. Reaction mixture was cooled to room temperature and poured into crushed ice. Excess POCl3 was quenched with 1 N NaOH and left overnight at room temperature. Ice-cold reaction mixture was then extracted (50 mL × 3) with EtOAc. Combined organic layer was concentrated on rotary evaporator and crude products were purified by silica gel (No.100-200) column chromatography to get indole-3-carboxaldehydes 1a-1h or 6 in 60-80% yield. |
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.666667h; Inert atmosphere;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0 - 35℃; for 1.33333h;
Stage #3: With sodium hydroxide In water; N,N-dimethyl-formamide at 100℃; for 0.5h; Cooling with ice; |
5.1.9. General procedure for the synthesis of 5-methyl-1H-indole-3-carbaldehyde
General procedure: To a 100 mL three-necked round flask was introduced anhydrous N,N-dimethylformamide (4.2 mL, 34.35 mmol) at 0 °C under argon followed by slow addition of phosphorus oxychloride (1.3 mL, 13.62 mmol). Solution was mixed at 0 °C for 40 min. A solution of 5-methyl-1H-indole (1.627 g, 12.41 mmol) in 2.5 mL of DMF was slowly added maintaining the temperature below 10 °C. The solution was stirred for 40 min at 0 °C and then 35 °C for additional 40 min. Then ice was added to the flask and a solution of sodium hydroxide (5.5 g, 137.25 mmol dissolved in 14.6 mL of water) was introduced using a dropping funnel. Solution was vigorously stirred during the addition, then heated to 100 °C for 30 min and left to reach room temperature. The brown precipitate was filtered off and washed with large volumes of water. The powder was dried in vacuum. Yield 71.5%. |
|
With trichlorophosphate |
|
|
With trichlorophosphate |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 40℃; for 2h;
Stage #2: With sodium hydroxide In water at 90℃; for 1h; |
|
|
With trichlorophosphate Cooling with ice; |
|
|
With sodium hydroxide; trichlorophosphate |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 20℃; Inert atmosphere;
Stage #2: With water; sodium hydrogencarbonate for 0.5h; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 15℃; for 0.25h;
Stage #2: 5-methoxylindole at 0 - 70℃; for 3h; |
General synthetic method
General procedure: Compound 2b was obtained by four steps ofreactions. Step 1: POCl3 (10.0mmol) wasadded into DMF (N,N-dimethylformamide;10.0mmol) and stirred at 158C for 15 min 5-Fluoroindole (1.0mmol) was dissolved inDMF, which was added in dropwise to themixture at 08C. The mixture was stirred at708C for 3 h. The reaction solution waspoured into icy water (50ml) and continuedstirring until precipitation appeared. Thesolid was filtered and washed with water.Step 2: 5-fluoro-indole-3-carboxaldehyde(1.2 mmol), 4b-amino-podophyllotoxin(413 mg, 1.0 mmol), and two drops ofglacial acetic acid in 95% C2H5OH(10.0ml) were stirred at r.t. for 12 h untilprecipitation appeared. The solid wasfiltered and washed with glacial aceticacid. Step 3: the compound b (1.0mmol)was reducted in anhydrous methanol byNaBH4 (4.0mmol) at 08C for 3h, 5% HClwas added to themixture to end the reaction.The saturated NaHCO3 was add to adjustpH.7. The solvent was evaporated undervacuum. Finally, the residue was dissolvedin anhydrous CH2Cl2 and chromatographedon silica gel plate using petroleum ether andethyl acetate (v/v 1:1) to give the correspondingsecondary amine 2b (0.31 g, 56%)as white solid. For 1H NMR spectral data(CDCl3) (see Table 2). HR-ESI-MSm/z: 561.2033 [M H] (calcd forC31H30FN2O7, 561.2037) |
|
With sodium hydroxide; trichlorophosphate In water at 0℃; for 6h; Reflux; |
|
|
With trichlorophosphate at 20℃; for 2h; |
General procedure for preparation of 1H-indole-3-carboxaldehyde (4-6)
General procedure: A solution of substituted indole 1-3 (82.6 mmol) in DMF (20 mL) was added dropwise to a stirring suspension of phosphorus oxychloride (7.0 mL, 75.0 mmol) in DMF (35 mL). After stirring at room temperature for 2 h, the reaction mixture was poured onto ice. The mixture was made basic with a solution of NaOH (330 mmol, 13.2 g) in H2O (44 mL), then was extracted with Et2O (2 * 50 mL). The combined organic layers were washed with brine, dried (MgSO4), and concentrated under vacuum. Flash chromatography on silica gel (10% ethyl acetate/hexanes) gave the title compounds 4-6 as an off-white solid, respectively. |
|
With trichlorophosphate at 20℃; |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 40℃; for 2.5h;
Stage #2: With sodium hydroxide In water at 90℃; for 1h; |
|
|
With trichlorophosphate at 20℃; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.5h;
Stage #2: 5-methoxylindole at 20℃; |
General Procedure for Synthesis of Bis(indolyl)-hydrazide-Hydazones (NMK-BH1-6).
General procedure: The intermediatesindole-2(3)-carbohydrazide (2) and indole-3-carboxaldehyde(4) were prepared starting from commercially available indole,as described previously. Indole-3-carboxylic acids 1were prepared from the reaction of indole with trifluoroaceticanhydride followed by hydrolysis with sodium hydroxide. Toobtain the corresponding ester, a solution of indole-3-carboxylicacid 1 (1 mmol) in absolute ethanol (20 mL) was refluxed witha catalytic amount of concentrated sulfuric acid for 20 h and theresidue was extracted with ethyl acetate (30 mL), washed with asaturated sodium bicarbonate solution (20 mL), dried, andevaporated. As shown in Scheme 1, a solution of theappropriate ester (1 mmol) and hydrazine hydrate (2 mmol)in ethanol (15 mL) was refluxed for 24 h and cooled, and thesolid that was obtained was filtered and recrystallized fromethanol to give pure indole-2(3)-carbohydrazide (2) in 85-90% yield.For preparation of indole-3-carboxaldehydes 4, withcontinuous stirring, phosphorous oxychloride (8.41 mL, 90mmol) was added to 0 °C dimethylformamide (28 mL, 370mmol) for 30 min followed by addition of a solution of indole 3(85.47 mmol) in DMF (10 mL, 130 mmol) at roomtemperature until it became a yellow paste. At the end of thereaction, 30 g of crushed ice was added to the paste followed bydropwise addition of a sodium hydroxide solution (1 N solution, 100 mL) while the mixture was being stirred. Theresulting suspension was heated rapidly to 90 °C, cooled atroom temperature, and refrigerated overnight. The product wasfiltered, washed with water (2 × 100 mL), and air-dried toafford the pure indole-3-carboxaldehydes 4 in 80-90% yields(Scheme 1).Finally, to the mixture of indole-2(3)-carboxylic acidhydrazide 2 (1 mmol) and indole-3-carboxaldehyde 4 (1mmol) in absolute ethanol (20 mL) was added a catalyticamount of glacial acetic acid, and the mixture was refluxed at 80°C for 5 h and cooled and the solid that separated out wasfiltered and recrystallized from ethanol to afford bis(indolyl)-hydrazide-hydrazones in 75-90% yields (Scheme 1). |
|
With trichlorophosphate at 0℃; for 2h; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate Cooling with ice;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 20℃; for 1h;
Stage #3: With sodium hydroxide In water at 100℃; for 0.25h; |
General procedure for the preparation of indolylnitroalkenes
General procedure: Phosphorus oxychloride (0.86 mL) was added dropwise to dimethylformamide (1.0 mL) with ice-bath cooling. The chosen indoles (1.0 g) were added as a dimethylformamide solution for preparation of corresponding indolecarbaldehydes. The resulting mixture was stirred at room temperature for one hour. The reaction mixture was quenched with chilled water, followed by the addition of aq Sodium hydroxide solution and refluxed at 100 °C for 15 min. The reaction mixture was cooled to room temperature and maintained at 0 °C overnight. The precipitates formed were collected and washed with water. After this, the -NH groups of different indolecarbaldehydes were protected with methyl, tosyl, benzyl, or methylsulfonyl by using corresponding halogenides (1.5 equiv) under different basic conditions, such as sodium hydride (1.5 equiv) using acetonitrile as a solvent. Then, the reaction of resulting N-protected indolecarbaldehydes (1.0 g) with nitromethane (5 mL) in the presence of 30 mol % of ammonium acetate as the catalyst to furnish the desired indolylnitroalkenes.9 |
|
Stage #1: N,N-dimethyl-formamide With thionyl chloride at 20℃; for 0.5h;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0 - 20℃; for 0.833333h;
Stage #3: With sodium hydroxide In water; N,N-dimethyl-formamide Reflux; |
tert-Butyl 5-Bromo-3-[(E)-2-nitrovinyl]-1H-indole-1-carboxylate (9b); Typical Procedure
General procedure: 5-Bromoindole (1.35 g, 6.9 mmol) was formylated as described for1H-indole-3-carbaldehyde to produce 6-bromo-1H-indole-3-carbaldehyde(1.50 g, 97%) with the exception that the obtained productwas not purified by refluxing in EtOH.5-Bromo-1H-indole-3-carbaldehyde (1.50 g, 6.7 mmol) and NH4OAc(0.52 g, 6.7 mmol) were heated at reflux in CH3NO2 (18 mL, 335mmol) for 45 min, cooled to r.t., and treated with H2O (70 mL). Theproduct was extracted with EtOAc (70 mL), washed with H2O (5 × 50mL) and NaCl solution (20 mL), and dried over anhydrous Na2SO4. Thesolvent was removed in vacuo to afford 6-bromo-3-[(E)-2-nitrovinyl]-1H-indole (1.72 g, 96%, brownish crystals), which was dried in vacuoand used without further purification.A suspension of 5-bromo-3-[(E)-2-nitrovinyl]-1H-indole (1.72 g, 6.4mmol) and DMAP (0.08 g, 0.64 mmol) in anhydrous THF (7 mL) wastreated with a solution of Boc2O (2.1 g, 9.7 mmol) in anhydrous THF(7 mL) dropwise at 0-5 °C over a period of 15 min, stirred at r.t. for 3h, and concentrated in vacuo. The residue was dissolved in CH2Cl2 (50mL), washed with citric acid solution (10%, 20 mL), H2O (20 mL), andconcd NaCl solution (15 mL), and dried over anhydrous Na2SO4. TheCH2Cl2 was evaporated in vacuo and the residue was purified by chromatographyon a column of silica gel (EtOAc-hexane, 6:1) to provide tert-butyl 5-bromo-3-[(E)-2-nitrovinyl]-1H-indole-1-carboxylate. Yield: 2.11 g (83% from 5-bromoindole); pale-yellow solid; mp 156-157 °C (MeOH); Rf = 0.55 (EtOAc-hexane, 1:8). |
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate for 0.333333h;
Stage #2: 5-methoxylindole at 0℃; for 0.5h; |
5-Methoxy-1H-indole-3-carbaldehyde (22)
DMF (3.0 mL, 38.9 mmol) and POCl3 (0.85 mL, 9.28 mmol) were stirred in an ice bath for 20 min until the yellow Vilsmeier compound formed. This Vilsmeier compound was then added to a solution of 21 (1.04 g, 6.45 mmol) in DMF (5 mL) and the reaction was stirred for 30 min in 0 °C. NaOH (2 M, 50 mL) was added and the solution was stirred for 10 min. Then the solution was extracted with DCM (2×100 mL), dried with Na2SO4 and evaporated. The obtained yellow solid 1.20 g (90%) was used without purification. 1H NMR (300 MHz, CDCl3): δ (ppm) 9.93 (s, 1H), 8.54 (d, J = 2.4 Hz, 1H), 7.93 (d, J = 9.0 Hz, 1H), 7.46 (s, 1H), 6.97 (dd, J = 9.0, 2.5 Hz, 1H), 3.72 (s, 3H), 1.24 (s, 1H); 13C NMR (75 MHz, DMSO-d6) δ (ppm) 197.18, 184.79, 147.90, 142.01, 138.27, 121.04, 113.21, 113.10, 102.50, 70.28; MS (ESI) m/z: 176.1 [M+H]+. |
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate for 0.5h;
Stage #2: 5-methoxylindole at 20℃; for 1h; |
4.1.3 General procedure for the synthesis of indole-3-carboxaldehydes (20)
General procedure: A round-bottomed flask containing (28mL, 370mmol) freshly distilled dimethylformamide (DMF) was cooled to 0°C for about 30min and freshly distilled phosphorus oxychloride (8.41mL, 90mmol) was subsequently added with stirring to the DMF over a period of 30min. A solution of indole 19 (85.47mmol) in DMF (10mL, 130mmol) was added to the yellow solution over a period of 1h. The solution was stirred at room temperature till it become a yellow paste. At the end of the reaction, 30g of crushed ice was added to the paste with stirring to make a clear cherry-red aqueous solution. Sodium hydroxide solution (1N, 100mL) was added dropwise with stirring to this cherry-red solution. The resulting suspension was heated rapidly to 90°C and allowed to cool at room temperature, after which it was refrigerated for overnight. The product was filtered, washed with water (2×100mL) and air-dried to afford pure indole-3-carboxaldehydes 20 in 80-90% yields. Indole-3-carboxaldehyde, pale yellow solid; mp 194-196°C (Lit. [37]196-197°C). |
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.25h; Inert atmosphere;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0 - 20℃; for 3.25h; Inert atmosphere;
Stage #3: With sodium hydroxide In water; N,N-dimethyl-formamide for 0.0833333h; Reflux; |
|
|
With trichlorophosphate at 20℃; for 8h; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 0℃; for 2h; |
|
|
With trichlorophosphate at 0℃; for 2h; |
4.1.2. General procedure for synthesis of 1H-indole-3-carboxaldehyde (2a-c)
General procedure: The relevant indole derivate (1a-c, 40 mmol) and phosphorusoxychloride (10 mL) were dissolved into DMF (20 mL), respectively.The solution of indole was added dropwise to POCl3 at 0 °C andstirred for 2 h. Then the reaction mixturewas diluted with icewater(300 mL), neutralized with NaOH to pH 9.0, and extracted withethyl acetate (50 mL x 3). The organic layer was washed with brine,dried over anhydrous sodium sulfate, filtered and concentrated invacuum. Recrystallization afforded compound 2a-c with yields of95.3-98.1%. |
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 20℃;
Stage #2: With water; sodium hydroxide at 0 - 90℃; for 2h; |
|
|
With trichlorophosphate at 0℃; for 0.166667h; |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate Sealed tube;
Stage #2: With potassium hydroxide In water Sealed tube; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.5h;
Stage #2: 5-methoxylindole In N,N-dimethyl-formamide at 20℃; for 3h; |
4.2.1. Synthesis of compound 3
General procedure: POCl3 (13.09 g, 85.36 mmol) was slowly added dropwise to DMF(10 mL) under ice bath. The mixture was stirred at ice bath for30 min. Then, DMF (6.24 g, 85.36 mmol) solution of indole (5 g,42.68 mmol) was added dropwise to the reaction system. Keepingthe reaction at room temperature for 3 h, ice water and 10% NaOHaq. were added into the reaction system to pH of 7-8 and continue to stir to a lot of white solid precipitation. The solid was recrystallizedfrom ethyl acetate and petroleum ether to obtain compound3 in yields of 97.6%. |
|
With trichlorophosphate at 0 - 60℃; for 6.5h; |
General synthetic procedure for the synthesis of indol-3-aldehydes (E)
General procedure: A solution of indole (1.71 mmol) in dimethyl formamide (1 ml) was added to a cooled solutionof trichlorophosphate (1.88 mmol) in dimethyl formamide (1 ml) and stirred at room temperature for30 min and allowed to stir for 6 h at 60 °C. The reaction mixture was allowed for cooling and pouredinto ice-cold water and added dropwise to an ice-cold solution of 2M NaOH. The suspension wasextracted with ethylacetate, the combined organic extract was concentrated to dryness to yield desiredindole-3-carboxaldehyde. |
|
With trichlorophosphate |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 40℃; for 2.5h;
Stage #2: With sodium hydroxide In water at 90℃; for 1h; |
|
|
Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃;
Stage #2: 5-methoxylindole for 1h; Heating; |
|
|
With trichlorophosphate at 5 - 20℃; for 12h; |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 40℃; for 2h;
Stage #2: With sodium hydroxide In water at 90℃; for 1h; |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate at 0 - 40℃; for 2.5h; Inert atmosphere;
Stage #2: With water; sodium hydroxide at 90℃; for 1h; Inert atmosphere; |
|
|
With trichlorophosphate at 0 - 20℃; for 2h; Inert atmosphere; |
|
|
With trichlorophosphate |
|
|
Stage #1: 5-methoxylindole; N,N-dimethyl-formamide With trichlorophosphate Inert atmosphere;
Stage #2: With potassium hydroxide In water Inert atmosphere; |
|

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 110K+ Compounds
110K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping